Posterior Alpha Power in Memory Encoding: Mechanisms, Biomarkers, and Clinical Applications
This article synthesizes current research on the role of EEG posterior alpha power in memory encoding, a key oscillatory pattern for understanding cognitive filtering and neural efficiency.
Posterior Alpha Power in Memory Encoding: Mechanisms, Biomarkers, and Clinical Applications
Abstract
This article synthesizes current research on the role of EEG posterior alpha power in memory encoding, a key oscillatory pattern for understanding cognitive filtering and neural efficiency. We explore its foundational mechanism in actively inhibiting task-irrelevant sensory regions to facilitate long-term memory formation. Methodological sections detail how alpha power predicts subsequent memory performance and serves as a biomarker in classifier models. The discussion extends to its alteration in clinical populations like ADHD and PTSD, and comparative analyses with other spectral features like gamma and theta activity. This resource is tailored for researchers, neuroscientists, and drug development professionals seeking to leverage EEG biomarkers for diagnosing cognitive disorders and evaluating therapeutic interventions.
The Inhibitory Gatekeeper: How Posterior Alpha Oscillations Control Memory Encoding
Posterior alpha power refers to the oscillatory activity in the 8-12 Hz frequency band, typically measured over the occipital and parietal cortices. This neural rhythm represents one of the most prominent features of the human electroencephalogram (EEG) and serves as a fundamental mechanism for regulating information processing in the brain. Within the context of memory encoding research, posterior alpha activity has emerged as a crucial gating mechanism that controls the flow of information into long-term storage by inhibiting task-irrelevant sensory input and facilitating internal cognitive processes. This application note provides a comprehensive overview of the neural origins, functional significance, and methodological protocols for investigating posterior alpha power in cognitive neuroscience research, with particular emphasis on memory encoding paradigms.
Neural Origins and Mechanisms
The neural generators of posterior alpha power originate from thalamocortical circuits connecting the thalamus with parietal and occipital cortical regions. Research using source localization techniques has identified two spatially distinct alpha sources that fulfill different functional roles:
- A sensory (visual) alpha source in occipital regions that reflects the "visual spotlight of attention" [1]
- A parietal alpha source modulated by attentional effort, showing lowest alpha power when attention was divided between modalities or spatial locations [1]
The alpha oscillation is generated from postsynaptic potentials in a cingulo-insular-thalamic network associated with attentional alertness, comprising the dorsal anterior cingulate cortex, anterior insula, anterior prefrontal cortex, and thalamus [2]. This network maintains tonic internalized alertness and correlates with fluctuations in involuntary attention.
Neurophysiological Basis
From a neurophysiological perspective, alpha oscillations typically reflect a state of cortical inhibition or disengagement of task-irrelevant regions. The prevailing model suggests that increased alpha power reflects decreased neuronal activity in the regions where it occurs, functioning as an active inhibitory mechanism [3] [4]. This inhibitory function is mediated through GABAergic neurotransmission, which is essential for generating alpha oscillations [3]. The thalamocortical dysrhythmia model of neuropsychiatric disorders further suggests that disruptions in normal oscillatory interactions between the thalamus and cortex can lead to excessive synchronized alpha rhythms [3].
Table 1: Neural Generators and Functional Roles of Posterior Alpha Power
| Anatomical Region | Functional Role | Experimental Paradigm |
|---|---|---|
| Occipital cortex | Sensory gating and visual attention | Visuospatial attention tasks [1] |
| Parietal cortex | Attentional effort and task difficulty modulation | Cross-modal attention division [1] |
| Temporoparietal junction | Inhibition of ventral attention network | Internal attention and mental imagery [5] |
| Thalamocortical circuits | Overall rhythmic generation and regulation | Resting-state EEG [2] |
Functional Significance in Cognitive Processing
Attention and Perceptual Gating
Posterior alpha power plays a critical role in attention modulation through power-dependent inhibition of sensory processing. The functional architecture involves region-specific changes where increased alpha power actively inhibits processing in task-irrelevant regions while decreased alpha power facilitates processing in task-relevant areas [1] [4]. This mechanism implements a sensory selection filter that gates information flow based on behavioral relevance.
In focused versus divided attention paradigms, prestimulus alpha power in visual areas decreases linearly with increasing attention to visual stimuli, whereas parietal alpha power is lower when attention is divided between modalities/hemifields compared with focused attention [1]. This demonstrates the dual mechanism of posterior alpha in implementing both sensory-specific attention and overall attentional resource allocation.
Memory Encoding and Cognitive Control
Research has established that alpha power increases during successful memory encoding reflect active inhibition of irrelevant information. In directed memory paradigms, posterior alpha activity increases prior to "No-Remember" cues while decreasing prior to "Remember" cues, with sources localized in the parietal cortex included in the dorsal attention network [4]. Subjects with larger cue-modulation of alpha activity demonstrate better memory for to-be-remembered items, indicating that inhibitory alpha mechanisms support memory efficiency.
During long-term memory formation, increased posterior alpha power during rehearsal periods creates an optimal brain state for successful encoding by actively reducing parieto-occipital activity that might interfere with memory consolidation [6]. This functional inhibition of sensory regions facilitates internal cognitive processes necessary for memory formation.
Table 2: Functional Roles of Posterior Alpha Power Across Cognitive Domains
| Cognitive Domain | Alpha Power Increase | Alpha Power Decrease |
|---|---|---|
| Attention | Inhibition of distracting sensory input [1] | Enhanced processing of relevant stimuli [1] |
| Memory Encoding | Blocking of irrelevant memories [4] | Facilitation of target encoding [6] |
| Working Memory | Information maintenance during retention [7] | Enhanced processing during encoding [7] |
| Emotional Processing | Regulation of fear circuitry [3] | Enhanced sensory processing of emotional stimuli |
Experimental Protocols and Methodologies
Standardized EEG Acquisition Parameters
For consistent measurement of posterior alpha power, the following acquisition parameters are recommended:
- Electrode Placement: International 10-20 system, with focus on O1, O2, P3, P4, Pz, T5, T6
- Reference Scheme: Linked ears or average reference; CSD transformation for improved localization [8]
- Sampling Rate: Minimum 500 Hz to adequately capture alpha morphology
- Filter Settings: High-pass at 0.1 Hz, low-pass at 100 Hz, notch filter at 50/60 Hz
- Impedance Criteria: Maintain below 5 kΩ for all electrodes
- Recording Conditions: Eyes-open (EO) and eyes-closed (EC) resting state, plus task conditions
Memory Encoding Paradigm
The following protocol assesses posterior alpha's role in memory encoding, adapted from successful implementations [6] [4]:
Stimuli and Apparatus:
- 640 real-life photographs of landscapes and buildings
- Presentation software (e.g., STIM2, Psychophysics Toolbox)
- EEG recording system with 64+ channels
- Response recording device
Procedure:
- Cue Period (2s): colored fixation cross indicates "Remember" (blue) or "No-Remember" (yellow)
- Stimulus Presentation (1s): picture display according to cue direction
- Inter-trial Interval (1s): blank screen
- Encoding Session: 440 trials (220 per condition), randomized
- Interference Task (5min): arithmetic equations to reduce recency effects
- Recognition Session: all encoded pictures plus 200 new foils, randomized
EEG Processing Pipeline:
- Preprocessing: filtering, artifact removal, bad channel interpolation
- Current Source Density (CSD) transformation to minimize volume conduction [8]
- Time-frequency analysis using Morlet wavelets or similar approach
- Frequency Principal Components Analysis (fPCA) for alpha quantification [8]
- Statistical analysis of alpha power during cue and encoding periods
Data Analysis and Quantification
Alpha Power Quantification:
- Frequency range: 8-12 Hz (individual alpha peak ±2 Hz)
- Time windows: -1 to 0s pre-stimulus, 0.5-1.5s post-stimulus
- Baseline correction: -1.5 to -0.5s pre-cue
- Regional masks: occipital (O1, O2, Oz), parietal (P3, P4, Pz), temporal-posterior (T5, T6)
Statistical Approaches:
- Repeated measures ANOVA with factors Condition (Remember/No-Remember) and Region
- Correlation analyses between alpha modulation and memory performance
- Source localization using L2-MNE or beamformer approaches
Signaling Pathways and Neural Networks
The following diagram illustrates the neural pathways and functional networks involved in posterior alpha generation and modulation:
Diagram 1: Neural Pathways of Posterior Alpha Generation and Modulation
Research Reagent Solutions and Materials
Table 3: Essential Research Materials for Posterior Alpha Investigations
| Category | Specific Product/Model | Function/Application |
|---|---|---|
| EEG Systems | 64+ channel EEG with active electrodes (e.g., BioSemi, BrainAmp) | High-quality signal acquisition for source localization |
| Stimulation Software | Psychophysics Toolbox, Presentation, STIM2 | Precise stimulus presentation and timing |
| Analysis Tools | EEGLAB, FieldTrip, Brainstorm, BESA | Signal processing and source analysis |
| CSD Toolbox | CSD Toolbox for MATLAB | Current Source Density transformation [8] |
| Source Modeling | SPM, MNE, sLORETA | Spatial localization of alpha generators |
| Frequency Analysis | Chronux Toolkit, custom wavelet scripts | Time-frequency decomposition |
| Statistical Packages | R, SPSS, MATLAB Statistics Toolbox | Quantitative analysis of alpha-power effects |
Applications in Clinical and Cognitive Research
Biomarker Potential
Posterior alpha power demonstrates significant potential as a neurophysiological biomarker for various clinical conditions:
- Depression: Elevated posterior alpha power is associated with depression and clinical response to antidepressants [8] [9]
- Trauma Disorders: Complex childhood trauma exposure is reflected in reduced temporal-posterior alpha power [3]
- Cognitive Assessment: Individual differences in alpha suppression correlate with working memory capacity [7]
Neuroregulation Applications
The robust functional significance of posterior alpha power makes it an ideal target for neurofeedback protocols and neuromodulation approaches. Research indicates that baseline resting-state alpha power can predict individual responses to interventions such as transcranial direct current stimulation (tDCS) [10], enabling personalized treatment approaches.
Posterior alpha power represents a fundamental neural mechanism for cognitive control, serving as a gating mechanism that regulates information flow to support optimal cognitive functioning. Its dual origin in sensory-specific and attention-control networks enables both focused perceptual inhibition and overall resource allocation. In memory encoding research, posterior alpha activity provides a crucial window into the neural mechanisms underlying successful information filtering and consolidation. The standardized protocols and analytical approaches outlined in this application note provide researchers with robust methodologies for investigating this important neural rhythm across basic and clinical research domains.
The Active Inhibition Hypothesis (AIH) posits that oscillatory alpha activity (~8-13 Hz) serves as an active gating mechanism that suppresses interference from task-irrelevant brain regions during cognitive processing [11] [4]. This framework redefines alpha oscillations from a mere idling rhythm to an active functional mechanism for resource allocation. Within memory encoding research, the AIH provides a parsimonious explanation for a seemingly paradoxical finding: increased posterior alpha power during successful memory formation, despite the visual nature of most memoranda. This suggests that inhibiting parieto-occipital areas mitigates potential sensory interference, thereby optimizing conditions for mnemonic processing in relevant networks [11] [6]. These insights are particularly valuable for drug development professionals targeting cognitive deficits, as the alpha rhythm presents a quantifiable electrophysiological biomarker for assessing therapeutic efficacy on network-level cognitive control.
Key Experimental Evidence
Empirical support for the AIH stems from studies demonstrating that alpha power in parieto-occipital cortex parametrically increases with cognitive demand and predicts subsequent behavioral performance.
Table 1: Key Experimental Findings Supporting the Active Inhibition Hypothesis
| Study Paradigm | Key Finding | Cognitive Interpretation | Source Location |
|---|---|---|---|
| Working Memory (Faces) | Parametric increase in alpha power with memory load during retention. | Higher load requires stronger inhibition of sensory input to protect maintenance. | Parieto-occipital sulcus [11] |
| Cued Long-Term Memory | "No-Remember" cues triggered alpha increase; "Remember" cues triggered alpha decrease. | Anticipatory alpha reflects top-down inhibition to gate irrelevant information. | Parietal cortex (Dorsal Attention Network) [4] |
| Sequence Rehearsal (LTM) | Higher alpha power during rehearsal predicted successful long-term memory formation. | Inhibition of posterior regions creates an optimal brain state for encoding. | Parieto-occipital regions [6] |
Table 2: Quantitative Summary of Alpha Power Modulations Across Studies
| Study Paradigm | Frequency Band | Power Change for Successful Inhibition/Encoding | Localization Method |
|---|---|---|---|
| Working Memory (Faces) | ~10.5 Hz | Significantly higher power with increasing memory load (1-4 faces). | Beamforming (DICS) coregistered with individual MRIs [11] |
| Cued Long-Term Memory | Alpha Band | Increase for "No-Remember" vs. decrease for "Remember" cues. | MEG source reconstruction [4] |
| Sequence Rehearsal (LTM) | 8-12 Hz | Markedly stronger for successfully vs. unsuccessfully encoded sequences. | MEG source analysis [6] |
The following diagram illustrates the core proposed mechanism of the Active Inhibition Hypothesis in a memory task context.
Detailed Experimental Protocols
This section provides detailed methodologies for key experiments that underpin the AIH, enabling replication and application in preclinical drug development.
Protocol: Investigating Alpha Power in Working Memory Retention
This protocol is adapted from a magnetoencephalography (MEG) study that identified the parieto-occipital sulcus as a key source of load-dependent alpha activity [11].
- Objective: To identify the neuronal sources of alpha activity that parametrically increase with working memory load during the retention interval.
- Subjects: 5 right-handed male subjects, age 23-26 years. Note: Future replications should include larger and more diverse cohorts.
- Task Design: A modified Sternberg task with faces as stimuli.
- Memory Load: Lists of 1, 2, 3, or 4 faces presented sequentially.
- Trial Structure:
- Encoding: Faces presented for 0.3 s each with 1.25 s intervals.
- Retention: A 2.7 s delay period.
- Probe: A single face is presented; subjects indicate if it was in the memory list.
- Data Acquisition:
- MEG: Recorded using a 151-sensor whole-head axial gradiometer system.
- Sampling: Low-pass filtered at 200 Hz, sampled at 600 Hz.
- Ancillary Recordings: Vertical electro-oculogram (EOG) for artifact identification.
- Co-registration: Head position coils aligned to individual MRIs.
- Data Analysis:
- Preprocessing: Exclude incorrect and artifact-contaminated trials.
- Time-Frequency Analysis: Compute spectrograms using Slepian tapers (2 Hz smoothing) to quantify power in the alpha band (8-13 Hz).
- Source Reconstruction: Apply a beamforming technique (Dynamical Imaging of Coherent Sources, DICS) to localize the load-dependent alpha power to anatomical images.
- Key Outcome Measure: The regression coefficient (slope) of alpha power in the retention interval against memory load, localized to the parieto-occipital sulcus.
Protocol: Testing Anticipatory Alpha in Gating Long-Term Memory
This protocol uses a cued memory paradigm to demonstrate how anticipatory alpha activity gates information into long-term memory [4].
- Objective: To examine how cue-directed alpha power modulation suppresses the encoding of irrelevant information.
- Subjects: 23 healthy, right-handed volunteers (11 males, 12 females, mean age 24.8 ± 3.1 years).
- Task Design: A cued long-term memory paradigm with "Remember" (R) and "No-Remember" (NR) instructions.
- Trial Structure:
- Cue Period (2 s): A colored fixation cross indicates the R or NR instruction.
- Item Presentation (1 s): A picture of a landscape or building is shown.
- Inter-Trial Interval (1 s).
- Stimuli: 640 real-life photographs, counterbalanced across conditions.
- Recognition Session: Old (both R and NR) and new pictures are presented; subjects make an old/new judgment.
- Trial Structure:
- Data Acquisition:
- MEG: Recorded during the encoding session.
- Analysis Epochs: Time-locked to the cue and item presentation.
- Data Analysis:
- Time-Frequency Analysis: Extract oscillatory power in the alpha band (8-13 Hz) following the cue.
- Source Localization: Reconstruct the neural generators of the cue-modulated alpha activity.
- Correlation Analysis: Relate the degree of cue-modulated alpha power to subsequent memory performance.
- Key Outcome Measures:
- Higher posterior alpha power following "No-Remember" vs. "Remember" cues.
- A positive correlation between the cue-induced alpha modulation and memory performance for R items.
The workflow for a typical MEG experiment investigating the AIH is summarized below.
The Scientist's Toolkit
Table 3: Research Reagent Solutions for Active Inhibition Research
| Item | Function/Description | Example Use Case |
|---|---|---|
| Whole-Head MEG System | Records magnetic fields generated by neuronal activity with high temporal resolution. | Core activity recording in all featured protocols [11] [4] [6]. |
| Structural MRI | Provides individual anatomical data for precise source localization of MEG/EEG signals. | Co-registration with MEG data to identify sources in parieto-occipital sulcus [11]. |
| Beamforming (e.g., DICS) | Spatial filtering algorithm for source reconstruction of oscillatory activity. | Localizing memory-load dependent alpha sources [11]. |
| High-Dimensional Statistical Framework | Addresses challenges of high dimensionality and temporal dependency in EEG/MEG data. | Identifying significant channels and testing changes in mean power [12]. |
| Sternberg/WM Paradigm | Classic working memory task with controlled encoding, retention, and probe periods. | Testing parametric effects of memory load on posterior alpha power [11]. |
| Cued Encoding Paradigm | Task with explicit instructions to remember or ignore subsequent items. | Testing top-down anticipatory inhibition via alpha rhythms [4]. |
Considerations and Future Directions
While the AIH is strongly supported by empirical evidence, a complete interpretation of alpha dynamics must also account for inter-individual variability. Some studies report that individuals can differ consistently in the direction of alpha power modulation associated with improved performance, suggesting different cognitive strategies may employ distinct neurophysiological pathways [13]. Furthermore, the brain operates as a system of dynamic networks. The Default Mode Network (DMN), active during rest and internal mentation, shows a reciprocal relationship with task-positive networks like the Executive Control Network (ECN). The AIH aligns with this framework, where increased alpha in the parieto-occipital cortex may facilitate a network switch away from the DMN's internal focus toward the ECN, supporting active maintenance and shielding from distraction [14] [15]. For drug development, this network perspective is critical, as therapeutics aimed at cognitive enhancement may exert effects by modulating the dynamic interplay between large-scale networks, with posterior alpha power serving as a key measurable outcome.
Within the framework of a broader thesis on EEG posterior alpha power in memory encoding research, a convergent body of evidence underscores the critical, albeit counterintuitive, role of high alpha oscillations (8-12 Hz) in facilitating long-term memory (LTM) formation. Contemporary neuroscience has moved beyond viewing alpha activity merely as a rhythm of an idling brain. Instead, it is recognized as an active inhibitory mechanism that suppresses processing in task-irrelevant regions, thereby optimizing cognitive function [6]. Subsequent memory paradigms, which contrast neural activity during the encoding of successfully versus unsuccessfully remembered items, provide direct evidence for this theory. Findings reveal that successful LTM formation is consistently associated with elevated posterior alpha power during encoding and rehearsal periods. This alpha-mediated suppression is hypothesized to minimize interference from parietal and occipital brain regions not directly involved in the memory task, creating an optimal brain state for the medial temporal lobe and prefrontal cortex to consolidate new information [6]. This application note synthesizes key evidence and provides detailed protocols for investigating this phenomenon.
Key Evidence and Quantitative Data Summaries
The following table consolidates quantitative and methodological findings from pivotal studies linking posterior alpha power to LTM success.
Table 1: Key Experimental Findings on Alpha Power and LTM Formation
| Study Paradigm | Key Finding on Successful LTM | Neural Metric & Location | Experimental Task |
|---|---|---|---|
| Word Sequence Encoding & Rehearsal [6] | Markedly stronger alpha power during rehearsal | Alpha power (8-12 Hz); Parieto-occipital cortex | Encode/maintain order of 3 words; 3.4s rehearsal interval |
| Post-Learning Rest [16] | Decrease in alpha power from baseline | Alpha power change; Cortical sources | Resting EEG after learning word pairs; 7-day retention |
| Trial-Level Alpha & Discourse Focus [17] | Pre-target alpha modulated subsequent memory effect | Pre-target alpha power; Centro-parietal sites | Spoken language comprehension; memory for focused words |
The Researcher's Toolkit: Essential Materials & Reagents
Table 2: Essential Research Reagents and Solutions for Alpha-LTM Studies
| Item Category | Specific Example & Function | Research Application |
|---|---|---|
| Neuroimaging Hardware | 275-sensor axial gradiometer MEG system (e.g., VSM/CTF); EEG systems with >64 channels | Records neuromagnetic/electrical brain activity with high temporal resolution [6]. |
| Data Acquisition Software | Custom MEG/EEG acquisition software (e.g., CTF software); Presentation/PsychoPy | Presents experimental stimuli and records synchronized neural data [6]. |
| Physiological Monitoring | Bipolar electrodes for Electrooculogram (EOG) and Electrocardiogram (ECG) | Monitors and artifacts from eye blinks and cardiac activity [6]. |
| Stimulus Material | High-frequency concrete nouns (e.g., from Celex database); word pairs; spoken discourse narratives | Provides standardized, ecologically valid verbal material for memory encoding [6] [17]. |
| Analysis Tools | MATLAB with toolboxes (e.g., FieldTrip, EEGLAB); Python (MNE-Python); Mixed-effects models in R | Processes MEG/EEG data, computes time-frequency representations, and performs statistical analysis [6] [17]. |
Detailed Experimental Protocols
Protocol 1: MEG Investigation of Alpha during Active Rehearsal
This protocol is adapted from the seminal study by [6] and is designed to isolate alpha activity during the rehearsal period.
3.1.1 Participant Preparation and Setup
- Participants: Recruit 20-25 right-handed, native-language speakers with no history of neurological or psychiatric disorders. Obtain written informed consent.
- MEG Setup: Position the participant in a supine position within the magnetically shielded room. Use a 275-sensor axial gradiometer MEG system. Recordings should be low-pass filtered at ~250 Hz and sampled at 1200 Hz.
- Auxiliary Recordings: Attach bipolar electrodes to record Electrooculogram (EOG) for eye movement and Electrocardiogram (ECG). Place three head position indicator coils (nasion, left and right ear canals) to track head position throughout the recording.
3.1.2 Stimulus and Task Design
- Stimuli: Utilize a large set (>2000) of high-frequency, concrete nouns. Match sequences for word frequency and ensure each word is presented only once.
- Procedure: The task consists of multiple blocks, each with two critical parts:
- LTM Trials: Instruct participants to "encode the order of three words in long-term memory." Present three words sequentially (0.6 s/word), followed by a 3.4 s rehearsal interval. Encourage subvocal sentence generation to link the words.
- WM Trials: Instruct participants to maintain word order in working memory for an immediate test. This contrasts the LTM condition.
- Memory Test: After a delay, present a retrieval test where participants reconstruct the order of the words from the LTM trials using button presses. Include catch trials (e.g., a word from another sequence) to ensure full sequence encoding.
3.1.3 Data Analysis Pipeline
- Preprocessing: Filter data, remove artifacts related to eye blinks and heartbeats using EOG/ECG recordings, and segment data into epochs around word presentation and rehearsal intervals.
- Time-Frequency Analysis: Calculate power spectral density for each epoch. Focus on the alpha band (8-12 Hz). Transform data into the time-frequency domain using methods like Morlet wavelets.
- Source Reconstruction: Use beamformer techniques (e.g., Synthetic Aperture Magnetometry) to localize the neuronal sources of the alpha activity, focusing on parieto-occipital regions.
- Subsequent Memory Contrast: Sort trials based on retrieval success (Remembered vs. Forgotten). Perform a statistical comparison (e.g., cluster-based permutation test) of alpha power during the rehearsal interval between these conditions.
Protocol 2: EEG and the Subsequent Memory Effect in Language
This protocol, based on [17], investigates how trial-level fluctuations in alpha power predict memory for spoken language.
3.2.1 Participant and EEG Setup
- EEG Recording: Use a high-density EEG system (e.g., 64-128 channels). Impedances should be kept below 10 kΩ. The sampling rate should be at least 500 Hz.
- Stimuli: Develop spoken discourse narratives where critical target words are either focused or defocused by the preceding context (e.g., using wh-questions).
3.2.2 Task Procedure
- Participants listen to the spoken narratives.
- Following the narrative, administer a surprise recognition or recall test for the target words to measure subsequent memory.
3.2.3 Data Analysis Focus
- Pre-Target Alpha: Segment EEG data into epochs immediately preceding the onset of critical target words.
- Alpha Power Extraction: Calculate alpha power for each epoch at centro-parietal electrode sites.
- Regression Analysis: Use mixed-effects models to determine if trial-by-trial fluctuations in pre-target alpha power predict the likelihood of subsequent memory for the target word, and whether this relationship is modulated by discourse focus.
Visualizing Workflows and Theoretical Models
Theoretical Model of Alpha-Mediated Memory Formation
The following diagram illustrates the proposed theoretical framework linking high posterior alpha power to successful long-term memory formation.
Experimental Workflow for an MEG Subsequent Memory Study
This flowchart outlines the end-to-end experimental procedure for a typical MEG study investigating the alpha-LTM link, as detailed in Protocol 3.1.
Electroencephalographic (EEG) posterior alpha oscillations (8-14 Hz) serve as a fundamental neural mechanism for cognitive control during memory tasks, operating through the functional inhibition of task-irrelevant cortical regions [18] [6]. The temporal dynamics of alpha power—specifically the contrast between stimulus processing and internal rehearsal intervals—provide critical insights into how the brain prioritizes and manages cognitive resources. During memory encoding, alpha power modulations reflect a delicate balance between external information processing and internal maintenance operations [18] [19]. Research consistently demonstrates that posterior alpha power decreases during stimulus presentation to facilitate perceptual processing, while increasing during rehearsal intervals to protect internal cognitive operations from potential interference [6]. This application note synthesizes current experimental evidence and methodologies for investigating these temporal dynamics, with particular emphasis on their implications for memory encoding research and pharmacological interventions targeting cognitive function.
Quantitative Synthesis of Alpha Power Modulations
Table 1: Temporal Dynamics of Posterior Alpha Power During Memory Phases
| Memory Phase | Alpha Power Change | Functional Interpretation | Typical Magnitude | Topography |
|---|---|---|---|---|
| Stimulus Presentation | Decrease | Gating of sensory input, facilitated perceptual processing | Lower during word presentation vs. rehearsal [6] | Parieto-occipital dominance |
| Rehearsal/Maintenance | Increase | Active inhibition of interfering visual input, internal focus | ~20-30% increase for successful LTM formation [6] | Posterior dominance |
| Retrospective Cueing | Lateralization | Spatiotopic access to WM representations | Contralateral to cued item [18] | Hemisphere-specific |
| Prospective Cueing | Lateralization | Anticipatory attention to upcoming stimuli | Contralateral to anticipated location [18] | Hemisphere-specific |
| Verbal WM Retention | Variable by presentation mode | Depends on temporal encoding structure | Below baseline in Fast+delay mode [19] | Posterior regions |
Table 2: Factors Influencing Alpha Modulation Patterns
| Factor | Impact on Alpha Power | Experimental Evidence |
|---|---|---|
| Stimulus Modality | Verbal vs. visual stimuli show different alpha topography | Left temporal alpha for verbal rehearsal [20] |
| Task Demands | Higher cognitive load increases posterior alpha during maintenance | Alpha power predicts successful LTM formation [6] |
| Temporal Structure | Simultaneous vs. sequential presentation affects alpha dynamics | Fast+delay mode shows unique below-baseline alpha [19] |
| Instruction Type | Pre-cues vs. retro-cues elicit different alpha lateralization | Retrocues enable spatiotopic access to WM [18] |
| Stimulus Semantics | Meaningful vs. abstract stimuli affect rehearsal efficacy | Phase-scrambled scenes benefit more from rehearsal [20] |
Experimental Protocols for Alpha Dynamics Investigation
Paired-Associates Memory Paradigm with Cued Instruction
Objective: To directly compare neural mechanisms of anticipatory and retrospective attention shifts during working memory [18].
Materials:
- 128-channel EEG system with active electrodes
- Stimulus presentation software (e.g., PsychToolbox, E-Prime)
- 2+2 visual memory arrays (e.g., oriented gratings, shapes)
- Spatial cues (central arrows indicating left/right locations)
Procedure:
- Initial Fixation: Display cross-hair for 500ms
- First Memory Array: Present two visual items to left/right of fixation for 500ms
- Delay Period: Maintain fixation for 1500ms
- Instruction Cue: Present spatial arrow for 300ms
- Pre-cue condition: Cue indicates which item from UPCOMING second array is relevant
- Retro-cue condition: Cue indicates which item from PREVIOUS first array is relevant
- Second Memory Array: Present two new visual items for 500ms (pre-cue) or blank screen (retro-cue)
- Delay Period: Maintain fixation for 1500ms
- Probe: Test memory for cued item with match/mismatch judgment
- Trial Structure: Equate stimulus timing and sequence across conditions
EEG Acquisition Parameters:
- Sampling rate: 500-1000 Hz
- Filter settings: 0.1-100 Hz bandpass, 50/60 Hz notch filter
- Reference: Common average or mastoid reference
- Electrode placement: International 10-20 system, extended to 64+ channels
Verbal Working Memory Task with Controlled Rehearsal
Objective: To investigate how maintenance strategy and stimulus presentation mode affect alpha dynamics during verbal working memory [20] [19].
Materials:
- MEG/EEG recording system
- Verbal stimuli (digits, words, or pseudowords)
- Strategy instruction prompts (rehearsal vs. suppression)
Procedure:
- Encoding Phase:
- Simultaneous condition: Present all stimuli (e.g., 7 digits) for 2800ms
- Sequential conditions: Present stimuli one at a time with varying rates
- Fast: 400ms per digit
- Slow: 1000ms per digit
- Fast+delay: 400ms per digit + 600ms free time
- Rehearsal Instruction:
- Rehearsal condition: Generate descriptive label and covertly rehearse
- Suppression condition: Repeat "the" continuously to prevent rehearsal
- Retention Interval: 6-second maintenance period with fixation
- Retrieval Phase: Serial recall of digits in correct order
- Strategy Verification: Post-trial reporting of strategy adherence
EEG Processing Steps:
- Preprocessing: Bad channel rejection, ocular correction, filtering
- Epoching: Lock to stimulus onset and rehearsal period separately
- Spectral analysis: Time-frequency decomposition using Morlet wavelets
- Alpha quantification: Mean power in 8-13Hz band at posterior electrodes
Figure 1: Experimental workflow for comparing pre-cue and retro-cue conditions in working memory task
The Scientist's Toolkit: Essential Research Reagents and Equipment
Table 3: Core Methodological Resources for Alpha Dynamics Research
| Category | Specific Tool/Resource | Function/Application | Key Considerations |
|---|---|---|---|
| EEG Systems | 128-channel Geodesic HydroCel System | High-density spatial sampling | Adequate for source localization of posterior alpha |
| Stimulus Software | PsychToolbox, E-Prime, Presentation | Precise timing control | Millisecond accuracy critical for temporal dynamics |
| Analysis Platforms | EEGLAB, FieldTrip, MNE-Python | Time-frequency analysis | Custom scripts for alpha lateralization metrics |
| Verbal Stimuli | CELEX Database, MRC Database | Controlled word characteristics | Word frequency, concreteness, imageability |
| Visual Stimuli | Oriented gratings, complex shapes | Visual working memory tasks | Control for luminance, contrast, complexity |
| Experimental Control | Articulatory suppression tasks | Prevent verbal rehearsal | "The" repetition for standard suppression |
Signaling Pathways and Neural Mechanisms
Figure 2: Neural mechanisms of alpha modulation during external processing versus internal rehearsal
Discussion and Research Applications
The temporal dynamics of posterior alpha power between stimulus presentation and rehearsal intervals represent a fundamental neural mechanism for cognitive control in memory processes. The robust finding that posterior alpha power increases during successful rehearsal for long-term memory formation [6] highlights its potential as a biomarker for memory efficacy. Furthermore, the differential effects of stimulus presentation mode on alpha dynamics [19] underscore the importance of methodological considerations in experimental design.
For pharmaceutical researchers, these alpha dynamics offer promising avenues for evaluating cognitive-enhancing compounds. The quantitative metrics provided in this application note can serve as sensitive endpoints for clinical trials targeting memory disorders. The experimental protocols establish standardized methodologies for assessing drug effects on specific memory phases, potentially revealing compound-specific mechanisms of action through their distinct effects on stimulus-processing versus rehearsal-related alpha modulation.
Future research directions should explore cross-species validation of these alpha dynamics to strengthen their translational relevance, investigate pharmacological modulation of these oscillations using compounds with known cognitive effects, and develop individualized alpha modulation profiles as potential biomarkers for cognitive disorders. The integration of these temporal alpha dynamics with other neurophysiological measures and behavioral outcomes will further enhance their utility in both basic memory research and applied pharmaceutical development.
Application Notes
This document details the application of source localization techniques to investigate the role of the Dorsal Attention Network (DAN), particularly the parietal cortex, in modulating posterior alpha oscillations for memory encoding. Converging evidence from magnetoencephalography (MEG) and electroencephalography (EEG) studies indicates that top-down controlled alpha activity in the DAN serves as a gating mechanism, inhibiting the processing of irrelevant information and thereby facilitating the encoding of relevant memories into long-term storage [4] [21] [22].
Key Functional Mechanisms
The parietal cortex, as a core node of the DAN, exhibits anticipatory alpha power changes that predict subsequent memory performance. This alpha modulation functionally inhibits sensory processing, which is reflected in suppressed gamma-band activity, creating a controlled gateway for memory formation [4]. The table below summarizes the direction of these oscillatory changes and their functional interpretations.
Table 1: Oscillatory Activities in the Parietal Cortex/DAN Related to Memory Encoding
| Cognitive Context | Alpha Band (8-13 Hz) Change | Gamma Band (>30 Hz) Change | Functional Interpretation |
|---|---|---|---|
| Cue to Ignore ("No-Remember") | Increase in power [4] [21] | Decrease in power [4] | Active inhibition of irrelevant sensory processing [4] |
| Cue to Remember ("Remember") | Decrease in power (desynchronization) [4] [22] | Increase in power [4] | Facilitation of sensory processing and encoding [4] |
| Attention to Item-Context Binding | Desynchronization in upper alpha band (10-13 Hz) [22] | Not Specified | Allocation of attention to episodic binding [22] |
Behavioral Correlation and Network Dynamics
The efficacy of this alpha-mediated gating mechanism directly correlates with memory performance. Individuals who demonstrate a larger cue-induced modulation of posterior alpha activity subsequently show better memory for the items they were instructed to remember [4] [21]. Furthermore, the anticipatory alpha power during the cue period negatively predicts the gamma power during the subsequent presentation of the memory item, highlighting a direct, mechanistic link between inhibitory alpha rhythms and the facilitatory gamma rhythms associated with detailed stimulus processing [4]. This suggests a model where the DAN uses alpha oscillations to implement top-down attentional control, which in turn gates information into long-term memory by regulating local gamma-synchronized processing [4].
Experimental Protocols
Protocol 1: Cued Memory Encoding with MEG/EEG
This protocol is designed to capture the oscillatory dynamics of the DAN during a directed memory encoding task [4] [21].
Objective: To quantify the pre-stimulus and stimulus-related oscillatory activity in the DAN associated with the intentional encoding versus ignoring of visual information.
Workflow: The following diagram illustrates the sequence and timing of a single trial in the cued memory encoding paradigm.
Procedure:
- Stimuli: Use a large set (e.g., 640) of real-life photographs (landscapes, buildings) to minimize repetition effects [4].
- Trial Structure: Each trial begins with a cue period (e.g., 2000 ms) where a colored fixation cross instructs the participant to either remember or not remember the upcoming picture. The color assignment (e.g., blue for "Remember," yellow for "No-Remember") should be counterbalanced across participants [4].
- Item Presentation: Following the cue, a picture is presented for a short duration (e.g., 1000 ms) [4].
- Task Engagement: To ensure perception, randomly intersperse trials (e.g., 10%) where participants must perform a perceptual decision (e.g., classify the picture as a landscape or building) after the offset of the picture [4].
- Recognition Test: After a delay filled with an interference task (e.g., arithmetic), present the old pictures randomly mixed with new pictures. Instruct participants to indicate whether each picture is "old," "new," or "don't know," regardless of the initial cue [4].
Data Acquisition & Source Analysis:
- Recording: Acquire high-density EEG or MEG data (e.g., 64+ channels for EEG) during the task, following standard procedures for electrode placement (10-20 system) and signal amplification [4] [23].
- Preprocessing: Filter raw data and remove artifacts (e.g., eye movements, muscle activity) using techniques like Independent Component Analysis (ICA) [23].
- Head Model: Create a forward model using individual or template (e.g., MNI) structural MRI to solve the forward problem of estimating scalp potentials from source activities [23].
- Source Localization: Solve the inverse problem to estimate the cortical sources of the recorded signals. Distributed source models (e.g., minimum norm estimate) are often suitable for locating oscillatory activity in the parietal cortex and other DAN regions [23].
- Time-Frequency Analysis: Compute time-frequency representations of source activity for alpha (8-13 Hz) and gamma (>30 Hz) bands. Focus analysis on the cue period and the item presentation period, comparing "Remember" vs. "No-Remember" conditions [4].
Protocol 2: Resting-State fMRI of the DAN
This protocol assesses the intrinsic functional architecture of the DAN, which can predict individual differences in attention and memory performance [24] [25].
Objective: To measure resting-state functional connectivity (FC) within the DAN and correlate it with behavioral performance on spatial attention and memory tasks.
Workflow: The following diagram outlines the core steps for analyzing resting-state fMRI data to investigate DAN connectivity.
Procedure:
- fMRI Acquisition: Acquire T1-weighted structural images and resting-state functional images (e.g., EPI sequence, TR=720ms, 440 volumes) while participants rest with their eyes open, minimizing structured mental activity [24].
- Behavioral Assessment: Administer spatial attention tasks (e.g., Posner cueing paradigm, visual search) outside the scanner to obtain behavioral metrics like disengagement cost (invalid vs. valid cue RT) and visual search speed [24] [25].
Data Analysis:
- Preprocessing: Preprocess fMRI data using standard pipelines (e.g., SPM, FSL) including realignment, coregistration, normalization, and smoothing. Apply band-pass filtering (e.g., 0.01-0.1 Hz) to isolate low-frequency fluctuations [24] [25].
- Seed-Based FC: Define seeds in core DAN regions: the Frontal Eye Fields (FEF) and Intraparietal Sulcus (IPS) in each hemisphere [24] [25].
- Connectivity Calculation: For each seed, extract the mean BOLD time series and compute correlation coefficients with the time series of every other voxel in the brain. Convert these correlations to Fisher's Z-values to create FC maps for each participant [24] [25].
- Correlation with Behavior: Correlate the strength of intra-hemispheric (e.g., left FEF-IPS) and inter-hemispheric (e.g., left FEF-right FEF) FC with the behavioral metrics obtained from the attention tasks [25].
The Scientist's Toolkit
Table 2: Essential Reagents and Materials for DAN and Alpha Oscillation Research
| Item Name | Specification / Example | Primary Function in Research |
|---|---|---|
| High-Density EEG System | 64+ channels, compatible with EEG caps and amplifiers. | Records scalp electrical activity with high temporal resolution to capture neural oscillations like alpha and gamma [23]. |
| MEG System | Whole-head superconducting quantum interference device (SQUID) sensors. | Measures magnetic fields induced by neuronal currents, offering excellent temporal and good spatial resolution for source localization [4]. |
| Structural MRI Scanner | 3T MRI scanner with T1-weighted MPRAGE sequence. | Provides high-resolution anatomical images for constructing individual head models for accurate EEG/MEG source localization [23]. |
| fNIRS System | Continuous-wave, portable fNIRS device with multiple source-detector pairs. | Measures cortical hemodynamic responses as a proxy for neural activity; useful for naturalistic settings and populations like infants [26]. |
| Analysis Software Suite | MATLAB with toolboxes (e.g., EEGLAB, SPM, FieldTrip), Brainstorm, FSL, Freesurfer. | Provides integrated environments for preprocessing, source localization, time-frequency analysis, and statistical analysis of neuroimaging data [23] [27]. |
| Standardized Electrode Cap | Cap based on the international 10-20 system. | Ensures consistent and standardized placement of EEG electrodes across subjects and studies [23]. |
| Visual Stimulation Platform | Software like STIM2, E-Prime, or PsychoPy. | Precisely presents visual cues and memory items with accurate timing control, which is critical for event-related design [4]. |
| Eye Tracker | Infrared video-based tracker. | Monitors gaze position and eye movements to ensure compliance with task instructions (e.g., fixation) and to control for overt attention confounds [25]. |
From Signal to Insight: Measuring Alpha Power and Predicting Memory Performance
EEG and MEG Methodologies for Capturing Posterior Alpha Oscillations
Posterior alpha oscillations (7-13 Hz) represent one of the most prominent rhythmic activities observable in the human brain, typically maximal over occipito-parietal regions during wakeful rest with eyes closed. Historically considered an "idling" rhythm, contemporary research reveals its active role in cognitive processes, particularly in memory encoding and information processing. These oscillations exhibit a complex structure, often consisting of multiple sub-components with distinct spatial and functional characteristics [28]. Understanding the methodologies for accurately capturing these rhythms is paramount for research investigating their role in memory formation.
The fine structure of posterior alpha rhythm has been systematically investigated, revealing that approximately 90% of healthy adults exhibit a multicomponent structure during resting-state recordings. The typical organization comprises a high-frequency occipito-parietal component (ARC1) and a low-frequency occipito-temporal component (ARC2), each demonstrating individual temporal dynamics and stability over weeks to months, representing individual EEG alpha phenotypes [28]. This heterogeneity necessitates methodological approaches capable of resolving these distinct components.
Comparative Analysis of EEG and MEG Methodologies
Biophysical Principles and Signal Characteristics
Electroencephalography (EEG) and Magnetoencephalography (MEG) provide complementary insights into brain activity, yet their distinct biophysical principles fundamentally influence how posterior alpha oscillations are captured and represented.
EEG measures electrical potential differences on the scalp surface generated primarily by radially oriented cortical pyramidal neurons. This method is particularly sensitive to activity from cortical regions with gyral orientations and is more strongly affected by various conductive tissues (skull, scalp, meninges) which blur the electrical field. In contrast, MEG detects the weak magnetic fields generated by intracellular currents, with maximal sensitivity to tangentially oriented sources located in sulcal walls. Magnetic fields are largely unaffected by conductive tissues, providing superior spatial fidelity for source localization [29].
Quantitative Performance Metrics
Table 1: Quantitative Comparison of EEG and MEG for Capturing Alpha Oscillations
| Parameter | EEG | MEG Planar Gradiometers | MEG Magnetometers |
|---|---|---|---|
| Spatial Resolution | ~10-20 mm | ~5-10 mm | ~5-10 mm |
| Temporal Resolution | <1 ms | <1 ms | <1 ms |
| Signal-to-Noise Ratio | Lower | Highest | Intermediate |
| Sensitivity to Radial Sources | High | Low | Low |
| Sensitivity to Tangential Sources | Moderate | High | High |
| Artifact Susceptibility | Higher (especially ocular, muscle) | Lower (especially for distant artifacts) | Lower (especially for distant artifacts) |
| Information Content for Physiology | More homogeneous components | Greater topographical variability, higher information | Intermediate |
Mutual information analysis indicates that MEG-derived independent components exhibit greater topographical variability and higher information content for neurophysiological activity, while EEG components are more homogeneous. Signal-to-noise ratio (SNR) analysis confirms that MEG planar gradiometers capture the highest total information, followed by magnetometers and then EEG [29].
Experimental Protocols for Alpha Oscillation Research
Protocol 1: Investigating Alpha Oscillations in Memory Encoding
This protocol is adapted from a magnetoencephalography study examining the role of posterior alpha activity during long-term memory formation [6].
Experimental Design and Paradigm
- Participants: 25 right-handed, native Dutch speakers without neurological or psychiatric history.
- Stimuli: 2,119 high-frequency concrete nouns divided into three frequency categories, presented in sequences of three words.
- Task Structure: The experiment consists of multiple blocks with three parts:
- LTM Encoding Trials: Participants encode word sequences for long-term memory retention.
- WM Maintenance Trials: Participants maintain word sequences in working memory.
- Memory Retrieval: Participants reconstruct learned sequences using button responses.
- Trial Structure: Three words presented sequentially (0.6 s/word) followed by a 3.4 s rehearsal interval.
- Instruction: Participants are instructed to subvocally construct sentences from the three words to facilitate order memory.
Data Acquisition Parameters (MEG)
- System: 275-sensor axial gradiometer MEG system (VSM/CTF Systems)
- Positioning: Subjects in supine position
- Filtering: Low-pass filter at ~250 Hz
- Sampling Rate: 1200 Hz
- Ancillary Recordings: Electrocardiogram (ECG) and electrooculogram (EOG) via bipolar electrodes
- Head Localization: Three coils placed at nasion and both ear canals
- Session Duration: Approximately 2 hours with a 15-minute break
Data Analysis Approach
- Time-Frequency Analysis: Focus on alpha band (8-12 Hz) during rehearsal intervals
- Source Localization: Identify neural generators of alpha activity
- Subsequent Memory Effect: Contrast alpha power between successfully vs. unsuccessfully encoded LTM sequences
- Key Comparison: Parieto-occipital alpha power during rehearsal for successful vs. unsuccessful encoding
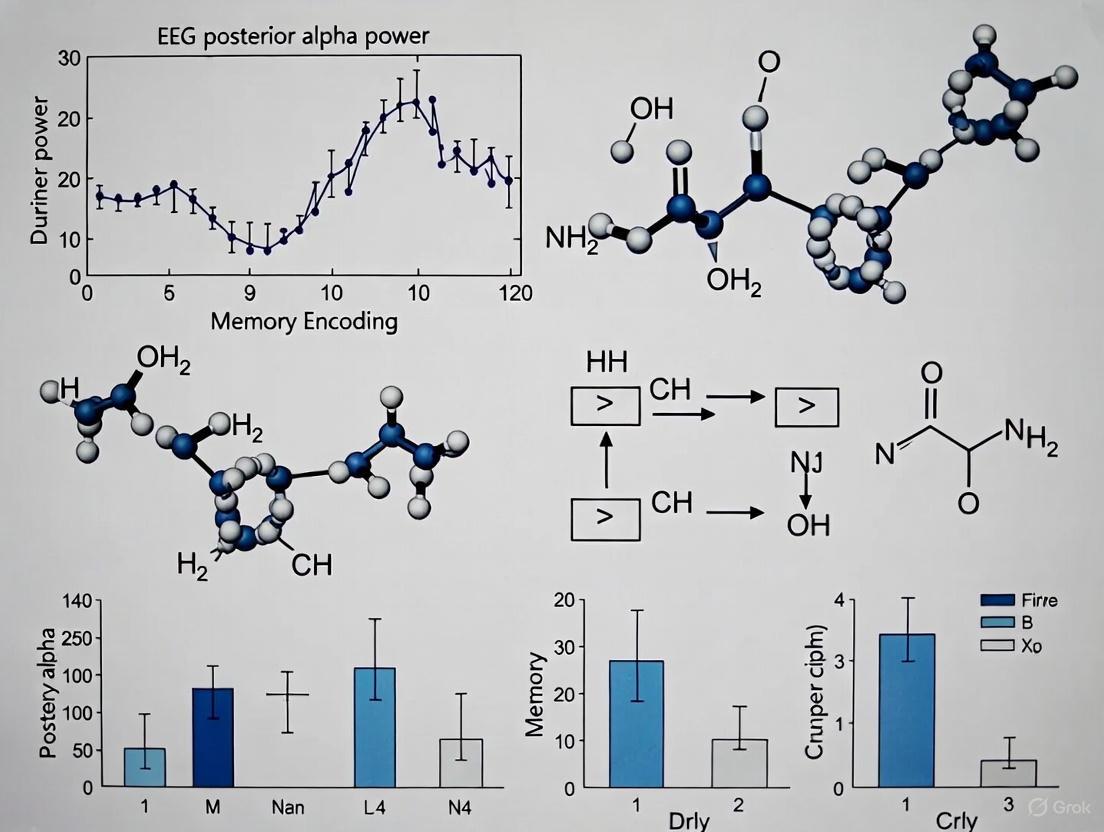
The critical finding from this protocol reveals that posterior alpha power during rehearsal periods is markedly stronger for successfully encoded long-term memory sequences, suggesting that high posterior alpha activity creates an optimal brain state for successful memory formation by actively reducing parieto-occipital activity that might interfere with sequence encoding [6].
Protocol 2: Separating Alpha Oscillations from Broadband Activity
This protocol addresses the challenge of distinguishing true alpha oscillations from overlapping broadband signals, particularly relevant for intracranial recordings [30].
Experimental Setup
- Participants: Patients with intracranial electrodes (ECoG) for clinical monitoring
- Visual Stimulation: Systematic variation of stimulus location across the visual field
- Recording Parameters: Standard clinical ECoG acquisition settings
Signal Separation Methodology
- Spectral Decomposition: Time-frequency analysis using discrete Short-Time Fourier Transform
- Component Separation: Model-based approach to disentangle alpha oscillations from broadband power changes
- Baseline Correction: Account for broadband response to isolate true alpha oscillatory power
- Population Receptive Field (pRF) Modeling: Fit separate models to broadband and alpha time series to characterize spatial tuning
Key Implementation Details
The separation of these components is crucial because visual stimulation typically causes both a decrease in alpha oscillation and a simultaneous increase in broadband power, which can cancel each other at alpha frequencies if not properly separated. The pRF modeling approach reveals that alpha pRFs have similar center locations to broadband pRFs but are several times larger and exhibit opposite response valence [30].
Component Separation and pRF Modeling Workflow
Research Reagent Solutions and Equipment
Table 2: Essential Materials for Alpha Oscillation Research
| Category | Specific Item/Technique | Function/Application | Example Parameters |
|---|---|---|---|
| Recording Systems | 64-channel EEG with active electrodes | High-density scalp recordings | 500-1000 Hz sampling, 0.1-100 Hz bandpass |
| 275-sensor MEG system (axial gradiometers) | Magnetic field detection | 1200 Hz sampling, low-pass ~250 Hz | |
| Intracranial ECoG grids/strips | Direct cortical recording | Clinical settings, high signal-to-noise | |
| Analysis Tools | Time-Frequency Analysis (STFT) | Spectral power quantification | 5-second Hann windows, 50% overlap, 0.2 Hz resolution |
| PARAFAC (Parallel Factor Analysis) | Multi-dimensional component separation | 3-way decomposition (space, frequency, time) | |
| Population Receptive Field (pRF) Modeling | Spatial characterization of neural responses | Difference of Gaussians model, circular symmetry | |
| Experimental Control | E-Prime Software | Precise stimulus presentation | 60 ms stimulus duration, jittered intervals |
| Eye tracking systems | Ocular artifact detection and control | Simultaneous with EEG/MEG recording | |
| Ancillary Equipment | Bipolar ECG/EOG electrodes | Physiological artifact monitoring | Standard clinical placement |
| Head localization coils (MEG) | Head position tracking | Nasion, left/right ear canal placement |
Methodological Considerations for Memory Research
When investigating posterior alpha oscillations in memory encoding contexts, several methodological factors require particular attention:
- Temporal Precision: The excellent temporal resolution of MEG enables separation of presentation (0.6 s/word) and rehearsal (3.4 s) intervals, allowing isolation of memory-specific alpha dynamics [6].
- Individual Alpha Phenotyping: The multicomponent structure of posterior alpha remains stable over weeks to months, suggesting that pre-characterization of individual alpha phenotypes may enhance experimental sensitivity to memory-related effects [28].
- Source Space Analysis: Transforming sensor-level data to cortical source space using appropriate forward models substantially improves the specificity of alpha oscillation localization, particularly for distinguishing dorsal and ventral visual stream contributions [28].
Memory Encoding Experimental Protocol
Advanced Analytical Approaches
Multi-Component Alpha Decomposition
The posterior alpha rhythm typically consists of multiple components that can be dissociated using advanced analytical approaches:
- PARAFAC Methodology: Applying 3-way parallel factor analysis to source-space EEG spectra decomposes alpha into spatial, frequency, and temporal components, revealing distinct occipito-parietal (high-frequency) and occipito-temporal (low-frequency) generators [28].
- Stability Assessment: Test-retest reliability of alpha components over weeks to months establishes their validity as individual neural phenotypes.
- Functional Correlates: Different alpha components show distinct reactivity patterns to experimental manipulations, with high-frequency components potentially more engaged in memory processes.
Source Localization and Connectivity Analysis
For both EEG and MEG data, transforming sensor-level recordings to source space enables more precise localization of alpha generators:
- Forward Modeling: Construct appropriate head models (single-shell for MEG, multi-layer for EEG) to calculate lead fields.
- Inverse Solutions: Apply beamforming, minimum norm estimation, or other inverse algorithms to estimate cortical source activity.
- Functional Connectivity: Calculate phase-based connectivity metrics (phase-locking value, imaginary coherence) between alpha sources to investigate network interactions relevant to memory processes.
Based on the comparative analysis of EEG and MEG methodologies for capturing posterior alpha oscillations, the following recommendations emerge for memory encoding research:
For studies prioritizing spatial precision and source localization of alpha generators, MEG provides superior performance, particularly for tangential sources in sulcal walls. The higher signal-to-noise ratio of MEG planar gradiometers makes them especially suitable for detecting subtle memory-related modulation of alpha rhythms [29].
For research requiring cost-effective acquisition or sensitivity to radially oriented sources, high-density EEG (64+ channels) with appropriate source modeling provides valuable alternatives, though with greater vulnerability to physiological artifacts.
For either modality, implementing component separation techniques is essential for distinguishing true oscillatory activity from broadband responses, particularly in task conditions where both may be simultaneously modulated [30].
The demonstrated relationship between enhanced posterior alpha during rehearsal and successful long-term memory formation [6] confirms the functional relevance of these oscillations and validates the methodological approaches for their quantification. Future research should continue to refine multi-component alpha decomposition to better understand the distinct contributions of various alpha generators to memory processes.
Spectral Analysis and Source Localization Techniques (e.g., sLORETA)
Electroencephalography (EEG) is a non-invasive, versatile functional brain imaging tool prized in neuroscience and clinical neurology for its excellent temporal resolution, low equipment cost, and safety profile [31]. It measures the electrical waveforms generated primarily by cortical pyramidal neurons in the cerebral cortex, which represent the summation of postsynaptic potentials [32] [31]. When a subject wears an EEG cap, electrodes positioned over various brain regions detect these electrical signals, which are then amplified and recorded for analysis [32]. Despite challenges such as environmental noise, movement artifacts, and considerable signal variability, EEG remains a cornerstone for understanding brain activity in normal function, disease states, and in response to therapeutic interventions [32].
The analysis of neural oscillations, or brain rhythms, provides a window into the dynamic and complex activity of the brain. These rhythms are categorized by their frequency range and are typically classified based on sleep or awake patterns [32]. The five main types of rhythms are Delta (0.5–4 Hz), Theta (4–8 Hz), Alpha (8–13 Hz), Beta (13–30 Hz), and Gamma (>30 Hz) [31] [33]. Each rhythm is associated with different brain states and cognitive functions. Notably, posterior alpha power has emerged as a critical biomarker in cognitive neuroscience, particularly in the study of memory encoding and neuropsychiatric disorders [32]. In the context of memory, alpha rhythms are linked to inhibitory processes and the regulation of information flow within the brain [33]. Patients with Major Depressive Disorder (MDD), for instance, show higher alpha-band coherence within the default mode network (DMN), a network most active at rest whose dysregulation may lead to behavioral symptoms like rumination and reduced concentration [32]. This positions alpha activity not just as a marker of brain state, but as a potential translational tool for confirming target engagement and comparing compounds in neurotherapeutic drug development programs [32].
Key Analytical Techniques
Spectral Analysis of EEG Signals
Spectral analysis is a fundamental approach in EEG analysis that quantifies the power or energy of various oscillatory frequency components within the signal. This method is crucial for identifying state changes in the brain related to sleep stages, seizures, emotional shifts, and cognitive processes like memory [31]. The core principle involves transforming the EEG signal from the time domain into the frequency domain to examine its power spectrum. Several computational methods are available for this purpose, each with distinct characteristics and applications [31].
Power Spectrum Analysis Methods
| Method | Description | Key Characteristics |
|---|---|---|
| Fast Fourier Transform (FFT) | A fast algorithm for computing the Discrete Fourier Transform (DFT), which decomposes a signal into its constituent sinusoidal frequencies [31]. | High-frequency resolution but susceptible to noise due to the assumption of signal stationarity [31]. |
| Welch's Method | An improvement on the FFT that involves splitting the signal into overlapping segments, windowing each segment, and averaging the periodograms [31]. | Reduces the variance of power spectral density estimation and is one of the most widely used methods for analyzing EEG power spectra [31]. |
| Autoregressive (AR) Model | A parametric method that models the EEG signal as the output of a linear system driven by white noise, described by a set of coefficients [31]. | Can provide a smoother power spectrum and requires less data length compared to FFT-based methods, but model order must be correctly selected [31]. |
These techniques are extensively applied in memory research. For example, studies on Alzheimer's Disease (AD) consistently show a "slowing" of EEG rhythms, characterized by increased power in lower frequencies (delta, theta) and a decrease in higher frequencies (alpha, beta) [34]. Furthermore, resting-state EEG (rsEEG) gamma power has been identified as a significant predictor of immediate and delayed recall performance in healthy adults, accounting for over 43% of the variance in memory performance [33].
Source Localization: sLORETA
While spectral analysis describes what frequencies are present in the EEG signal, it does not precisely pinpoint where in the brain these rhythms are generated. This is the domain of source localization techniques. Scalp EEG signals are a blurred mixture of electrical activity from multiple, often deep, cortical sources. Source localization uses mathematical models to solve the "inverse problem"—estimating the intracranial sources that gave rise to the recorded scalp potentials [31].
sLORETA (standardized Low-Resolution Brain Electromagnetic Tomography) is a widely used and validated source localization method. Its key principle is to assume that the EEG signal originates from a collection of discrete sources (voxels) within the brain. sLORETA computes a linear solution to the inverse problem and standardizes the current density estimate at each voxel. The primary assumption behind sLORETA is that neighboring neural sources are synchronously active, providing a "smooth" localization solution that has been shown to yield zero localization error under ideal, noise-free conditions for single point sources [31].
This technique is particularly powerful for investigating the role of specific brain networks in memory encoding. For instance, while scalp EEG might show posterior alpha synchronization, sLORETA can be used to verify that this activity is indeed stemming from key regions of the Default Mode Network (DMN), such as the posterior cingulate cortex or medial prefrontal cortex, thereby confirming its hypothesized link to internal mentation and reduced external attention during memory encoding [32].
Application Note: Protocol for Investigating Alpha Power in Memory Encoding
This protocol outlines a comprehensive procedure for using spectral analysis and sLORETA to investigate the role of posterior alpha power in a verbal memory encoding task, suitable for both basic research and clinical trials.
Experimental Workflow
The following diagram illustrates the end-to-end experimental workflow, from data acquisition to final interpretation.
Detailed Methodology
Participant Preparation and EEG Recording
- Participants: Recruit subjects according to study design (e.g., healthy controls, patients with MCI). Ensure they provide informed consent. Screen for history of neurological or psychiatric conditions, and ensure they have had adequate sleep prior to recording [33].
- EEG Setup: Use a high-density EEG system (e.g., 64-128 channels). Apply electrode cap according to the international 10-20 system. Keep impedances below 5 kΩ. Set sampling rate to a minimum of 500 Hz to capture high-frequency activity and avoid aliasing [33] [34].
- Data Acquisition:
- Resting-State EEG: Record a 5-minute, eyes-closed resting-state EEG. Instruct the participant to relax but remain awake. This provides a baseline measure of intrinsic neural activity [33].
- Task EEG: Present the memory encoding task. A sample verbal task involves displaying a series of words on a screen, each for 2 seconds, with an inter-stimulus interval of 1-2 seconds. Instruct participants to memorize the words for a later recall test. The EEG is recorded continuously throughout the task.
Data Pre-processing Protocol
- Filtering: Apply a band-pass filter (e.g., 0.5 - 45 Hz) to remove slow drifts and high-frequency noise [34]. A notch filter (e.g., 50/60 Hz) can be applied to remove line interference.
- Re-referencing: Re-reference the data to a common average reference.
- Artifact Removal:
- Manual Inspection: Visually inspect the data to identify and remove bad channels and segments with large artifacts (e.g., movement, sweat).
- Ocular Correction: Use algorithms like Independent Component Analysis (ICA) to identify and remove components associated with eye blinks and eye movements.
- Epoching: For task data, segment the continuous EEG into epochs time-locked to the onset of each word stimulus (e.g., from -1000 ms pre-stimulus to 2000 ms post-stimulus).
Spectral Analysis Protocol
This protocol focuses on calculating alpha power during the memory encoding task.
- Power Calculation: For each epoch and channel, compute the power spectral density (PSD) using Welch's method. Use a 1-second Hamming window with 50% overlap. This will yield power values for standard frequency bands, including alpha (8-13 Hz).
- Alpha Power Extraction: For each epoch, calculate the average log-power within the alpha band for each electrode. Focus statistical analysis on posterior electrodes (e.g., Pz, P3, P4, O1, O2).
- Analysis: Compare alpha power between conditions (e.g., subsequently remembered vs. forgotten words) or between groups (e.g., healthy controls vs. MCI patients) using statistical tests like repeated-measures ANOVA.
Source Localization Protocol using sLORETA
This protocol is used to identify the cortical generators of the alpha activity observed in the spectral analysis.
- Head Model: Use a standardized head model (e.g., the MNI152 template) co-registered with the electrode positions.
- Input Data: The input for sLORETA is the cross-spectral density matrix within the alpha frequency band, calculated from the pre-processed EEG epochs of interest.
- sLORETA Computation: Run the sLORETA algorithm to estimate the 3D distribution of current density for the alpha rhythm. The output is a volumetric image of neural activity.
- Statistical Analysis:
- Perform voxel-wise statistical comparisons (e.g., paired t-tests for within-subject designs, independent t-tests for between-group designs) on the sLORETA images.
- Correct for multiple comparisons across all voxels using a non-parametric permutation method (e.g., SnPM) or threshold-free cluster enhancement (TFCE).
The Scientist's Toolkit: Essential Research Reagents and Materials
The following table details key solutions and materials required for conducting EEG experiments in memory encoding research.
Table: Essential Reagents and Materials for EEG Memory Research
| Item | Function & Application in Research |
|---|---|
| High-Density EEG System | Core hardware for recording electrical brain activity. A 64-128 channel system is recommended for sufficient spatial sampling for source localization techniques like sLORETA [31]. |
| Electroconductive Gel | Ensures a stable, low-impedance electrical connection between the scalp and EEG electrodes, which is critical for obtaining high-quality data with a high signal-to-noise ratio [32]. |
| Stimulus Presentation Software | Software (e.g., E-Prime, PsychoPy) used to present controlled auditory or visual stimuli (e.g., words, images) during memory encoding and retrieval tasks [33]. |
| EEG Data Analysis Suite | Software platforms (e.g., EEGLAB, BrainVision Analyzer, MNE-Python) that provide integrated environments for data pre-processing, artifact removal, spectral analysis, and statistical comparison [31] [34]. |
| sLORETA Software Package | A specialized, freely available software tool for performing source localization. It is used to compute the 3D intracerebral electrical sources underlying the scalp-recorded EEG activity [31]. |
| Neuropsychological Test Battery | Standardized paper-and-pencil or computerized tests (e.g., Digit Span, Rey Auditory Verbal Learning Test) used to independently assess and validate behavioral memory performance [33]. |
Data Interpretation and Integration
The final phase involves synthesizing results from the spectral and source analyses to draw meaningful conclusions. A typical finding in memory encoding research might be that words which are successfully remembered are associated with a greater decrease in posterior alpha power (event-related desynchronization) during encoding compared to forgotten words [32]. The sLORETA analysis would then reveal whether this desynchronization originates specifically from hubs of the DMN, such as the posterior cingulate cortex.
This integrated approach provides a powerful, non-invasive method for probing the neural mechanisms of memory. The robustness of these techniques is evidenced by their application in clinical populations; for example, computational analyses of EEG signals using methods like wavelet coherence have shown high accuracy in discriminating between Alzheimer's disease patients and healthy elderly controls [34]. Furthermore, these quantitative EEG markers are increasingly used in drug development to confirm target engagement and objectively measure a compound's impact on brain network dynamics, accelerating the search for new neurotherapeutics [32].
Subsequent memory effects (SMEs) are neural processes during encoding that predict whether an experience will be successfully remembered or forgotten at a later time [35]. Research using electrophysiological methods like electroencephalography (EEG) and magnetoencephalography (MEG) has identified oscillatory activity in the alpha band (8-13 Hz) as a robust predictor of encoding success. This application note details the role of posterior alpha power as a key biomarker in memory encoding research, providing structured data, detailed experimental protocols, and analytical workflows for researchers and drug development professionals investigating cognitive function and therapeutic efficacy. The content is framed within a broader thesis that posterior alpha power reflects a cortical state of functional inhibition that facilitates successful memory formation by suppressing processing in task-irrelevant regions [6].
Spectral Correlates of Successful Memory Encoding
Research consistently demonstrates that oscillatory power in specific frequency bands during stimulus encoding can predict subsequent memory performance. The following table summarizes key spectral correlates of successful memory formation identified in recent studies.
Table 1: Spectral Correlates of Successful Memory Encoding
| Frequency Band | Direction of Change for Successful Encoding | Brain Regions | Proposed Functional Role | Citation |
|---|---|---|---|---|
| Alpha (8-13 Hz) | Decrease (in some contrasts) | Parieto-occipital cortex | Reduced power may reflect disinhibition of visual processing areas during stimulus presentation. | [35] |
| Alpha (8-12 Hz) | Increase (during rehearsal) | Parieto-occipital cortex | Increased power may actively suppress interfering inputs from task-irrelevant regions during maintenance. | [6] |
| High-Frequency Activity (44-100 Hz) | Increase | Broad cortical network | Engages task-relevant areas; reflects detailed item processing. | [35] [36] |
| Theta (3-8 Hz) | Inconsistent (increases and decreases reported) | Varies by region (e.g., frontal vs. hippocampal) | May reflect two separate mechanisms; power decreases across cortex and increases in frontal cortex support memory. | [35] |
Posterior Alpha Power and Subsequent Memory Effect
A critical finding in the literature is the specific role of posterior alpha power during the rehearsal period, which distinguishes it from alpha activity during stimulus presentation.
Table 2: Specific Role of Posterior Alpha Power in Memory Encoding
| Experimental Phase | Alpha Power Change | Relationship to Subsequent Memory | Interpretation |
|---|---|---|---|
| Stimulus Presentation | Lower in parieto-occipital regions | Weak or negative correlation with success | Reflects engagement of visual cortical areas for perceptual processing. |
| Rehearsal/Maintenance Interval | Higher in parieto-occipital regions | Stronger for successfully encoded sequences | Actively suppresses interfering external input, facilitating internal memory consolidation. |
Experimental Protocols
This section provides a detailed methodology for a key experiment that established the role of posterior alpha power during rehearsal in long-term memory formation, based on the MEG study by [6].
Protocol: Investigating Alpha Power during Memory Encoding and Rehearsal
Objective: To quantify the role of oscillatory brain activity, particularly in the alpha band, during the encoding and rehearsal of word sequences for long-term memory (LTM) and working memory (WM).
Experimental Design:
- Design Type: Within-subjects, subsequent memory paradigm.
- Primary Measure: Brain activity (MEG/EEG) during task performance, later sorted based on memory accuracy.
Participants:
- 25 right-handed, native language speakers with no history of neurological or psychiatric disorders.
- Obtain written informed consent approved by the local Institutional Review Board (IRB).
Stimuli:
- Stimulus Type: High-frequency, concrete nouns (e.g., >90 occurrences per million).
- Source: Celex database or equivalent spoken word corpus.
- List Construction: Each trial contains three words, matched for frequency and randomized across serial positions. Each word is used only once per participant.
Procedure: The experiment is conducted in multiple blocks, each with three distinct parts, as illustrated in the workflow below.
Data Acquisition:
- Recording System: 275-sensor axial gradiometer MEG system (e.g., VSM/CTF systems).
- Parameters: Data low-pass filtered at ~250 Hz and sampled at 1200 Hz.
- Ancillary Recordings: Electrooculogram (EOG) and electrocardiogram (ECG) to monitor artifacts.
- Head Localization: Head position monitored using coils placed at nasion and in both ear canals.
Data Analysis:
- Preprocessing: Filtering, artifact rejection (e.g., eye blinks, muscle activity).
- Time-Frequency Analysis: Compute spectral power (e.g., using Morlet wavelets) for predefined intervals:
- Encoding/Stimulus Period: Epoch aligned to word presentation.
- Rehearsal Period: Epoch from the 3.4s interval following word offset.
- Statistical Comparison: Contrast spectral power from trials with successfully vs. unsuccessfully encoded sequences (LTM trials) using non-parametric cluster-based statistics.
Signaling Pathways and Cognitive Workflow
The relationship between posterior alpha power, cognitive processes, and memory success can be conceptualized as a functional model of cortical regulation.
Workflow Interpretation:
- The cognitive demand to internally rehearse verbal information initiates a top-down regulatory signal.
- This signal manifests as a synchronized increase in alpha band oscillations in the parieto-occipital cortex.
- The high alpha power actively inhibits or disengages processing in task-irrelevant visual and parietal regions, preventing external sensory input from interfering with the internal mnemonic process.
- This suppression of interference creates an optimal brain state.
- This state facilitates the binding of items to their context and consolidation of the memory trace, leading to successful long-term memory encoding [6].
The Scientist's Toolkit: Research Reagent Solutions
This section catalogs the essential materials, tools, and software required to conduct SME research focusing on alpha oscillations.
Table 3: Essential Resources for EEG/MEG Studies of Memory Encoding
| Category | Item/Reagent | Specification/Function | Example |
|---|---|---|---|
| Recording Equipment | MEG/EEG System | High-temporal-resolution recording of neural oscillations. | 275-sensor CTF MEG system; 128-channel Geodesic Sensor Net [6]. |
| Stimulus Presentation | Experiment Software | Precise control of stimulus timing and response collection. | Presentation, PsychToolbox, E-Prime. |
| Data Analysis | Programming Environment | Data preprocessing, time-frequency analysis, and statistics. | MATLAB with toolboxes (EEGLAB, FieldTrip), Python (MNE-Python). |
| Biomarker Validation | Classification Algorithms | Multivariate pattern analysis to validate predictive power of spectral features. | Logistic Regression, Support Vector Machines (SVMs) [36]. |
| Stimulus Material | Word Pool | Standardized verbal items for memory tasks. | High-frequency concrete nouns from Celex database or Penn WordPool [35] [6]. |
Advanced Analytical Framework: EEG Decoders for Tracking Memory
Modern multivariate analyses can leverage EEG spectral features to predict memory outcomes on a single-trial basis, offering a more sensitive measure for interventional studies.
Protocol: Single-Trial EEG Classification of Memory Success
- Classifier Type: Logistic regression or support vector machines (SVMs).
- Input Features: Spectral power values across multiple frequency bands and electrodes from specific time windows (e.g., during encoding or pre-retrieval).
- Training Paradigm:
- Standard Method: Classifier is trained on all encoding events to distinguish subsequently recalled from non-recalled items. This captures a mix of signals related to memory success and other factors like serial position.
- Event Re-sampling Method: To isolate pure mnemonic signals, training events are re-sampled to ensure an equal ratio of remembered/forgotten items from each serial position in a list [36].
- Output: A classifier model that can assign a probability of subsequent recall to each new item based on its encoding-related EEG signature.
Key Findings:
- Classifiers trained with re-sampling rely more heavily on alpha/beta power decreases to predict successful encoding [36].
- These classifiers are more sensitive to the successful encoding of items that are later recalled in temporal or semantic clusters, linking the neural signal to item-to-context binding.
- Retrieval success (correct recall vs. intrusions) can also be decoded from increases in high-frequency activity (HFA) in the 500ms preceding vocalization [36].
Electroencephalography (EEG) provides a non-invasive window into the neural dynamics of memory processes. Within this domain, the role of posterior alpha power (8-13 Hz) is of particular significance for memory encoding research. Contemporary research has shifted from purely localization-based approaches to distributed models of cognition, leveraging multivariate statistical methods and machine learning to decode cognitive states from complex neural data [36]. This application note details how multivariate decoders, trained on specific EEG spectral features, can classify memory success with high accuracy, and places these findings within the context of posterior alpha activity's recognized role in creating optimal brain states for memory formation [6].
Spectral Features of Memory Success: A Quantitative Synthesis
Multivariate classifiers distinguish successful from unsuccessful memory processes by learning patterns from multiple EEG features simultaneously. The table below synthesizes key spectral features identified by research as predictive of memory encoding and retrieval success.
Table 1: Spectral Features Predictive of Memory Encoding and Retrieval Success
| Memory Process | Spectral Feature | Direction of Change | Brain Regions | Classifier Performance |
|---|---|---|---|---|
| Encoding | Alpha/Beta (10-30 Hz) | Decrease [36] | Widespread | Reliable single-trial prediction [36] |
| Encoding | Theta (4-8 Hz) | Increase [36] [37] | Anterior | Linked to item-context binding [36] |
| Encoding | High-Frequency Activity (HFA) | Increase (without re-sampling) [36] | Cortical/Subcortical Network | - |
| Retrieval | High-Frequency Activity (HFA) | Increase [36] | - | Distinguishes correct recalls from intrusions [36] |
| Retrieval | Theta-band Relative Energy | Key predictor [37] | Left & Right Anterior Superior | Gradient Boosting Classifier: 81.97% accuracy, 91.62% AUC [37] |
| Rehearsal | Posterior Alpha Power | Increase for successful LTM [6] | Parieto-occipital | Marks successful long-term memory formation [6] |
The application of Explainable AI (XAI) methods, such as SHapley Additive exPlanations (SHAP), has further clarified that low theta-band relative energy and root mean square (RMS) values in anterior superior regions are particularly indicative of encoding states [37].
Experimental Protocols for EEG Memory Decoding
Protocol 1: Classifying Encoding Success Using a Delayed Recall Task
This protocol is designed to train a classifier to predict whether an encoded item will be subsequently recalled.
- Objective: To build a multivariate decoder that predicts subsequent memory performance from EEG spectral features recorded during word encoding [36].
- Participants & Task:
- A large cohort (e.g., N=98) each performing a high number of trials (e.g., 576 lists) across multiple sessions in a delayed free recall task [36].
- During encoding, words are presented sequentially. The subsequent recall period requires participants to vocally produce as many words as they can remember from the list.
- EEG Recording & Pre-processing:
- Feature Extraction:
- For each epoch, compute time-frequency representations using methods like Continuous Wavelet Transform (CWT) [38] or Discrete Wavelet Transform (DWT) [37] to extract power in key frequency bands (Delta, Theta, Alpha, Beta, Gamma).
- Critical Step (Event Re-sampling): To isolate neural correlates of encoding success from signals related to list position (e.g., primacy/recency effects), re-sample the data to ensure a matching ratio of subsequently recalled and non-recalled items across all list positions [36].
- Classifier Training & Validation:
Protocol 2: Distinguishing Correct Recall from Intrusion Errors
This protocol focuses on the retrieval phase, classifying whether a recalled item is correct or an intrusion.
- Objective: To train a classifier that distinguishes, prior to vocalization, between correct recalls and intrusion errors (prior-list or extra-list intrusions) [36].
- Participants & Task: Utilize the same behavioral paradigm and participants as in Protocol 1.
- EEG Recording & Pre-processing:
- Follow similar steps as in Protocol 1.
- Epoch the 500 ms immediately preceding the vocalization of each recalled item [36].
- Feature Extraction:
- Compute time-frequency power as in Protocol 1.
- Apply event re-sampling to match recall accuracy rates across the recall output period, controlling for neural changes over time [36].
- Classifier Training & Validation:
- Train a classifier (e.g., logistic regression) on the spectral features from the pre-vocalization epochs to classify recalls as correct or intrusions [36].
- Validate using held-out session data.
Visualization of Analytical Workflows and Spectral Relationships
EEG Memory Decoding Workflow
The following diagram illustrates the end-to-end pipeline for building a multivariate decoder of memory success from raw EEG signals.
Spectral Feature Relationships in Memory
This diagram summarizes the directionality of key spectral features associated with successful memory encoding and retrieval, highlighting the role of posterior alpha.
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials and Tools for EEG Memory Decoding Research
| Item | Function/Description | Example Use Case |
|---|---|---|
| High-Density EEG System | Records electrical brain activity from the scalp; 20+ channels recommended. | Core data acquisition for all memory task paradigms [38]. |
| Stimulus Presentation Software | Presents auditory or visual stimuli and records behavioral responses with precise timing. | Controlling word presentation and recording vocal recalls in delayed recall tasks [36]. |
| Continuous Wavelet Transform (CWT) | Generates time-frequency representations for extracting power in specific frequency bands from EEG signals. | Creating scalograms for feature input into deep learning models [39] [38]. |
| Logistic Regression / Gradient Boosting | Machine learning classifiers that can be trained on spectral features to predict memory outcomes. | Classifying single-trial encoding or retrieval success [36] [37]. |
| Explainable AI (XAI) Tools (e.g., SHAP) | Interprets complex machine learning models and identifies the most important features for predictions. | Revealing that theta-band energy is a key predictor for classifying encoding states [37]. |
| Event Re-sampling Procedure | A statistical method to balance data across conditions (e.g., list position) to avoid confounds. | Isolating neural correlates of pure encoding success, separate from primacy/recency effects [36]. |
Multivariate decoding of EEG spectral features represents a powerful framework for classifying memory success on a single-trial basis. The synthesis of findings reveals a complex orchestration of frequency-specific effects, where decreases in alpha/beta power and increases in theta and posterior alpha power are instrumental for successful encoding, while increased high-frequency activity heralds successful retrieval. The integration of robust experimental protocols, machine learning, and explainable AI provides researchers and drug development professionals with a refined toolkit to quantify memory function, with profound implications for developing biomarkers and evaluating cognitive therapeutics.
Electroencephalography (EEG) posterior alpha power (8-13 Hz) is a prominent neural oscillation that serves distinct, task-dependent functions during memory processing. A growing body of research demonstrates that alpha oscillations play fundamentally different roles in working memory (WM) and long-term memory (LTM) encoding tasks, primarily through mechanisms of functional inhibition and cortical disengagement. This Application Note synthesizes current research findings to delineate these differential roles and provides detailed methodological protocols for researchers investigating alpha oscillations in memory paradigms. Within the broader context of EEG posterior alpha power research in memory encoding, understanding these distinct patterns is crucial for designing targeted experiments and developing precise neural biomarkers for cognitive assessment and pharmaceutical interventions.
Comparative Alpha Dynamics in WM and LTM
Table 1: Directional Changes in Posterior Alpha Power Across Memory Tasks
| Memory Type / Task Phase | Alpha Power Change | Functional Interpretation | Neural Mechanism |
|---|---|---|---|
| Working Memory Encoding | Decrease (ERD) [40] | Active visual processing [40] | Cortical activation of task-relevant areas |
| Working Memory Retention | Increase (ERS) [40] [41] | Protection from interference [40] | Inhibition of irrelevant visual input |
| LTM Encoding (Stimulus) | Decrease [42] | Enhanced sensory processing [42] | Disinhibition for perceptual encoding |
| LTM Encoding (Rehearsal) | Increase [6] | Active interference reduction [6] | Disengagement of task-irrelevant regions |
Table 2: Spectral and Spatial Characteristics of Posterior Alpha
| Characteristic | Working Memory | Long-Term Memory |
|---|---|---|
| Peak Frequency | ~12 Hz during retention [40] | Not explicitly specified |
| Spatial Topography | Parieto-occipital regions [41] | Parieto-occipital regions [6] |
| Functional Subtypes | Two distinct rhythms: Alpha1 (power increase) and Alpha2 (power decrease) [41] | Single rhythm modality reported |
| Relationship to Performance | Positive correlation for Alpha1, negative for Alpha2 [41] | Stronger during successful sequence encoding [6] |
Signaling Pathways and Theoretical Frameworks
The differential roles of alpha in WM and LTM tasks can be conceptualized through distinct neural pathways that emphasize their unique computational demands.
Detailed Experimental Protocols
Protocol 1: Investigating Alpha in Working Memory Retention
This protocol outlines the methodology for studying alpha oscillations during the retention phase of a Sternberg-type working memory task [40].
Experimental Design:
- Task Structure: Implement a modified Sternberg task with sequential presentation of 5 consonant letters (0.5s/item) followed by a 2.0s retention interval and a probe pair recognition test.
- Participant Instructions: Participants judge whether probe letter pairs appeared in the same or reversed order from the study list.
- EEG Recording: Record MEG/EEG during the entire task with particular focus on the 2.0s retention interval.
- Data Analysis: Perform time-frequency analysis on the retention interval data. Calculate event-related synchronization (ERS) in the alpha band (8-14 Hz) relative to baseline.
Key Measurements:
- Alpha power during retention vs. encoding intervals
- Peak alpha frequency during retention
- Alpha-gamma cross-frequency coupling
- Correlation between alpha power and reaction time/accuracy
Protocol 2: Assessing Alpha in LTM Encoding of Sequences
This protocol details the investigation of alpha dynamics during LTM formation using a word sequence encoding paradigm [6].
Experimental Design:
- Stimuli: Present sequences of three concrete nouns (0.6s/word) followed by a 3.4s rehearsal interval.
- Task Conditions: Include separate LTM trials (tested after delay) and WM trials (tested immediately).
- Participant Instructions: Instruct participants to encode word order into LTM using sentence generation strategies.
- Testing Procedure: After a delay period, test participants' ability to reconstruct the order of the three words using button presses.
- EEG Recording: Record MEG/EEG throughout, with separate analysis of word presentation and rehearsal intervals.
Key Measurements:
- Posterior alpha power during rehearsal intervals
- Subsequent memory effect: alpha power for successfully vs. unsuccessfully encoded sequences
- Source localization of alpha generators
Protocol 3: Dissociating Multiple Posterior Alpha Rhythms
This advanced protocol enables researchers to identify and distinguish functionally separate alpha rhythms in posterior cortex [41].
Experimental Design:
- Task: Implement a visual working memory task requiring retention of spatial directions (0.25s cue → 3s delay → test).
- Manipulation: Include trials with and without visual distractors during retention.
- EEG/MEG Recording: High-density recording sufficient for source separation.
Analysis Pipeline:
- Preprocessing: Standard artifact removal and filtering
- ICA Application: Use Independent Component Analysis to separate spatially distinct alpha sources
- Component Clustering: Identify Alpha1 (power-increasing) and Alpha2 (power-decreasing) components
- Characterization: Compare spectral profiles (peak frequency, bandwidth), source locations, and functional responses to distractors
Validation: Confirm that Alpha1 and Alpha2 components show opposite relationships with behavioral accuracy.
The Scientist's Toolkit
Table 3: Essential Research Reagents and Solutions
| Item | Function/Application | Example Specifications |
|---|---|---|
| High-Density EEG Systems | Recording oscillatory activity with sufficient spatial resolution | 64+ channels; compatible with EEG analysis software (e.g., EEGLab, MNE-Python) |
| MEG Systems | Recording neural oscillations with excellent spatial and temporal resolution | 275+ channels; shielded room requirements [6] [40] |
| Stimulus Presentation Software | Precise timing control for memory paradigms | MATLAB with Psychtoolbox; Presentation; E-Prime |
| ICA Algorithms | Separating distinct alpha rhythms from mixed signals | EEGLab RUNICA; Infomax algorithm [41] |
| Time-Frequency Analysis Tools | Quantifying event-related changes in alpha power | MATLAB Wavelet Toolbox; MNE-Python time-frequency implementation |
| Aperiodic Component Removal Tools | Isolating periodic alpha activity from background spectrum | FOOOF algorithm; IRASA method [43] |
Methodological Considerations
When implementing these protocols, researchers should account for several critical factors:
Aperiodic Component Removal: Recent evidence indicates that the EEG power spectrum contains both periodic (oscillatory) and aperiodic components. For accurate alpha power measurement, use parameterization methods to isolate the periodic component [43].
Individual Alpha Frequency Variability: Account for individual differences in peak alpha frequency (typically 8-13 Hz) by using individualized frequency bands rather than fixed cutoffs.
Source Separation Imperative: Sensor-level analyses may conflate multiple distinct alpha rhythms. Implement source separation techniques (e.g., ICA) to dissociate functionally distinct alpha generators [41].
Autonomic and Subjective Confounds: Alpha power correlates with autonomic measures (heart rate variability, respiration) and subjective states (fatigue, arousal). Measure and control for these factors to isolate memory-specific effects [43].
Posterior alpha oscillations play fundamentally different roles in working memory versus long-term memory tasks, primarily implementing inhibitory mechanisms tailored to specific task demands. In WM, alpha increase during retention protects internal representations from interference, while in LTM, alpha increase during rehearsal suppresses competing processing in task-irrelevant regions. The provided protocols enable precise characterization of these distinct alpha functions, with particular importance placed on separating multiple alpha rhythms that may have been conflated in earlier research. These differentiation protocols offer valuable tools for basic memory research and the development of targeted cognitive assessments in pharmaceutical development contexts.
Challenges and Clinical Translation: Alpha Power in Atypical Populations and Intervention
Electroencephalographic (EEG) alpha oscillations (8-13 Hz), particularly originating from posterior cortical regions, represent a fundamental rhythm of the brain's idling and inhibitory control mechanisms. Within memory encoding research, posterior alpha power has been established as a critical neurophysiological marker, where its increase during rehearsal phases facilitates successful long-term memory formation by actively inhibiting task-irrelevant visual input [6]. This application note explores the translational potential of posterior alpha power beyond cognitive neuroscience, examining its emerging role as a diagnostic biomarker in two distinct clinical populations: Attention-Deficit/Hyperactivity Disorder (ADHD) and trauma-related disorders. The investigation is framed within the established context of alpha-mediated inhibitory control mechanisms and their disruption across neuropsychiatric conditions, providing researchers with validated experimental protocols and analytical frameworks for biomarker development.
Alpha Oscillations as Neural Mechanisms and Diagnostic Biomarkers
Functional Significance of Posterior Alpha
Posterior alpha oscillations serve a fundamental inhibitory function in cognitive processing, acting as a "gating mechanism" that suppresses information flow from task-irrelevant visual regions during internal cognitive operations. This functional role is precisely quantified through power spectral density analysis of EEG signals, typically measured in microvolts squared (μV²/Hz). The table below summarizes key functional correlates of posterior alpha power alterations across cognitive and clinical domains:
Table 1: Functional Correlates of Posterior Alpha Power Alterations
| Alpha Power Alteration | Functional Correlation | Associated Condition/Process |
|---|---|---|
| ↑ Increased during rehearsal | Successful long-term memory formation [6] | Cognitive encoding in healthy subjects |
| ↑ Increased in temporal-posterior regions | Fear conditioning/learning circuits [3] | Trauma-related psychopathology |
| ↓ Attenuated | Attention and cognitive control deficits [44] | ADHD pathophysiology |
| ↓ Reduced in eyes-open condition | Complex childhood trauma exposure [3] | Developmental trauma disorder |
Biomarker Classification Framework
For neuropsychiatric biomarker development, the field has established standardized classification systems. According to Davis et al. (2014), biomarkers can be categorized as: (1) risk biomarkers identifying at-risk individuals; (2) diagnostic/trait biomarkers enabling definitive diagnosis; (3) state/acuity biomarkers reflecting current symptom severity; (4) stage biomarkers indicating illness progression; (5) treatment response biomarkers predicting therapeutic outcomes; and (6) prognostic biomarkers forecasting disease course [45]. This framework provides essential context for evaluating the potential clinical applications of alpha oscillations across different disorders.
Alpha Alterations in ADHD: Biomarker Evidence and Protocols
Empirical Findings in ADHD
ADHD research has consistently identified aberrant alpha oscillatory activity as a promising physiological biomarker. Current diagnostic reliance on subjective behavioral assessments remains problematic, with approximately 35% of children diagnosed with ADHD showing minimal functional impairment, highlighting the urgent need for objective biomarkers [44]. EEG studies have revealed that individuals with ADHD demonstrate attenuated resting-state alpha power, particularly during eyes-open conditions, suggesting dysregulated arousal mechanisms and impaired attentional engagement [44] [46]. Theta/beta ratio (TBR) has historically been investigated as a potential ADHD biomarker, though recent evidence suggests posterior alpha power may provide superior discriminatory value, especially when combined with other electrophysiological features.
Table 2: Alpha Oscillation Findings in ADHD and Trauma-Related Disorders
| Disorder | Alpha Alteration Pattern | Brain Regions | Clinical Correlation |
|---|---|---|---|
| ADHD | Attenuated resting-state alpha power [44] | Posterior, parieto-occipital | Inattention, cognitive dysfunction |
| ADHD | Elevated theta/beta ratio [44] | Frontocentral | Impaired cognitive control |
| Complex Childhood Trauma | Reduced temporal-posterior alpha (eyes-open) [3] | T5, T6 electrodes | Trauma-related symptomatology |
| PTSD | Reduced frontal alpha power [47] | AF4, frontotemporal | Hyperarousal symptoms |
| Memory Encoding (Healthy) | Increased parieto-occipital alpha during rehearsal [6] | Parieto-occipital | Successful long-term memory formation |
ADHD Alpha Assessment Protocol
Objective: To quantify resting-state posterior alpha power alterations in ADHD for diagnostic classification.
Equipment:
- High-density EEG system (≥64 channels)
- Electrode caps with international 10-20 placement
- Electromyography (EMG) and electrooculography (EOG) for artifact monitoring
- Electrically shielded, sound-attenuated recording room
- Stimulus presentation system for eyes-open/closed conditions
Procedure:
- Participant Preparation: Apply conductive gel to achieve electrode impedances <5 kΩ.
- Experimental Conditions:
- Eyes-Closed Resting State: 5 minutes of quiet rest with eyes closed
- Eyes-Open Resting State: 5 minutes of fixation on a central crosshair
- Data Acquisition:
- Sampling Rate: ≥1000 Hz
- Online Filtering: 0.1-100 Hz bandpass
- Reference: Linked mastoids or average reference
- Preprocessing:
- Bandpass filtering: 0.5-45 Hz (zero-phase shift filters)
- Ocular and muscular artifact rejection using independent component analysis (ICA)
- Epoch segmentation into 2-second segments with 50% overlap
- Automated and manual artifact rejection (threshold: ±100 μV)
- Spectral Analysis:
- Fast Fourier Transform (FFT) with Hanning window (10%)
- Power spectral density calculation (μV²/Hz) for alpha band (8-13 Hz)
- Regional averaging: parieto-occipital (Pz, P3, P4, O1, Oz, O2) and temporal-posterior (T5, T6) clusters
- Feature Extraction:
- Mean absolute and relative alpha power for each condition and region
- Theta/beta ratio (4-8 Hz/13-30 Hz) at Cz
- Alpha peak frequency (individual alpha frequency)
Analysis:
- Between-group comparisons (ADHD vs. controls) using multivariate ANOVA
- Receiver operating characteristic (ROC) analysis for diagnostic classification
- Correlation analysis with clinical symptom severity (e.g., ADHD rating scales)
Alpha Alterations in Trauma-Related Disorders: Biomarker Evidence and Protocols
Empirical Findings in Trauma Populations
Complex childhood trauma (CCT) exposure produces distinctive alterations in temporal-posterior alpha oscillations, reflecting disruptions in fear circuitry and emotional regulation networks. A recent study demonstrated significantly reduced temporal-posterior alpha power (at T5/T6 electrodes) during eyes-open resting conditions in adolescents exposed to CCT compared to matched controls [3]. This specific alpha reduction pattern aligns with the neurobiological model of trauma, where temporal lobe structures (including amygdala and hippocampus) play crucial roles in fear conditioning and emotional memory processing. Similarly, adults with PTSD exhibit reduced frontal alpha power, which correlates with symptom severity and reflects decreased cortical inhibition underlying hyperarousal symptoms [47].
Trauma Disorder Alpha Assessment Protocol
Objective: To characterize trauma-related alpha oscillatory alterations in temporal-posterior regions.
Equipment:
- Mobile EEG systems (e.g., EMOTIV EPOC X) acceptable for clinical settings [47]
- 14-channel configuration focusing on fronto-temporal coverage
- Validated trauma assessment scales (PCL-5, CTQ)
- Comfortable clinical assessment room
Procedure:
- Participant Screening:
- Administer trauma exposure and symptom measures (e.g., PCL-5, CTQ)
- Document trauma history, type, and chronicity
- EEG Recording:
- Conditions: Eyes-closed (180s) and eyes-open (180s) resting state
- Sampling Rate: 128 Hz (mobile systems) to 1000 Hz (research systems)
- Electrodes of Interest: T5, T6, P3, P4, O1, O2, AF3, AF4
- Data Preprocessing:
- Filtering: 1-40 Hz bandpass, 60 Hz notch filter (if in North America)
- Artifact rejection using automated algorithms
- Epoch segmentation into 2-second segments
- Quantitative Features:
- Absolute and relative alpha power at temporal-posterior sites
- Alpha asymmetry indices (right-left hemisphere)
- Shannon entropy in fronto-temporal regions [47]
- Advanced Analysis:
- Source localization using LORETA for temporal lobe structures
- Functional connectivity metrics (coherence, phase-locking value) in alpha band
Analysis:
- Correlation of alpha metrics with trauma symptom severity scores
- Group comparisons between trauma-exposed and control participants
- Machine learning classification (SVM, random forests) for diagnostic accuracy
Integrated Experimental Workflow and Signaling Pathways
The following diagram illustrates the integrated experimental workflow for assessing alpha alterations across clinical populations, highlighting shared methodological approaches and disorder-specific analytical considerations:
Diagram 1: Experimental workflow for alpha biomarker assessment in ADHD and trauma disorders. The pathway illustrates shared methodological foundations (yellow), core processing steps (green), disorder-specific analytical approaches (blue/red), and validation phases (red).
The neurobiological pathways underlying alpha alterations in ADHD and trauma disorders involve distinct yet partially overlapping neural circuits, as visualized in the following diagram:
Diagram 2: Neurobiological pathways of alpha alterations in ADHD and trauma disorders. The diagram illustrates shared oscillation mechanisms (yellow), disorder-specific pathological processes (blue/red), resulting alpha alterations (solid colors), and common cognitive correlates (green).
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Research Materials and Analytical Tools for Alpha Biomarker Studies
| Category | Specific Tool/Solution | Research Function | Example Application |
|---|---|---|---|
| EEG Hardware | High-density EEG systems (64-256 channels) | Signal acquisition with spatial precision | Source localization of alpha generators |
| EEG Hardware | Mobile EEG headsets (e.g., EMOTIV, Muse) | Ecological validity and scalability | Trauma studies requiring clinical settings [47] |
| Software Tools | EEGLAB, BrainVision Analyzer | Preprocessing and ICA artifact removal | Ocular correction in resting-state data |
| Software Tools | FieldTrip, MNE-Python | Time-frequency analysis | Alpha power spectral density calculation |
| Software Tools | LORETA, sLORETA | Source localization | Temporal lobe alpha source estimation |
| Analytical Metrics | Power Spectral Density (μV²/Hz) | Alpha oscillation quantification | Group comparisons of resting alpha |
| Analytical Metrics | Shannon Entropy | Brain signal complexity measurement | Trauma symptom correlation [47] |
| Analytical Metrics | Theta/Beta Ratio (TBR) | Complementary ADHD biomarker | Differentiation from alpha measures [44] |
| Statistical Tools | Support Vector Machines (SVM) | Diagnostic classification | ADHD vs. control discrimination |
| Statistical Tools | ROC Curve Analysis | Biomarker sensitivity/specificity | Diagnostic accuracy assessment |
Discussion and Future Directions
The converging evidence from ADHD and trauma research highlights the robust biomarker potential of posterior alpha oscillations while emphasizing disorder-specific topographic patterns. In ADHD, attenuated posterior alpha reflects core deficits in attentional engagement and arousal regulation, whereas in trauma disorders, reduced temporal-posterior alpha specifically implicates dysregulated fear circuitry and emotional processing networks. These distinct spatial patterns suggest that alpha oscillations may serve as differential biomarkers when combined with precise topographic analysis.
Future research should prioritize multisite validation studies with standardized protocols to establish reproducible alpha biomarker thresholds across diverse populations. Longitudinal designs tracking alpha dynamics alongside symptom progression will clarify whether these measures function as state or trait biomarkers. Furthermore, integrating alpha metrics with complementary biomarkers—such as eye-tracking measures of pupil diameter [48] or structural neuroimaging—may yield multimodal biomarker panels with enhanced diagnostic specificity. The development of portable EEG systems already enables ecological momentary assessment of alpha dynamics in real-world settings, opening new avenues for monitoring treatment response and disease progression [47].
Within memory encoding research frameworks, these clinical findings extend our understanding of alpha's functional significance beyond cognitive operations to encompass emotional regulation and trauma processing. The demonstrated role of posterior alpha in inhibiting task-irrelevant processing during memory formation [6] provides a mechanistic foundation for interpreting its disruption in clinical populations where intrusive thoughts and impaired cognitive control are hallmark features.
Alpha oscillations, particularly in posterior and temporal regions, represent promising diagnostic biomarkers for both ADHD and trauma-related disorders. The documented alterations—reduced posterior alpha in ADHD and diminished temporal-posterior alpha in trauma populations—reflect disorder-specific disruptions in fundamental inhibitory mechanisms supported by thalamocortical circuits. The experimental protocols and analytical frameworks presented herein provide researchers with validated methodologies for quantifying these biomarkers with high translational potential. As the field advances toward biologically-defined neuropsychiatric diagnoses, alpha oscillations offer objectively measurable targets for diagnostic refinement, treatment personalization, and therapeutic monitoring across diverse clinical populations.
In electroencephalography (EEG) research on memory encoding, the accurate interpretation of neural signals, particularly posterior alpha power (8-13 Hz), is complicated by the presence of confounding variables. Two significant confounds—serial position and cognitive load—systematically influence brain activity and memory outcomes, potentially leading to spurious findings if not properly controlled. The serial position effect describes the robust finding that items presented at the beginning (primacy) and end (recency) of a list are remembered better than middle items, forming a U-shaped recall curve [49]. Concurrently, cognitive load—the mental effort required to process information—fluctuates throughout memory tasks and directly impacts neural resources available for encoding [50]. When researchers investigate the relationship between posterior alpha power and memory encoding, these factors introduce variance that can obscure true effects or create illusory correlations. This application note provides methodologies to identify, measure, and control for these confounds, ensuring more valid interpretations of EEG data in memory research.
Theoretical Framework and Key Concepts
The Nature of the Confounds
Serial position effects emerge from multiple cognitive mechanisms that directly modulate EEG activity. Primacy items benefit from elevated attention and greater opportunity for rehearsal, while recency items remain accessible in short-term memory buffers with minimal interference [49]. Critically, the presentation order itself creates a systematic bias in neural engagement throughout a memory task. Studies reveal that event-related potential (ERP) components, including the late positive complex, show amplitude variations across serial positions independent of subsequent memory performance [49]. This demonstrates that neural activity measured during encoding confounds intrinsic memory processes with position-dependent neural adaptation, resource allocation, and rehearsal dynamics.
Cognitive load represents the total mental effort being used in working memory, which fluctuates during complex tasks. As load increases, it consumes shared attentional resources necessary for memory encoding, potentially suppressing alpha rhythm modulation that would otherwise indicate successful formation of memory traces [51] [50]. The relationship between cognitive load and posterior alpha power is particularly relevant, as alpha oscillations are known to reflect functional inhibition of task-irrelevant cortical regions during cognitive processing. High cognitive load may therefore manifest as either alpha suppression (indicating cortical engagement) or alpha enhancement (reflecting inhibitory processes), depending on task demands and brain regions involved.
Impact on Posterior Alpha Power Interpretation
Posterior alpha power during memory encoding is theorized to reflect functional inhibition of sensory cortical regions, potentially facilitating memory formation by reducing interference from extraneous visual input. However, serial position systematically modulates this signal—early list items may show enhanced alpha due to focused attention, while middle items might exhibit alpha suppression from interference or fatigue [49] [51]. Similarly, cognitive load influences alpha dynamics through resource competition—high load conditions may suppress posterior alpha as visual processing resources are recruited for maintenance, while low load permits the alpha synchronization associated with successful encoding [50]. When unaccounted for, these confounds can create the false appearance of alpha-memory relationships or mask genuine effects.
Quantitative Characterization of Confounds
Table 1: Empirical Effects of Serial Position and Cognitive Load on EEG Measures
| Experimental Manipulation | EEG Measure | Effect Size/Direction | Behavioral Correlation | Key Brain Regions |
|---|---|---|---|---|
| Serial Position (Primacy) | Late Positive Component Amplitude | Increased by 1.5-2.0 μV [49] | Higher recall success (70-80%) [49] | Parietal, Prefrontal |
| Serial Position (Middle) | Late Positive Component Amplitude | Decreased by 1.0-1.5 μV [49] | Lower recall success (40-50%) [49] | Parietal, Prefrontal |
| Serial Position (Recency) | Late Positive Component Amplitude | No significant change [49] | Higher recall success (70-80%) [49] | Parietal, Temporal |
| High Cognitive Load | Gamma Power (30-100 Hz) | Increased by 15-25% [50] | Reduced accuracy (10-15% decrease) [50] | Frontal, Temporal |
| Low Cognitive Load | Theta Power (4-7 Hz) | Increased by 10-20% [33] | Improved delayed recall | Frontal, Hippocampal |
| High Cognitive Load | Posterior Alpha Power | Variable (Task-Dependent) [51] | Inconsistent memory performance | Parietal, Occipital |
Table 2: Statistical Control Methods for Confounds in EEG Memory Studies
| Control Method | Implementation Approach | Advantages | Limitations |
|---|---|---|---|
| Event Re-sampling | Equalize trial counts across serial positions and performance categories [52] | Isletes mnemonic from non-mnemonic signals; Reduces positional bias | Requires large trial numbers; Complex implementation |
| Confound Regression | Remove variance associated with serial position before memory analysis [53] | Straightforward implementation; Works with standard designs | Can introduce negative bias if not cross-validated [54] |
| Stratified Analysis | Analyze serial positions separately, then meta-analyze effects | Intuitive interpretation; No special statistical methods | Reduces power through data splitting; Multiple comparisons |
| Multivariate Decoding | Train classifiers to predict memory while controlling for confounds [52] | Models complex interactions; High sensitivity to distributed patterns | Black box interpretation; Computationally intensive |
| Behavioral Covariates | Include recall timing, intrusion rates as covariates in models | Accounts for performance differences; Adds ecological validity | Does not control for neural specificity of confounds |
Experimental Protocols for Confound Control
Protocol 1: Serial Position-Controlled Memory Paradigm
Purpose: To measure EEG correlates of memory encoding while controlling for serial position effects.
Stimuli and Materials:
- Word Lists: 20 lists of 12 common nouns each, matched for frequency, concreteness, and length [49]
- Presentation Software: E-Prime, PsychoPy, or Presentation
- EEG System: 64+ channel system with active electrodes
Procedure:
- Participant Preparation: Apply EEG cap following standard 10-20 system. Impedance should be maintained below 10 kΩ.
- Task Structure:
- Present words sequentially for 250 ms each with 1500 ms interstimulus interval [49]
- Fixation cross displayed during intervals to minimize eye movement artifacts
- After each list, participants engage in 30-second distractor task (e.g., arithmetic problems)
- Implement free recall period (90 seconds) where participants verbally recall words
- EEG Recording Parameters:
- Sampling rate: 1000 Hz
- Online filters: 0.1-100 Hz bandpass
- Reference: Linked mastoids or average reference
- Serial Position Control:
- Counterbalance list orders across participants
- Implement event re-sampling during analysis to equalize trials across positions [52]
- Tag each trial with serial position (1-12) for stratified analysis
Analysis Pipeline:
- Preprocess EEG data (filtering, artifact rejection, ICA)
- Segment data into -200 to 1200 ms epochs around each word presentation
- Bin trials by serial position (primacy: 1-3, middle: 4-9, recency: 10-12) and memory outcome (recalled/not recalled)
- Compute ERPs and time-frequency representations for each bin
- Apply statistical controls for serial position before testing memory effects [52]
Figure 1: Experimental workflow for serial position-controlled EEG memory paradigm
Protocol 2: Cognitive Load Manipulation and Measurement
Purpose: To quantify and control for cognitive load effects on posterior alpha power during memory encoding.
Stimuli and Materials:
- Dual-Task Paradigm: Primary memory task with secondary load manipulation (e.g., tone counting, motor sequence)
- Difficulty Levels: 3-5 levels of cognitive load manipulation
- Physiological Measures: Pupillometry, heart rate variability in addition to EEG
Procedure:
- Load Manipulation:
- Implement simultaneous task demands (e.g., memory encoding while performing n-back task) [50]
- Vary load across blocks with counterbalanced order
- Include practice sessions to ensure task understanding
- EEG Recording:
- High-density EEG (64+ channels) with synchronized pupillometry
- Sampling rate: 1000 Hz or higher to capture gamma activity
- Record electrooculogram (EOG) for ocular artifact correction
- Load Quantification:
- Collect subjective load ratings after each block (NASA-TLX)
- Measure performance on secondary task as objective load index
- Extract pupillary dilation as physiological load indicator [51]
Analysis Approach:
- Preprocess EEG data with special attention to artifact removal
- Compute time-frequency representations for posterior electrodes (Pz, POz, Oz)
- Extract alpha power (8-13 Hz) during encoding periods
- Model relationship between alpha power and load metrics using multiple regression
- Residualize alpha power against load measures before memory analysis
Figure 2: Cognitive load measurement and control protocol
Analytical Framework for Confound Adjustment
Statistical Control Methods
Cross-Validated Confound Regression represents the gold standard for controlling continuous confounds like cognitive load [54]. This approach involves:
- Feature Selection: Identify EEG features of interest (e.g., posterior alpha power)
- Confound Measurement: Quantify confounds (serial position, load metrics, time-on-task)
- Nested Regression: Within each cross-validation fold, regress confounds from EEG features
- Memory Analysis: Use residuals from regression in subsequent memory comparisons
Implementation Code Concept:
Multivariate Pattern Analysis with confound control offers an alternative approach [52]:
- Feature Space Construction: Include both neural features and confound measures
- Regularized Classification: Train classifiers to predict memory outcome while penalizing confound-related variance
- Feature Importance: Interpret resulting models to identify confound-resistant neural signals
Validation and Sensitivity Analysis
Positive Control Analyses verify that confounds are adequately measured:
- Confirm expected serial position effects on behavior and ERPs [49]
- Validate cognitive load manipulations through performance and physiological measures [50]
Negative Control Analyses ensure confound control doesn't introduce bias:
- Test for absence of relationship between adjusted EEG features and confounds
- Verify that control methods don't remove genuine memory signals through simulation
Sensitivity Analysis quantifies robustness of findings across control methods:
- Compare results across different statistical control approaches
- Estimate bounds of true effects given residual confounding
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Resources for Controlling Confounds in EEG Memory Research
| Resource Category | Specific Tools/Measures | Primary Function | Implementation Considerations |
|---|---|---|---|
| Stimulus Presentation | E-Prime, PsychoPy, Presentation | Precise timing of serial word presentation | Ensure millisecond accuracy for ERP studies; Counterbalance lists |
| EEG Acquisition | Biosemi ActiveTwo, BrainVision, EGI Geodesic | High-fidelity neural recording | 64+ channels; Sampling rate ≥1000 Hz; Low impedance (<10 kΩ) |
| Cognitive Load Measures | NASA-TLX, Behavioral Performance, Pupillometry | Quantify cognitive load during encoding | Synchronize with EEG; Multiple measures for validation |
| Artifact Handling | ICA, MARA, FASTER | Remove ocular/movement artifacts | Balance artifact removal with signal preservation; Document rejection rates |
| Serial Position Control | Event Re-sampling Algorithms [52] | Equalize position distribution in analysis | Custom scripts; Requires large trial numbers (>20 per position) |
| Statistical Control | Cross-Validated Confound Regression [54] | Remove confound variance from EEG features | Implement nested cross-validation to avoid bias |
| Multivariate Analysis | MVPA-Light, scikit-learn | Decode memory states while controlling confounds | Pattern classification; Feature importance analysis |
| Data Quality Metrics | Trial Counts, Signal-to-Noise Ratio, ERP Quality | Quantify data quality for each serial position | Report by position group; Exclude participants with position-specific artifacts |
Effective control for serial position and cognitive load confounds requires a multi-method approach combining experimental design and analytical solutions. Researchers should prioritize event re-sampling for serial position control [52] and cross-validated confound regression for continuous covariates like cognitive load [54]. For studies of posterior alpha power specifically, we recommend:
- Design Stage: Counterbalance stimuli across serial positions; include cognitive load manipulations
- Data Collection: Record sufficient trials (>20 per serial position); monitor data quality by position
- Analysis Phase: Implement event re-sampling or stratified analysis; validate control methods
- Reporting: Explicitly describe confound control methods; report effects both with and without controls
These protocols enable more precise characterization of the relationship between posterior alpha power and memory encoding by isolating neural signals specific to mnemonic processes from those reflecting structural task demands. Through rigorous confound management, researchers can advance our understanding of the neural mechanisms of memory while developing more sensitive biomarkers for clinical application.
Electroencephalography (EEG) alpha power (8-12 Hz), particularly over posterior brain regions, is a central biomarker in cognitive neuroscience research for investigating the neural underpinnings of working memory. Contemporary theoretical frameworks posit that posterior alpha oscillations during memory retention reflect an active gating mechanism, serving to inhibit distracting visual input and protect internal representations [55] [56]. A critical question within this domain is whether this neural activity exhibits experience-dependent plasticity. This Application Note synthesizes current evidence to address whether targeted working memory interventions can systematically modulate posterior alpha power, providing researchers with a consolidated overview of empirical findings, detailed experimental protocols, and essential methodological tools.
Synthesized Evidence: Effects of Interventions on Alpha Power
The following tables summarize key quantitative findings from recent studies on how different types of working memory interventions influence alpha power and behavioral performance.
Table 1: Working Memory Training Studies and Their Impact on Alpha Power
| Study & Intervention | Population | Design | Key Alpha Power Findings | Behavioral Outcomes |
|---|---|---|---|---|
| Cogmed WM Training [57] [56] | 136 ADHD students; 41 healthy peers | RCT; Pre/Post EEG | Marginal increase in posterior alpha power after training vs. waitlist control. No dose-effect. | No significant training effects on task accuracy. |
| Alpha Neurofeedback [58] | 40 healthy adults | Randomized, Single-blind, Sham-controlled | Significant alpha increase within NF sessions. No change in alpha ERSP during Sternberg task. | Significant improvement in visual WM capacity (Corsi-block) and precision (color-recall). |
| Structured WM Training [59] | 24 healthy older adults | Double-blind, Active Control | No direct alpha power measure. Increased high-beta/alpha ratio at rest post-training. | Medium-large gains in WM criterion task and intrusion errors (inhibitory control). |
Table 2: Non-Training Studies on Alpha's Functional Role in WM
| Study & Focus | Population | Design | Key Alpha Power Findings | Interpretation / Mechanism |
|---|---|---|---|---|
| EEG-Guided tACS [60] | 25 healthy adults | Crossover tACS Stimulation | 8 Hz prefrontal tACS improved verbal WM accuracy vs. 40 Hz/sham. | Alpha activity is causally involved in successful WM performance. |
| Attention Direction [55] | 30 & 41 healthy adults | VR & Lab Experiments | ERS (Increase): During internal mental arithmetic. ERD (Decrease): During external visual attention. | Alpha power crucially isolates a subject from the environment to support internal processing. |
| Intrinsic Motivation [61] | 48 healthy adults (32 F) | EEG during Sternberg Task | Parietal alpha during encoding decreased with higher subjective difficulty and motivation. | Motivational state and personality traits modulate oscillatory correlates of WM. |
Detailed Experimental Protocols
Protocol: Alpha Neurofeedback Training for Visual Working Memory
This protocol is adapted from the randomized, single-blind, sham-controlled study by [58].
1. Objective: To investigate whether up-regulating alpha power via neurofeedback (NF) leads to improvements in the quantity (capacity) and quality (precision) of visual working memory.
2. Participants: Healthy adults with normal or corrected-to-normal vision. Sample size: 40 participants (20 in NF group, 20 in sham group).
3. Equipment & Reagents:
- EEG System: 64-channel wireless amplifier.
- Software: NF processing software (e.g., EEGLAB, BCILAB), PsychToolbox for task presentation.
- Stimuli Display: Standard computer monitor.
- Behavioral Tasks: Corsi-block tapping task, color-recall task, and modified Sternberg task.
4. Procedure:
- Phase 1: Pre-Test Assessment (Day 1)
- Record baseline EEG during a modified Sternberg task.
- Administer behavioral tasks (Corsi-block, color-recall) to assess baseline WM capacity and precision.
- Phase 2: Intervention (5 Consecutive Days)
- NF Group: Participants complete five 30-minute NF sessions. The real-time EEG alpha power (8-12 Hz) from pre-defined electrodes is converted into visual feedback. Participants are instructed to mentally increase the feedback to learn alpha self-regulation.
- Sham Control Group: Participants undergo an identical procedure but receive feedback based on a non-relevant frequency band or pre-recorded EEG signals.
- Phase 3: Post-Test Assessment
- Post-test 1: Immediately after the final training session.
- Post-test 2: 7 days after training completion (follow-up).
- Repeat the pre-test assessments (EEG and behavioral tasks).
5. Data Analysis:
- EEG: Analyze within-session and across-session alpha amplitude. Compute alpha Event-Related Spectral Perturbation (ERSP) and pre-stimulus alpha power during the Sternberg task.
- Behavior: Compare pre- and post-test scores on the Corsi-block (capacity) and mean absolute error in the color-recall task (precision) using repeated-measures ANOVA or Friedman's test.
Protocol: Working Memory Training in Clinical (ADHD) Populations
This protocol is based on the randomized controlled trial (RCT) by [57] [56].
1. Objective: To examine if neural indices of working memory maintenance (posterior alpha) differ in adults with ADHD and if they are malleable through intensive working memory training.
2. Participants: College students with a confirmed ADHD diagnosis and a matched healthy control group.
3. Equipment & Reagents:
- EEG System: Standard clinical EEG system.
- Training Software: Cogmed Working Memory Training (CWMT) program.
- Assessment Task: Visual delayed-match-to-sample task.
4. Procedure:
- Study 1 (Group Comparison):
- Record EEG from the ADHD group and healthy peers during the delayed-match-to-sample task.
- Study 2 (RCT):
- Randomly assign participants with ADHD to one of three groups:
- Standard-Length Training: 45 minutes/day of CWMT.
- Shortened-Length Training: 15 minutes/day of CWMT.
- Waitlist Control: Equal contact with a coach, no active training.
- The training is conducted over several weeks.
- Post-intervention, all participants undergo the same EEG recording and behavioral assessment as in Study 1.
- Randomly assign participants with ADHD to one of three groups:
5. Data Analysis:
- Behavior: Analyze task accuracy (hit rate minus false alarm rate) using ANOVA with factors Group and Memory Load.
- EEG: Extract and compare posterior alpha power during the maintenance phase of the task. Use mixed-model ANOVAs to test for group differences (Study 1) and training-induced changes (Study 2).
Conceptual Workflow and Signaling Pathways
The following diagram illustrates the hypothesized pathway from intervention to cognitive improvement, integrating key concepts from the reviewed literature on how working memory training may modulate alpha oscillations and behavior.
The Scientist's Toolkit: Key Research Reagents & Materials
Table 3: Essential Materials for EEG Research on Working Memory and Alpha Plasticity
| Item / Solution | Specification / Function | Exemplar Use Case |
|---|---|---|
| High-Density EEG System | 64+ channels; wireless systems beneficial for VR/ecological designs. | Core neural data acquisition in all cited studies [60] [55] [58]. |
| tACS Device | Non-invasive brain stimulator for modulating cortical oscillations. | Applying 8 Hz stimulation to prefrontal regions to enhance verbal WM [60]. |
| Neurofeedback Software Suite | Real-time EEG signal processing and feedback presentation. | Alpha amplitude training protocol [58]. |
| Cognitive Task Programming Tool | e.g., MATLAB with PsychToolbox, Presentation, E-Prime. | Presentation of n-back, delayed-match-to-sample, and Sternberg tasks [60] [56]. |
| Virtual Reality (VR) Platform | Immersive environment for controlling external attention. | Studying alpha ERS during internal tasks amidst rich external stimuli [55]. |
| Spectral Decomposition Algorithm | e.g., FOOOF (Fitting Oscillations & One Over F). | Separating periodic (oscillatory) from aperiodic (1/f) components of the EEG signal [62]. |
| Working Memory Training Program | Adaptive, computerized tasks (e.g., Cogmed). | Investigating near-transfer and neural plasticity in clinical populations [57] [56]. |
Electroencephalographic (EEG) alpha rhythms (8-13 Hz), particularly those generated in posterior cortical regions, have transcended their historical status as mere idling rhythms to be recognized as fundamental components of active cognitive processing. Research increasingly implicates these oscillations in core cognitive functions, with their modulation presenting a promising frontier for non-invasive cognitive enhancement [63]. This field is anchored in a foundational thesis investigating the specific role of posterior alpha power in memory encoding processes. The posterior dominant rhythm, a hallmark of the awake, resting brain, is not a monolithic entity but a dynamic phenomenon whose power and frequency are precisely tuned by cognitive demands. This document provides detailed Application Notes and experimental Protocols to guide researchers and drug development professionals in exploring and manipulating posterior alpha rhythms to improve memory and cognitive function, framing all content within the established context of memory encoding research.
Quantitative Data Synthesis: Alpha Rhythm Signatures in Cognition
The following tables synthesize key quantitative findings from recent research, providing a reference for experimental design and biomarker identification.
Table 1: Alpha Band Spectral Signatures in Cognitive States and Disorders
| Condition / State | Alpha Band Change | Specific Frequency / Location | Quantitative Correlation / Ratio | Associated Cognitive Function |
|---|---|---|---|---|
| Working Memory Load [64] | Lower Alpha (8-9 Hz) ERD* | Widespread, posterior | Increases with set size | Attentional allocation to memory items |
| Working Memory Spatial Extent [64] | Upper Alpha (10-12 Hz) ERD* | Posteriorly localized | Associated with spatial extent | Suppression of irrelevant sensory input |
| Healthy Aging [63] | Reduced Spatial Variability (S values) | Frontal region | Lower S values vs. young adults | Neurophysiological aging |
| Psychosis Risk (ARMS-P) [63] | Increased Beta1 Power | Left Middle Frontal Gyrus | Higher β1/alpha power ratio | Biomarker for psychosis prediction; correlates with physical anxiety |
| Alzheimer's Disease [65] | Overall Alpha Power Reduction | Posterior regions | Spectral slowing (↑δ/θ, ↓α/β) | Synaptic dysfunction, cholinergic deficit |
*ERD: Event-Related Desynchronization (power decrease); ERS: Event-Related Synchronization (power increase)
Table 2: Quantitative EEG (qEEG) Indices for Monitoring Neurocognitive States
| qEEG Index | Abbreviation | Calculation / Definition | Neurocognitive Interpretation | Application Context |
|---|---|---|---|---|
| Delta/Alpha Ratio [66] | DAR | (Delta Power) / (Alpha Power) | Indicator of cortical slowing or dysfunction | Stroke recovery, dementia monitoring |
| Brain Symmetry Index [66] | BSI | Quantifies power asymmetry between hemispheres | Marker of lateralized brain pathology | Post-stroke mental disorders, epilepsy |
| Alpha Peak Frequency [67] | APF | Center frequency of the posterior alpha rhythm | Faster alpha linked to enhanced memory retention | Working memory performance, cognitive enhancement |
| Objective Sleepiness Scale [68] | OSS | Classifies sleepiness into 5 levels based on alpha/theta duration | Direct measure of drowsiness and alertness | Occupational safety, fatigue monitoring |
| Infra-slow Fluctuations [63] | - | Very slow fluctuations of alpha power | Marker of cognitive efficiency and resource availability | Cognitive load assessment |
Experimental Protocols for Alpha Rhythm Research
This section outlines detailed methodologies for key experiment types relevant to investigating alpha rhythms in memory encoding.
Protocol: Closed-Loop Auditory Stimulation for Alpha Frequency Modulation
Objective: To laterally modulate posterior alpha frequency during the memory retention period to test its causal role in working memory performance [67].
Background: Alpha oscillations during the retention interval of a working memory task are functionally significant. This protocol uses real-time analysis to deliver auditory stimulation at a specific phase of the alpha rhythm, leveraging the phase-dependent modulation of alpha frequency by sound.
Materials:
- EEG system with at least 2 posterior electrodes (e.g., O1, O2, P3, P4).
- Auditory stimulation system (headphones).
- Software for real-time EEG analysis and closed-loop stimulation (e.g., Psychtoolbox, OpenVIBE).
Procedure:
- Participant Setup: Apply EEG cap according to standard 10-20 system. Ensure impedance is below 5 kΩ. Instruct participants on the task.
- Baseline Recording (5 mins): Record resting-state EEG (eyes-open and eyes-closed) to determine individual alpha peak frequency for each hemisphere.
- Working Memory Task:
- Encoding: Present a memory array containing stimuli (e.g., oriented gratings) in one or both visual hemifields for 500-1000 ms.
- Retention: A blank screen maintenance period of 2000-3000 ms. During this period, the closed-loop stimulation is active.
- Retrieval: Present a probe stimulus and require the participant to indicate if it matches one of the memorized items.
- Real-Time Processing & Stimulation:
- Signal Extraction: Continuously read EEG data from the posterior electrodes contralateral to the memorized hemifield(s).
- Phase Estimation: Apply a real-time bandpass filter (e.g., individual alpha frequency ±2 Hz) and extract the instantaneous phase using the Hilbert transform.
- Stimulation Trigger: When the instantaneous alpha phase crosses a pre-defined value (e.g., 0° or 180°), trigger a brief, low-volume auditory click or tone burst.
- Condition Design: Employ a within-subjects design with three conditions: (a) Left Hemisphere Stimulation, (b) Right Hemisphere Stimulation, (c) No Stimulation (Control). Stimulation is targeted to the hemisphere contralateral to the memorized stimuli.
- Data Analysis:
- Compare behavioral performance (accuracy, reaction time) between stimulation conditions.
- Analyze induced lateralization of alpha frequency and power between hemispheres during the retention interval.
- Correlate the degree of induced frequency lateralization with the behavioral asymmetry in task performance [67].
Protocol: Dissociating Lower and Upper Alpha Sub-bands in Visual Working Memory
Objective: To independently characterize the roles of lower (8-9 Hz) and upper (10-12 Hz) alpha sub-bands in encoding the number (set size) and spatial distribution of memory items [64].
Materials:
- High-density EEG system (64+ channels recommended).
- Stimulus presentation software (e.g., E-Prime, PsychoPy).
- EEG processing toolbox (e.g., EEGLAB, FieldTrip).
Procedure:
- Experimental Design:
- Use a whole-field change detection paradigm.
- Independently vary Set Size (e.g., 2, 4, 6 items) and Spatial Extent (e.g., items clustered centrally vs. spread widely) of the memory array in a factorial design.
- EEG Recording: Record continuous EEG during task performance at a sampling rate ≥ 500 Hz.
- Preprocessing:
- Apply high-pass (0.1 Hz) and low-pass (40 Hz) filters.
- Remove bad channels and interpolate.
- Perform Independent Component Analysis (ICA) to remove ocular and muscular artifacts.
- Segment data into epochs time-locked to the memory array onset.
- Time-Frequency Analysis:
- Compute time-frequency representations of power for each electrode, epoch, and condition. Use Morlet wavelets spanning the alpha range (e.g., 2-30 Hz).
- Separate the analysis into lower alpha (8-9 Hz) and upper alpha (10-12 Hz) bands.
- Calculate event-related desynchronization/synchronization (ERD/ERS) as the percentage change in power from a pre-stimulus baseline.
- Neural Decoding Analysis:
- Use multivariate pattern analysis (MVPA) or a similar decoding approach (e.g., support vector machine) to decode Set Size and Spatial Extent from the EEG data.
- Train and test classifiers separately on the lower and upper alpha band activity across different time windows during the maintenance phase.
- Statistical Comparison:
- Compare the time courses of ERD/ERS for lower vs. upper alpha.
- Compare the decoding accuracies for Set Size and Spatial Extent between the two sub-bands across early and late maintenance phases [64].
Signaling Pathways and Experimental Workflows
Alpha Rhythm Modulation in Memory Encoding
Closed-Loop Alpha Stimulation Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials and Reagents for Alpha Rhythm Research
| Item / Solution | Function / Application | Example / Specification |
|---|---|---|
| High-Density EEG System | Recording brain electrical activity with high spatial resolution. | 64-channel to 256-channel systems with active electrodes; impedance < 10 kΩ. |
| Electrolyte Gel / Paste | Ensures conductive connection between scalp and EEG electrodes. | Chloride-based, non-abrasive gels (e.g., SignaGel, Electro-Gel). |
| Closed-Loop Stimulation Software | Enables real-time EEG analysis and precise, phase-locked stimulus delivery. | OpenVIBE, Psychtoolbox, Brainstorm, or custom MATLAB/Python scripts. |
| Auditory Stimulation Equipment | Delivers phase-locked auditory stimuli without introducing electrical artifact. | MRI-compatible in-ear headphones or calibrated external speakers. |
| Visual Stimulation Presentation Software | Prescribes precise timing and content of cognitive tasks. | E-Prime, PsychoPy, Presentation, or custom code (JavaScript/Python). |
| EEG Preprocessing & Analysis Toolbox | Processes raw EEG data for artifact removal, filtering, and time-frequency analysis. | EEGLAB, FieldTrip, MNE-Python, Brainstorm. |
| Individual Alpha Frequency (IAF) Estimation Algorithm | Determines the subject-specific peak frequency for targeted stimulation. | Custom script to calculate power spectral density from resting-state EEG (eyes-closed). |
| Multivariate Decoding Software | Analyzes neural data to decode cognitive states (e.g., memory load). | Scikit-learn (Python), LIBSVM, or custom MVPA implementations in MATLAB. |
Electroencephalography (EEG), particularly posterior alpha power (8-13 Hz), is a key biomarker in memory encoding research due to its non-invasive nature and high temporal resolution. Its established role in reflecting attentional processes and thalamocortical interactions makes it highly relevant for studying cognitive functions and neurodegenerative diseases [33] [36]. However, its translation into clinical practice and drug development is hampered by significant challenges related to specificity, sensitivity, and individual variability. This article outlines these limitations and provides detailed protocols to help researchers address them in their experimental designs.
Key Limitations and Supporting Quantitative Data
The clinical application of EEG biomarkers faces specific, quantifiable hurdles. The table below summarizes the core limitations related to individual variability and signal specificity, synthesizing key findings from recent research.
Table 1: Key Limitations in the Clinical Application of EEG Biomarkers
| Limitation Category | Key Quantitative Findings | Clinical/Research Implication |
|---|---|---|
| Individual Variability & Long-Term Stability | Nonlinear EEG measures demonstrated greater one-year temporal stability (ICC) than band powers in healthy adults [69]. Some individuals exhibited annual fluctuations in band power comparable to or exceeding inter-individual variability [69]. | Group-level reliability does not preclude substantial within-subject variability, complicating the detection of true physiological changes in longitudinal studies [69]. |
| Signal Specificity & Confounding Factors | Posterior high-gamma power (51-100 Hz) positively correlated with memory recall, while posterior theta power was negatively correlated [33]. This inverse pattern highlights the frequency-specific nature of EEG biomarkers. HFA increases and alpha/beta decreases collectively marked successful memory encoding, indicating that multiple spectral features are involved [36]. | A single biomarker (e.g., posterior alpha) may be insufficient; multi-feature models are often necessary for accurate prediction [36]. Signals can be confounded by non-mnemonic processes related to list position or pre-motor activity [36]. |
| Subject-Level Consistency | Spectral markers of successful retrieval showed a higher degree of consistency across participants compared to encoding-related markers [36]. The HFA component of the subsequent memory effect showed notable variability across individuals [36]. | Classifiers and biomarkers may not generalize equally well across all individuals or between encoding and retrieval phases [36]. |
Detailed Experimental Protocols
To address the limitations above, rigorous experimental protocols are essential. The following sections provide detailed methodologies for assessing individual variability and optimizing EEG analysis for memory research.
Protocol for Assessing Long-Term Individual Variability
This protocol is designed to establish the stability of EEG measures within individuals over time, a prerequisite for using EEG as a longitudinal biomarker.
1. Objective: To evaluate the one-year temporal stability of linear and nonlinear EEG measures at the individual level. 2. Participants: Cohort of healthy adults; consider stratifying by age and sex. Patient cohorts should be well-characterized. 3. EEG Acquisition:
- Equipment: High-density EEG system (e.g., 19-channel or more).
- Setup: Resting-state, eyes-closed paradigm in a sound-attenuated room.
- Duration: Minimum 5 minutes of clean data per session.
- Schedule: 12 monthly recording sessions to capture intra-individual fluctuations [69]. 4. Data Processing & Analysis:
- Preprocessing: Apply standard filters (e.g., 0.5-70 Hz bandpass, 50/60 Hz notch). Perform artifact removal (e.g., ICA) and bad channel interpolation.
- Feature Extraction:
- Linear Measures: Compute absolute band power (Theta: 4-8 Hz, Alpha: 8-13 Hz, Beta: 13-30 Hz, Gamma: >30 Hz) for predefined regions of interest (ROIs), such as the posterior region [69] [33].
- Nonlinear Measures: Calculate complexity metrics like Higuchi’s Fractal Dimension (HFD) and Lempel-Ziv Complexity (LZC) [69].
- Statistical Analysis: Calculate Intraclass Correlation Coefficients (ICCs) to assess reliability across sessions for each measure and ROI. Plot individual trajectories of feature values over time to visualize within-subject variability [69].
Protocol for Memory Encoding-Specific EEG Analysis
This protocol employs advanced classification methods to isolate neural signals specific to memory success from other confounding neural processes.
1. Objective: To train a classifier that distinguishes neural correlates of successful memory encoding and retrieval while controlling for non-mnemonic confounds. 2. Task Design: Verbal delayed free-recall task.
- Encoding Phase: Participants study lists of words (e.g., 576 lists across multiple sessions) [36].
- Retrieval Phase: Participants freely recall words from the studied list after a distractor interval. Intrusions (recall of non-list words) are recorded. 3. EEG Acquisition:
- During Encoding: Epoch EEG data from the word presentation interval.
- During Retrieval: Epoch EEG data from the 500 ms immediately preceding vocalization of a recalled word [36]. 4. Data Analysis:
- Feature Engineering: Extract time-frequency power across all standard frequency bands from multiple scalp electrodes.
- Event Re-sampling: To control for serial position effects (e.g., primacy, recency), sub-sample encoding events so that the ratio of subsequently recalled vs. non-recalled items is matched across all list positions. Apply a similar re-sampling for retrieval events across output positions [36].
- Classifier Training & Validation:
- Use regularized logistic regression to predict subsequent memory performance during encoding and to distinguish correct recalls from intrusions during retrieval.
- Employ a leave-one-session-out cross-validation approach to ensure generalizability.
- Model Interpretation: Analyze classifier feature weights to identify the spectral and spatial features most critical for prediction (e.g., posterior alpha suppression, frontal theta increase) [36].
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials and Tools for EEG Memory Research
| Item Name | Function/Application | Specific Examples & Notes |
|---|---|---|
| High-Density EEG System | Captures electrical brain activity with high temporal resolution and sufficient spatial sampling. | 19-channel or 64-channel systems; Ensure amplifier has a suitable sampling rate (e.g., >500 Hz) for gamma activity [36] [66]. |
| Stimulus Presentation Software | Precisely controls the timing and delivery of memory task stimuli (words, images). | E-Prime, PsychoPy, or Presentation. Millisecond precision is critical for time-locking EEG epochs. |
| Automated Preprocessing Pipelines | Standardizes and streamlines data cleaning to reduce manual bias and improve reproducibility. | EEGLAB, MNE-Python, or FieldTrip. Use for filtering, artifact removal (ICA), and epoching. |
| Machine Learning Libraries | Enables the development of multivariate classifiers to decode memory states from EEG features. | Scikit-learn (for Python) or similar toolkits for implementing logistic regression and other classifiers [36]. |
| Interpretability Frameworks | Provides post-hoc explanations for classifier decisions, identifying impactful features. | SHAP (SHapley Additive exPlanations) or LIME; critical for moving beyond "black box" models and understanding biomarker contributions [70]. |
Workflow Visualization
The following diagrams, generated with Graphviz, illustrate the logical flow of the key experimental and analytical protocols described in this article.
Memory EEG Analysis Workflow
Individual Variability Assessment
Beyond Alpha: Integrating Spectral Signatures for a Holistic Memory Model
Electroencephalography (EEG) research has evolved beyond interpreting alpha oscillations (8-13 Hz) as a mere "idling rhythm." Contemporary evidence positions posterior alpha power as an active mechanism for inhibitory gating and temporal organization of neural processing [71] [72]. This inhibitory function complements the role of faster gamma oscillations (>30 Hz), typically associated with active information processing and feature binding. Within memory encoding research, this dynamic interplay creates windows of opportunity for information transfer and storage by selectively inhibiting task-irrelevant pathways and releasing relevant neural ensembles from inhibition [71]. This article details the application of this framework to experimental protocols investigating posterior alpha power in memory encoding.
Theoretical Framework & Signaling Pathways
The functional roles of alpha and gamma oscillations are realized through specific thalamocortical circuits. The following diagram illustrates the core signaling pathways involved in their generation and interaction.
Diagram Title: Neural Pathways for Alpha and Gamma Oscillation Generation
Pathway Description
- Alpha Generation: High-threshold bursting neurons in the thalamus, particularly the lateral geniculate nucleus, act as a pacemaker, generating rhythmic activity at alpha frequencies (8-13 Hz) [72]. This rhythm is synchronized via gap junctions and projected to the cortex, where it is amplified by pyramidal neurons in supragranular layers (I-III) [72]. Cortical GABAergic inhibitory interneurons further shape this rhythm, leading to pulsed inhibition that gates sensory processing [71] [72].
- Gamma Generation: Gamma oscillations are primarily generated in the cortex by the reciprocal interaction between pyramidal cells and fast-spiking GABAergic interneurons, facilitating local feature binding and active information processing [71].
- Cross-Frequency Coupling: The phase of alpha oscillations can modulate the amplitude of gamma activity, illustrating a mechanism by which pulsed inhibition (alpha) creates temporal windows for localized active processing (gamma) [71].
Quantitative Signatures in Memory Research
The interplay between alpha and gamma oscillations manifests in quantifiable EEG patterns that are predictive of memory encoding success.
Table 1: Key Oscillatory Signatures in Memory Encoding
| Oscillatory Metric | Protocol for Measurement | Functional Interpretation | Association with Memory Encoding |
|---|---|---|---|
| Posterior Alpha Power Decrease (ERD) | Calculate event-related desynchronization (ERD) in the alpha band (8-13 Hz) over occipital-parietal electrodes during stimulus presentation versus a pre-stimulus baseline [71] [72]. | Release of inhibition in visual cortical regions, facilitating sensory processing and resource allocation [71] [72]. | A stronger pre-stimulus alpha power decrease predicts better subsequent memory performance, indicating preparatory disinhibition [17]. |
| Alpha-Gamma Phase-Amplitude Coupling | Compute the modulation index between the phase of posterior alpha rhythms and the amplitude of gamma oscillations (30-100 Hz) in the same or connected regions [71]. | Pulsed inhibition (alpha phase) structuring periods of excitability for local cortical computation (gamma amplitude) [71]. | Effective memory encoding is associated with specific alpha phase windows that facilitate high gamma amplitude in sensory and associational cortices. |
| Lateralization of Alpha Power | Calculate a lateralization index (e.g., Contralateral ERD vs. Ipsilateral ERS) during visuospatial attention tasks using electrodes over left/right occipital cortices [71] [72]. | Active suppression of distracting visual input presented to the unattended hemifield (ipsilateral ERS) [71] [72]. | Successful encoding correlates with strong alpha lateralization, reflecting efficient suppression of irrelevant information to prevent interference [72]. |
| Trial-Level Alpha Fluctuations | Analyze single-trial pre-stimulus alpha power at centro-parietal sites and correlate it with subsequent memory performance on a trial-by-trial basis [17]. | Fluctuations in the state of preparatory attention and cortical excitability immediately before a to-be-remembered event [17]. | Trial-level fluctuations in pre-target alpha power predict the depth of processing and subsequent memory for new information, especially for discourse-focused words [17]. |
Experimental Protocols
Protocol 1: Assessing Pre-stimulus Alpha and Subsequent Memory (Dm Effect)
This protocol is based on recent research investigating spoken language comprehension and memory [17].
1. Research Question: How do trial-level fluctuations in pre-stimulus alpha power predict the subsequent memory (Dm effect) for information that is focused within a discourse context?
2. Experimental Design:
- Participants: Native language speakers with normal hearing.
- Stimuli: Spoken language narratives where target words are either focused or defocused by a preceding discourse question.
- Task: Participants listen to narratives for comprehension. A subsequent surprise recognition memory test is administered to assess memory for the target words.
- EEG Recording: Continuous EEG is recorded from 64+ scalp sites, with a focus on centro-parietal electrodes. Data is referenced to the average of all electrodes.
3. Data Analysis Workflow: The following diagram outlines the key steps from data acquisition to statistical modeling.
Diagram Title: Analysis Workflow for Pre-stimulus Alpha and Memory
4. Key Steps:
- Preprocessing: Filter data, remove artifacts using Independent Component Analysis (ICA), and epoch data from -1500 ms to +2000 ms around the onset of each target word.
- Time-Frequency Analysis: Use Morlet wavelet transform or similar on each epoch to extract power in the alpha band (8-13 Hz).
- Trial Sorting: For each participant, calculate the mean pre-stimulus alpha power (e.g., -500 ms to 0 ms) for each target word. Sort trials based on subsequent memory performance into "remembered" and "forgotten" conditions.
- Statistical Modeling: Employ linear mixed-effects (LME) modeling to test the effect of pre-stimulus alpha power (continuous predictor), discourse focus (categorical predictor), and their interaction on the probability of successful memory encoding, with random effects for subjects and items [17].
Protocol 2: Probing Inhibitory Gating via Alpha-Gamma Coupling
1. Research Question: Does the phase of posterior alpha oscillations modulate gamma power in sensory cortices during successful memory encoding?
2. Experimental Design:
- Stimuli: Presentation of visual objects for subsequent memory test.
- Task: Participants make a semantic judgment (e.g., "Is it living?") on each object.
- EEG Recording: High-density EEG (128 channels) is required for adequate spatial resolution of gamma activity.
3. Data Analysis:
- Preprocessing: Follow steps in Protocol 1, ensuring minimal filtering in the gamma range to avoid artifacts.
- Alpha Phase Extraction: Apply a bandpass filter in the individual alpha frequency (IAF) range to the signal from a posterior electrode (e.g., POz). Extract the instantaneous phase using the Hilbert transform.
- Gamma Amplitude Extraction: Filter the signal from visual electrodes (e.g., O1, Oz, O2) in a gamma band (e.g., 30-80 Hz). Extract the instantaneous amplitude envelope using the Hilbert transform.
- Coupling Calculation: Use the Modulation Index to quantify the strength of phase-amplitude coupling between the posterior alpha phase and visual cortex gamma amplitude. Compare this coupling strength between "remembered" and "forgotten" trials.
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials and Tools for EEG Memory Research
| Item / Solution | Function in Research | Example & Specification |
|---|---|---|
| High-Density EEG System | Records electrical potentials from the scalp with high temporal resolution. Essential for capturing event-related potentials and oscillatory dynamics. | Systems from Brain Products, BioSemi, or Electrical Geodesics Inc. with 64-128 channels or more. |
| Active Electrodes | Improve signal-to-noise ratio by amplifying the signal at the source, reducing interference from movement and cable noise. | Brain Vision actiCAP electrodes. |
| Electrode Gel / Paste | Ensures stable, low-impedance electrical connection between the scalp and EEG electrodes. | SignaGel from Parker Laboratories, Abralyt HiCl from EasyCap. |
| Presentation Software | Precisely controls the timing and presentation of experimental stimuli. | MATLAB with Psychtoolbox, E-Prime, Presentation. |
| Analysis Toolbox | Provides a suite of functions for preprocessing, time-frequency analysis, and statistical testing of EEG data. | EEGLAB, FieldTrip, MNE-Python. |
| Individual Alpha Frequency (IAF) Identification | Calculates the subject-specific peak alpha frequency, which is crucial for defining analysis bands that account for individual variability. | Custom scripts using FFT on resting-state data, or toolboxes like EEGLAB. |
The study of neural oscillations has revealed that cognitive functions, particularly working memory (WM), rely on the precise coordination of activity across different frequency bands, not just the activity within isolated bands. Phase-amplitude coupling (PAC), a specific type of cross-frequency coupling where the phase of a lower frequency rhythm modulates the amplitude of a higher frequency rhythm, is a critical mechanism for this coordination [73]. The coupling between the theta (4–8 Hz) phase and gamma (30–80 Hz) amplitude is especially vital for WM processes, including the encoding and maintenance of information [74] [73]. Concurrently, oscillations in the alpha band (8–12 Hz), particularly those generated in posterior cortical regions, play a fundamental role in gating information during memory encoding by inhibiting task-irrelevant brain areas [75] [76] [77].
This Application Note details the experimental and analytical protocols for investigating the interplay between posterior alpha oscillations and theta-gamma PAC. We frame this within the context of WM and memory encoding research, providing a practical guide for researchers and drug development professionals to quantify these neural phenomena and assess their functional significance in cognitive performance and potential dysregulation in clinical populations.
Theoretical Background and Significance
The Functional Roles of Alpha, Theta, and Gamma
Posterior Alpha Oscillations: Alpha oscillations are not merely a cortical "idling" rhythm. A growing consensus indicates that they serve a functional inhibitory role in cognition. During visuospatial WM tasks, a decrease in alpha power (alpha desynchronization) over occipital-parietal regions is required for the successful encoding of visual stimuli. This suppression is thought to facilitate sensory processing by releasing inhibition in task-relevant visual areas [75] [76]. Conversely, alpha synchronization may actively suppress distracting information. This modulation is a key biomarker; its attenuation has been documented in ADHD and serious mental illnesses (e.g., schizophrenia, bipolar disorder), where it is linked to WM encoding deficits [75] [77].
Theta-Gamma PAC (TGC): Theta and gamma oscillations form a coupled system crucial for WM. Theta rhythms, often dominant in the frontal cortex, are linked to the temporal organization of WM items, while gamma oscillations are associated with the active maintenance of individual memory representations [73]. Theta-gamma PAC is thought to be the mechanism that packages multiple gamma-nested memory items within a single theta cycle, thereby structuring the limited capacity of WM [74] [73]. Dysregulation of this coupling is observed in conditions like Mild Cognitive Impairment (MCI) and is a target for neuromodulation therapies [74].
The Integrated System
The interaction between alpha and the theta-gamma system is hypothesized to be a sequential gating-and-packaging mechanism. Posterior alpha desynchronization during encoding may permit sensory information to enter the memory system. Once encoded, this information is then structured and maintained via frontal-to-posterior theta-gamma PAC. Therefore, a failure in the initial alpha gating mechanism could lead to a cascade of impairments in downstream maintenance processes, ultimately manifesting as WM deficits [75].
Experimental Protocols for EEG Investigation
This section outlines a standardized protocol for a visuospatial working memory (SWM) task, suitable for investigating the roles of alpha and theta-gamma PAC.
Visuospatial Working Memory Task Protocol
Table 1: Visuospatial Working Memory Task Parameters
| Component | Specification | Rationale |
|---|---|---|
| Participants | Patients (e.g., ADHD, MCI, psychosis) and matched Healthy Controls (HCs) | Allows for comparison of neural mechanisms between clinical and neurotypical groups [75] [74] [77]. |
| Task Design | Computerized Spatial Working Memory (SWM) Task [75] or change detection task [74]. | Isolates specific WM phases (encoding, maintenance, retrieval). |
| Stimuli | Presentation of 1, 3, 5, or 7 yellow dots (or colored squares) on a screen for location memory [75]. | Manipulation of WM load. |
| Trial Structure | 1. Fixation (500 ms)2. Encoding (2000 ms): Stimuli presentation.3. Maintenance (3000 ms): Blank screen.4. Retrieval/Probe (3000 ms): Single dot appears; participant indicates if it matches a previous location. | Clearly demarcates cognitive phases for EEG analysis [75] [74]. |
| Behavioral Measures | Accuracy (%), Reaction Time (RT), and RT standard deviation (RTsd). | Provides performance metrics to correlate with neural data [75]. |
EEG Data Acquisition and Preprocessing
Table 2: EEG Acquisition and Analysis Parameters
| Parameter | Recommended Specification | Notes |
|---|---|---|
| EEG System | 64-channel cap (10-20 system) with active electrodes. | Ensures full scalp coverage and high-quality signal [28] [78]. |
| Reference | Common Average Reference (CAR) or linked ears. | CAR is common for analysis, though CSD is recommended for source separation [79]. |
| Sampling Rate | ≥ 500 Hz | Prevents aliasing and allows for gamma analysis. |
| Impedance | < 10 kΩ | Critical for signal quality [75]. |
| Filtering | Band-pass: 0.5 - 100 Hz; Notch: 50/60 Hz. | Removes line noise and slow drifts [74]. |
| Preprocessing | Automated and manual artifact rejection, Independent Component Analysis (ICA) for ocular and muscle artifacts. | ICA is highly effective for removing blinks and eye movements [75] [74]. |
| Epoching | Segmented into trials aligned to encoding, maintenance, and retrieval onsets. | Standard event-related potential/oscillation analysis. |
The following workflow diagram summarizes the experimental and analytical pipeline:
Figure 1: Experimental and Analytical Workflow for investigating cross-frequency dynamics in a working memory task.
Data Analysis Protocols
Analyzing Alpha Power Modulation
To analyze event-related changes in alpha power:
- Time-Frequency Decomposition: Apply methods like wavelet convolution or multitaper Fourier transform to EEG epochs from posterior electrodes (e.g., O1, Oz, O2, P3, Pz, P4) to compute power across frequencies and time.
- Baseline Correction: Normalize power during the task (e.g., encoding period) to a pre-stimulus baseline period (e.g., -500 to 0 ms before stimulus onset) to calculate percent change or decibel change.
- Extract Alpha Power: Average power within the 8–12 Hz band during key intervals (e.g., the first 500 ms of encoding) [75] [76].
- Statistical Analysis: Compare alpha power between groups (e.g., ADHD vs. controls) or conditions (high vs. low load) using t-tests or ANOVAs. Correlate alpha power with behavioral outcomes (e.g., WM accuracy, precision) and clinical symptom scores [75] [77].
Analyzing Theta-Gamma PAC
To quantify theta-gamma phase-amplitude coupling:
- Filtering: Band-pass filter the signal at the theta (e.g., 4–8 Hz) and gamma (e.g., 30–80 Hz) bands of interest.
- Extraction: Extract the phase time-series from the theta-filtered signal using the Hilbert transform. Extract the amplitude envelope from the gamma-filtered signal using the same method.
- Calculate Coupling Strength: Quantify the modulation of gamma amplitude by theta phase using the Modulation Index (MI). This involves assessing whether the distribution of gamma amplitude values across different theta phase bins is non-uniform [74] [73].
- Spatial and Temporal Focus: Analyze PAC separately for different task phases (encoding vs. maintenance) and brain regions (e.g., frontal theta with occipital gamma) [74].
Key Quantitative Findings and Their Interpretation
The following table synthesizes key empirical results from recent studies, highlighting the relationship between neural oscillations and cognition.
Table 3: Key Quantitative Findings on Alpha, Theta-Gamma, and Working Memory
| Study Population | Neural Finding | Behavioral Correlation | Clinical/Experimental Intervention |
|---|---|---|---|
| ADHD Children [75] | ↓ Alpha desynchronization during WM encoding. ↑ Occipital alpha during maintenance (compensatory). | Weak encoding alpha linked to ↓ executive function & ↑ ADHD symptoms. | N/A (Case-control study) |
| MCI Patients [74] | Dysregulated TGC; ↑ occipital gamma power. | ↑ Gamma correlated with ↓ WM performance. | 10 Hz rTMS to DLPFC ↓ gamma power, improved WM accuracy/capacity. |
| Healthy Adults [76] | ↑ Alpha power during encoding predicts ↑ precision of memory recall. | No change in guess rate, only precision. | N/A (Correlational study) |
| Serious Mental Illness [77] | ↓ Post-stimulus alpha suppression (encoding deficit). Prestimulus alpha not a primary mechanism. | Reduced suppression linked to ↓ likelihood of memory storage. | N/A (Case-control study) |
| Young vs. Old [73] | TGCp-tACS ↓ delta/theta power, ↑ high gamma. | Mixed effects: ↓ accuracy/RT in older adults; ↑ accuracy in young on some tasks. | Theta-gamma coupled tACS showed age-dependent behavioral effects. |
The Scientist's Toolkit: Research Reagent Solutions
Table 4: Essential Tools for Cross-Frequency Coupling Research
| Tool / Resource | Function / Purpose | Examples & Notes |
|---|---|---|
| High-Density EEG System | Recording electrical brain activity with high temporal resolution. | 64+ channel systems (e.g., Biosemi ActiveTwo, Neuracle). Critical for source localization and connectivity [74] [28]. |
| EEG Analysis Software | Preprocessing, time-frequency analysis, and PAC calculation. | EEGLAB, MNE-Python, FieldTrip. Custom MATLAB/Python scripts for MI calculation. |
| Current Source Density (CSD) | A reference-free technique to sharpen EEG topographies and localize activity. | Mitigates volume conduction; clarifies posterior alpha generators [79]. |
| Normative Databases (QEEG) | Benchmarking individual EEG metrics (e.g., alpha power) against a healthy population. | Used to identify significant deviations in clinical groups [78]. |
| Transcranial Magnetic Stimulation (TMS) | Non-invasive neuromodulation to test causal relationships. | 10 Hz rTMS to DLPFC can modulate TGC and improve WM in MCI [74]. |
| Transcranial Alternating Current Stimulation (tACS) | Entrain specific brain oscillations to probe function. | Theta-gamma cross-frequency tACS (TGCp-tACS) can directly manipulate PAC [73]. |
The interaction between posterior alpha power and frontal-posterior theta-gamma coupling represents a complex, multi-layered system for controlling and maintaining information in working memory. The provided protocols offer a roadmap for rigorously investigating this system. Future research should leverage causal interventions like TMS and tACS to move beyond correlation and establish direct mechanistic links. Furthermore, integrating these EEG metrics with pharmacological challenges holds great promise in drug development, providing sensitive, quantitative biomarkers for assessing the efficacy of novel compounds targeting cognitive deficits in neurological and psychiatric disorders. The application of these standardized protocols will enhance the reproducibility and translational potential of research in this rapidly advancing field.
Understanding the distinct contributions of neural oscillations in different brain regions is crucial for elucidating the mechanisms of memory. This application note focuses on the spatial and functional dissociation between posterior alpha (8-12 Hz) and frontal theta (4-8 Hz) rhythms during memory processes. Within the context of a broader thesis on EEG posterior alpha power in memory encoding research, we detail how these rhythms support dissociable aspects of memory function. We provide a synthesized analysis of quantitative findings, detailed experimental protocols for investigating these rhythms, and visualizations of their unique roles in memory networks, serving as a practical guide for researchers and drug development professionals.
Quantitative Data Synthesis
The following tables consolidate key quantitative findings from recent studies on posterior alpha and frontal theta oscillations, highlighting their predictive relationships with memory performance.
Table 1: Spectral Power Correlates of Memory Performance [33]
| Frequency Band | Brain Region | Memory Process | Direction of Correlation | Variance Explained (R²) |
|---|---|---|---|---|
| High-Gamma (51-100 Hz) | Temporal / Frontal | Immediate Recall (IR) & Delayed Recall (DR) | Positive | > 43% |
| High-Gamma (51-100 Hz) | Posterior | Immediate Recall (IR) & Delayed Recall (DR) | Negative | > 43% |
| Low-Gamma (30-49 Hz) | Temporal | Delayed Recall (DR) | Positive | Not Specified |
| Theta (4-8 Hz) | Posterior / Frontal | Immediate Recall (IR) | Positive | Significant |
Table 2: Frontal Theta and Theta/Beta Ratio in Memory Consolidation [80] [81]
| Oscillation Metric | Experimental Manipulation | Memory Process | Key Finding | Statistical Significance |
|---|---|---|---|---|
| Frontal-Midline Theta Power | Sequential WM with Spatial Regularity | Working Memory (WM) | Theta power increased monotonically with sequence complexity. | p < .05 |
| Fronto-Posterior Theta Connectivity | Sequential WM with Spatial Regularity | Working Memory (WM) | Connectivity modulated by sequence regularity, but not scenario difficulty. | p < .05 |
| Theta/Beta Ratio (TBR) | Post-learning Neurofeedback Upregulation | Spatial & Long-Term Memory | Increased TBR led to faster task completion times (performance gain) up to one week. | p < .05 |
Experimental Protocols
Protocol for Investigating Posterior Alpha in Memory Encoding
This protocol is designed to assess the role of posterior alpha power as an inhibitory mechanism during memory encoding [33].
- Objective: To determine the relationship between resting-state posterior alpha power and subsequent memory performance for visual and verbal stimuli.
- Participants: 20-30 healthy adult volunteers, screened for neurological/psychiatric history.
- Stimuli & Task:
- Stimuli: A set of 200 unique items, including concrete nouns (verbal) and line drawings of common objects (visual).
- Encoding Task: Participants make animacy judgments ("Is this object/living or non-living?") for each presented stimulus.
- Memory Test: Following a 20-minute delay filled with a distractor task, participants complete a recognition memory test containing old and new items.
- EEG Acquisition:
- Setup: 64-channel EEG system with active electrodes.
- Resting-State: 5 minutes of eyes-closed rest, followed by 5 minutes of eyes-open rest, performed before the task.
- Task-Related: Continuous EEG recorded during the encoding phase.
- Preprocessing: Band-pass filtering (0.1-100 Hz), artifact removal (ocular, muscle, cardiac), and bad channel interpolation.
- Data Analysis:
- Spectral Analysis: Compute power spectral density for resting-state data. For task data, segment epochs from -1000 ms to 1500 ms relative to stimulus onset.
- Grouping: Separate encoding trials based on subsequent memory performance into "Remembered" and "Forgotten" conditions.
- Statistical Test: Use cluster-based permutation tests to compare alpha power (8-12 Hz) between conditions, focusing on posterior electrodes (Pz, P3, P4, POz, O1, O2).
Protocol for Investigating Frontal Theta in Working Memory
This protocol examines how frontal theta oscillations support the processing of spatial sequences and respond to cognitive load [81].
- Objective: To investigate the modulation of frontal-midline theta (FM-theta) power and connectivity by spatial regularity in a sequential working memory task.
- Participants: 25-35 healthy adult volunteers.
- Stimuli & Task - Sequential WM with Spatial Regularity:
- Stimuli: A sequence of dots appears at different locations on a screen. The spatial relation between successive items is manipulated.
- Manipulation: "High Regularity" sequences contain more repetitions of the same spatial displacement, forming fewer unique "chunks." "Low Regularity" sequences have more unique displacements.
- Task: Participants encode, maintain, and then reproduce the temporal order of the spatial sequence.
- Scenario: The task is embedded in both a simple (focused layout) and a complex scenario (meaningful context, dispersed layout) to assess ecological validity.
- EEG Acquisition:
- Setup: High-density EEG system (e.g., 128 channels).
- Recording: Continuous EEG during the entire task, with markers for encoding, maintenance, and retrieval phases.
- Data Analysis:
- Time-Frequency Decomposition: Calculate power in the 4-30 Hz range during the delay (maintenance) period.
- Connectivity Analysis: Compute phase-based connectivity (e.g., Phase-Locking Value) between frontal (Fz) and posterior (Pz) electrodes in the theta band.
- Statistical Model: Use repeated-measures ANOVA to test the effects of "Regularity" (High vs. Low) and "Scenario" (Simple vs. Complex) on FM-theta power and fronto-posterior theta connectivity.
Signaling Pathways and Workflow Visualizations
Neural Mechanism of Posterior Alpha
Neural Mechanism of Frontal Theta
Experimental Workflow for Protocol 3.1
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials and Equipment for EEG Memory Research
| Item | Function / Rationale | Example Specification / Note |
|---|---|---|
| High-Density EEG System | Records electrical brain activity from the scalp with high spatial resolution. | 64+ channels; active electrodes recommended for reduced noise [33] [81]. |
| Electroencephalography (EEG) | Core technique for measuring oscillatory power and functional connectivity. | N/A |
| Quantitative EEG (QEEG) Software | Enables numerical analysis of spectral power, coherence, and source localization. | Used for analyzing spectral properties like alpha asymmetry and theta/beta ratio [9]. |
| Stimulus Presentation Software | Precisely controls the timing and presentation of experimental stimuli. | Software like PsychoPy, E-Prime, or Presentation. |
| Virtual Reality Platform (e.g., Minecraft) | Provides an immersive, ecologically valid environment for spatial navigation and memory tasks [80]. | Customizable virtual worlds for assessing real-life episodic memory. |
| Neurofeedback (NFB) System | Allows real-time modulation of brain oscillations to test causal roles in memory. | Used to upregulate Theta/Beta ratio post-learning to enhance consolidation [80]. |
| Cognitive Reserve Index (CRI) Questionnaire | Assesses an individual's resilience to cognitive decline, a key covariate. | Should be administered to participants to control for confounding factors [33]. |
Within electrophysiological research on human memory, two robust neural signatures have emerged: posterior alpha oscillations (8-13 Hz) and the event-related potential (ERP) component known as the Late Positive Component (LPC). Individually, they are established markers of cognitive processes, yet their interrelationship is a critical area of investigation for understanding the brain's dynamical system. This application note details the empirical links between these signals, places them within the theoretical framework of memory encoding, and provides standardized protocols for researchers, particularly in clinical and drug development settings, to quantify their interaction.
The LPC (or left parietal old/new effect), observed between 500–800 ms post-stimulus over posterior parietal electrodes, is a well-validated neural correlate of episodic recollection, sensitive to the retrieval of contextual details and the overall quality of memory [82]. Concurrently, posterior alpha oscillations reflect a top-down inhibitory mechanism that suppresses distracting sensory input and gates information flow to facilitate cognitive operations [4] [83]. Framing these signals within the context of a dynamical system theory suggests that spontaneous brain activity (e.g., alpha rhythms) is intrinsically linked to and shapes evoked responses (e.g., the LPC) [84].
Empirical Evidence and Quantitative Data
The relationship between alpha power and the LPC is not a simple inverse correlation but is nuanced, depending on brain state, timing, and cognitive domain. The table below summarizes key quantitative findings from the literature.
Table 1: Summary of Key Empirical Findings on Alpha-LPC Relationships
| Cognitive Domain | Alpha Power Effect | LPC Effect | Observed Relationship | Key Reference |
|---|---|---|---|---|
| Long-Term Memory Encoding | ↑ Posterior alpha during rehearsal predicts successful encoding [6]. | Larger amplitude for successfully encoded items [82]. | High anticipatory alpha facilitates encoding, reflected in a larger subsequent LPC during retrieval. | Hanslmayr et al., 2011 [6] |
| Episodic Recollection | Alpha/beta desynchronization during retrieval correlates with memory precision [82]. | Amplitude is sensitive to overall memory quality and spatial precision [82]. | Alpha desynchronization and LPC amplitude are correlated, reflecting complementary aspects of recollection. | Cooper & Ritchey, 2025 [82] |
| Cross-Individual Variability | Higher resting-state alpha amplitude. | Stronger brain response magnitude (P3). | Individuals with higher resting alpha show stronger evoked responses, but only when confounding factors are controlled [84]. | PMC, 2025 [84] |
| Working Memory Maintenance | Increased posterior alpha during retention. | Not typically measured in maintenance. | Alpha serves to inhibit task-irrelevant areas, creating an optimal state for memory integrity, which may be reflected in later retrieval-related LPC. | Freunberger et al., 2010 [6] |
| Attentional Inhibition | ↑ Alpha in sensory cortices processing distracting information [83]. | N/A | Demonstrates the inhibitory gating role of alpha, a proposed mechanism that influences the content available for later recollection (LPC). | Foxe & Snyder, 2011 [83] |
Experimental Protocols
This section provides detailed methodologies for experiments that can simultaneously capture alpha oscillations and the LPC.
Cued Long-Term Memory Encoding Task
This protocol is adapted from studies investigating the gating function of alpha and its effect on subsequent memory as measured by the LPC [4] [6].
Objective: To determine how anticipatory alpha power following a memory cue predicts subsequent memory performance and retrieval-related LPC.
Materials:
- Stimuli: A large set of images (e.g., landscapes, buildings) or words.
- EEG System: 64-channel (or higher) EEG system with active electrodes.
- Software: Stimulus presentation software (e.g., PsychoPy, E-Prime), EEG recording software, and analysis tools (e.g., EEGLAB, FieldTrip, MNE-Python).
Table 2: Research Reagent Solutions for Cued LTM Task
| Item | Specification / Function |
|---|---|
| EEG Acquisition System | 64+ channel system; recommended impedance < 10 kΩ. |
| Stimulus Set | 600+ trial-unique images/words to avoid repetition effects. |
| Electrode Caps | Standard 10-10 or 10-5 system placement. |
| Analysis Software | EEGLAB/FieldTrip for time-frequency & ERP analysis. |
Procedure:
- Task Design:
- Each trial begins with a fixation cross (500 ms).
- A cue is presented (e.g., colored fixation cross) for 2000 ms, instructing the participant to either "Remember" or "No-Remember" the upcoming item.
- A stimulus (image or word) is presented for 1000 ms.
- This is followed by an inter-trial interval (ITI) of 1000-1500 ms.
- Memory Test:
- After a delay filled with a distractor task, a recognition test is administered.
- Participants are shown old and new stimuli and must indicate "old"/"new" and potentially their confidence or recollection of details.
- EEG Recording:
- Record continuous EEG at a sampling rate ≥ 500 Hz.
- Ensure proper grounding and reference electrode placement.
- Data Analysis:
- Preprocessing: Filter data (e.g., 0.1-100 Hz), re-reference to average mastoids, segment into epochs, and perform artifact rejection (e.g., ocular, muscle).
- Alpha Power: Time-frequency analysis (e.g., using Morlet wavelets) on the cue period (-500 to 2000 ms relative to cue onset). Extract alpha power (8-13 Hz) from posterior electrodes (e.g., Pz, P3, P4, O1, Oz, O2) during the final 500 ms of the cue period.
- LPC Analysis: ERP analysis on the test period. Filter data (e.g., 0.1-30 Hz), segment (-200 to 1000 ms relative to test stimulus onset), and baseline correct. Measure the mean amplitude of the LPC (500-800 ms) at parietal electrodes (e.g., Pz).
- Statistics: Correlate cue-period alpha power with subsequent LPC amplitude for correctly remembered "Remember" trials. Compare alpha power and LPC amplitude between "Remember" vs. "No-Remember" trials and between subsequently remembered vs. forgotten items.
Experimental workflow for the cued long-term memory task, highlighting the time points for alpha and LPC extraction.
Memory Retrieval with Continuous Measures
This protocol leverages continuous measures of memory precision to investigate graded relationships between alpha/beta desynchronization and the LPC [82].
Objective: To assess how the precision of retrieved memory features modulates alpha/beta oscillatory power and the amplitude of the LPC.
Materials:
- Stimuli as described in [82] (items with multiple continuous features like color, spatial location in a 360° panorama, and sound).
- Equipment and software as in Protocol 3.1.
Procedure:
- Encoding Phase:
- Present participants with trial-unique items, each possessing multiple features (e.g., a specific color, spatial location, and sound).
- Retrieval Phase:
- Present a cue to recall a specific feature of a studied item (e.g., "Recall the location").
- Participants provide a continuous response (e.g., adjusting a dial to indicate the remembered location on a circle).
- EEG Recording:
- Record continuous EEG during the retrieval phase, focusing on the period before and during the retrieval cue and response.
- Data Analysis:
- Behavioral Data: Calculate precision (error) for each retrieved feature.
- Oscillatory Power: Calculate alpha/beta (8-30 Hz) power during the retrieval epoch. Use regression models to test if power desynchronization predicts retrieval precision for each feature.
- LPC Analysis: Extract the LPC (500-800 ms) time-locked to the retrieval cue onset. Test if LPC amplitude is graded by overall memory quality or precision of specific features.
- Integration: Perform mediation analysis to test for a potential relationship where alpha desynchronization during later retrieval stages mediates the effect of early retrieval processes (indexed by the LPC) on memory precision.
Signaling Pathways and Logical Workflow
The following diagram synthesizes the proposed neuro-cognitive pathway linking alpha oscillations to the generation of the LPC, based on the reviewed evidence.
A proposed neuro-cognitive model linking alpha oscillations to the LPC. Top-down control induces alpha rhythms to inhibit interference and gate encoding. Successful gating leads to precise memory traces, the reactivation of which during retrieval involves alpha/beta desynchronization and generates the LPC.
Advanced Analysis Considerations
- Separating Periodic and Aperiodic Activity: When analyzing alpha oscillations, it is critical to separate the periodic (oscillatory) component from the 1/f aperiodic background signal. Conflating the two can lead to spurious findings [84]. Use tools like the FOOOF algorithm to parameterize the power spectrum.
- Trial-Level Variability and Adaptation: Brain responses adapt across trials. The response to the first stimulus in a block is often more representative of the brain's genuine response and may show a clearer relationship with spontaneous activity like alpha [84]. Consider analyzing early trials separately.
- Multivariate Pattern Analysis (MVPA): Beyond traditional univariate analyses, MVPA can decode specific feature information (e.g., color, shape) from EEG signals during working and long-term memory tasks, providing a more detailed view of content-specific representations that may be governed by alpha/LPC dynamics [85] [36].
By implementing these protocols and adhering to the analytical considerations outlined, researchers can rigorously investigate the interplay between alpha oscillatory mechanisms and the LPC, advancing our understanding of human memory and providing robust biomarkers for evaluating cognitive function in health and disease.
The formation of durable memories represents a complex neural achievement requiring precisely coordinated interactions across distributed brain networks. Within this framework, oscillations in the alpha (8-12 Hz), theta (4-8 Hz), and gamma (30-200 Hz) bands play complementary and functionally specific roles. This application note synthesizes current evidence into a unified model of how these rhythms orchestrate successful memory encoding, with particular emphasis on EEG posterior alpha power as a critical gating mechanism. The model posits that successful encoding emerges from cross-frequency interactions where theta rhythms provide a temporal organizing framework, gamma rhythms facilitate local information processing, and alpha rhythms actively inhibit task-irrelevant regions to minimize interference [6] [86] [87]. This integrated perspective provides researchers with a neurophysiological framework for developing biomarkers and therapeutic interventions targeting memory function.
Quantitative Synthesis of Oscillatory Correlates
Table 1: Spectral Signatures of Successful Memory Encoding and Retrieval
| Frequency Band | Direction for Success | Brain Regions | Functional Role | Key Supporting Evidence |
|---|---|---|---|---|
| Posterior Alpha (8-12 Hz) | ↑ Power during rehearsal | Parieto-occipital cortex | Inhibits task-irrelevant visual areas to reduce interference | MEG study: Stronger posterior alpha during rehearsal predicts successful LTM formation [6] |
| Frontal Theta (4-8 Hz) | ↑ Power & synchronization | Prefrontal cortex, Hippocampus | Provides temporal framework for encoding; supports interhemispheric integration | tACS study: Theta-gamma cross-frequency stimulation enhances working memory [87] |
| Gamma (30-100 Hz) | ↑ Power & precision timing | Neocortical layers, Prefrontal cortex | Facilitates local feature binding and information processing | Laminar recordings: Gamma amplitude coupled to alpha phase in superficial layers [86] |
| Alpha-Theta-Gamma Coupling | ↑ Cross-frequency coupling | Prefrontal-visual-hippocampal network | Enables hierarchical information processing across scales | Residual network: Theta-gamma tACS boosts memory more than theta alone [87] |
Table 2: Quantitative EEG Markers of Mnemonic Success from Recent Studies
| Experimental Paradigm | Spectral Effect Size | Temporal Dynamics | Spatial Topography | Practical Applications |
|---|---|---|---|---|
| Subsequent Memory Effect | Decreased 10-30 Hz power during encoding predicts recall [36] | 1.6s encoding epochs; re-sampling controls for serial position | Anterior-superior HFA increases; posterior low-frequency decreases | Biomarker for encoding strength; target for neurofeedback |
| Retrieval Success | Increased HFA distinguishes correct recalls from intrusions [36] | 500ms pre-vocalization period; matches output time | Distributed cortical networks with HFA increases | Differentiates true vs. false memory; intrusion error prediction |
| Working Memory Maintenance | Theta-gamma cross-frequency coupling improves performance [87] | Gamma bursts locked to theta peaks (80-100 Hz most effective) | Prefrontal cortex critical for spatial working memory | Non-invasive stimulation target for cognitive enhancement |
| Long-Term Memory Formation | Posterior alpha power increase during rehearsal intervals [6] | 3.4s rehearsal period following word sequence presentation | Parieto-occipital regions show strongest effects | Neurofeedback protocol development for memory disorders |
Proposed Unified Model: Mechanisms and Interactions
The unified model integrates three core mechanisms through which alpha, theta, and gamma oscillations collectively enable successful memory encoding:
Alpha-Mediated Functional Inhibition
Posterior alpha power serves an active inhibitory role during memory formation by suppressing task-irrelevant visual processing regions. This creates an optimal brain state for encoding by minimizing potential interference from extraneous sensory input [6]. The model specifies that high parieto-occipital alpha activity during rehearsal periods is particularly crucial for successful long-term memory formation, possibly by actively reducing parieto-occipital activity that might otherwise interfere with the cognitive processes required for sequence encoding [6].
Theta-Gamma Cross-Frequency Coupling
Theta oscillations provide a temporal scaffold that organizes gamma-band activity into discrete processing units. Evidence demonstrates that gamma bursts occurring at the peak of theta waves significantly enhance working memory performance, whereas gamma activity occurring during theta troughs proves ineffective [87]. This phase-specific relationship is most effective in the 80-100 Hz high gamma range and enhances global neocortical connectivity, suggesting a mechanism for integrating information across distributed brain regions [87].
Laminar-Specific Cross-Frequency Hierarchy
A hierarchical organization exists across cortical layers, where alpha phase in infragranular layers modulates gamma amplitude in granular and supragranular layers [86]. This inter-laminar coupling creates a top-down control mechanism where deeper layer alpha rhythms phasically inhibit superficial layer processing, demonstrating an anticorrelated relationship between alpha and gamma power across cortical layers [86].
Experimental Protocols and Methodologies
Protocol 1: Assessing Subsequent Memory Effects Using EEG
Objective: To identify spectral features predicting successful memory encoding.
Experimental Design:
- Participants complete a delayed free recall task with 576 word lists across multiple sessions [36]
- EEG recorded during 1.6s word encoding epochs and 500ms pre-recall periods
- Critical innovation: Implement event re-sampling to equalize contribution of recalled and non-recalled items across list positions [36]
Data Analysis Pipeline:
- Preprocessing: Standard EEG preprocessing including filtering, artifact removal, and referencing
- Spectral Feature Extraction: Compute time-frequency representations for encoding and retrieval epochs
- Classifier Training: Train logistic regression classifiers on spectral features to predict subsequent recall success
- Validation: Use held-out sessions for validation rather than held-out lists to assess generalizability
Key Controls:
- Match temporal distributions of correct recalls and intrusion errors
- Control for serial position effects in encoding analysis
- Control for output position effects in retrieval analysis [36]
Protocol 2: Cross-Frequency tACS for Memory Enhancement
Objective: To causally enhance working memory through theta-gamma transcranial alternating current stimulation (tACS).
Stimulation Parameters:
- Target region: Prefrontal cortex
- Theta component: 4-8 Hz continuous stimulation
- Gamma component: 80-100 Hz bursts synchronized to theta peaks [87]
- Control conditions: Theta-only tACS; gamma on theta troughs; low gamma protocols
Assessment Metrics:
- Spatial working memory performance pre-/post-stimulation
- Global neocortical connectivity changes via EEG
- Comparison of different gamma frequencies (30-50 Hz vs. 80-100 Hz) [87]
Implementation Considerations:
- Gamma bursts must precisely coincide with theta peaks for maximal effect
- High gamma frequencies (80-100 Hz) show superior efficacy to low gamma
- Effect specific to phase relationship, not just presence of both frequencies [87]
Visualization of Core Model Components
Unified Model of Oscillatory Interactions
Laminar Organization of Cross-Frequency Coupling
Experimental Workflow for Memory Encoding Research
Table 3: Research Reagent Solutions for Oscillatory Memory Research
| Tool/Category | Specific Examples | Function & Application | Key Considerations |
|---|---|---|---|
| Neurostimulation Platforms | tACS systems; GENUS (Gamma Entrainment Using Sensory stimuli) | Causally manipulate oscillatory activity; test functional roles | Phase precision critical for cross-frequency protocols [87] [88] |
| EEG/MEG Systems | High-density EEG (32+ channels); MEG systems with axial gradiometers | Record neural oscillations with high temporal resolution | MEG optimal for posterior alpha sources [6]; EEG sufficient for scalp-level analysis [36] |
| Analysis Toolboxes | EEGLAB; FieldTrip; MNE-Python; LIMO EEG | Preprocess data, compute time-frequency representations, statistical analysis | Different toolboxes have complementary strengths for visualization [89] |
| Classification Frameworks | Logistic regression; Deep ConvNets; FBCSP (Filter Bank Common Spatial Patterns) | Decode memory states from spectral features; identify predictive patterns | Deep ConvNets enable end-to-end learning from raw EEG [90] |
| Spectral Analysis Tools | Wavelet transforms; FFT algorithms; Phase-amplitude coupling metrics | Quantify power, phase, and cross-frequency interactions | Critical for identifying theta-gamma and alpha-gamma coupling [86] [87] |
| Neurofeedback Systems | Real-time EEG processing; BCI++ platform; Custom feedback displays | Train participants to self-regulate oscillatory activity | Effective for alpha/theta modulation in older adults [91] |
The unified model presented herein establishes a foundational framework for understanding how alpha, theta, and gamma oscillations collectively orchestrate successful memory encoding. The empirical evidence demonstrates that these frequency bands do not operate in isolation but instead engage in precisely coordinated interactions across multiple spatial and temporal scales. From a translational perspective, this oscillatory triad offers promising targets for therapeutic interventions in populations with memory impairment. The specific role of posterior alpha power as an active inhibitory mechanism provides a parsimonious explanation for how the brain allocates resources during encoding by suppressing competing visual processing. Future research should prioritize developing closed-loop systems that dynamically adapt to individual oscillatory profiles, potentially yielding more effective and personalized cognitive enhancement strategies.
Conclusion
Posterior alpha power is established as a critical neural mechanism for successful memory encoding, primarily through the active inhibition of distracting sensory input. Its reliability as a biomarker is evidenced by its predictive value in subsequent memory paradigms and its sensitivity to clinical conditions characterized by memory and attention deficits. Future research must focus on standardizing measurement protocols, elucidating the causal role of alpha via neuromodulation, and developing integrated models that account for its interaction with other spectral features. For biomedical research, this holds profound implications for developing objective diagnostic tools for neurocognitive disorders and validating the efficacy of pharmacological and neurostimulation therapies aimed at enhancing memory function.
References
- 1. Two Spatially Distinct Posterior Alpha Sources Fulfill Different ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6733553/]
- 2. Resting-state posterior alpha power changes with prolonged ... [https://cognitiveresearchjournal.springeropen.com/articles/10.1186/s41235-020-00247-0]
- 3. Temporal–Posterior Alpha Power in Resting-State ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11201643/]
- 4. Blocking of irrelevant memories by posterior alpha activity ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6869719/]
- 5. Alpha power increases in right parietal cortex reflects ... [https://www.sciencedirect.com/science/article/pii/S0028393214000621]
- 6. Increase in posterior alpha activity during rehearsal ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6870165/]
- 7. Load-dependent alpha suppression is related to working ... [https://www.sciencedirect.com/science/article/abs/pii/S0006899322002189]
- 8. Neuronal generators of posterior EEG alpha reflect individual ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3893140/]
- 9. Quantitative and qualitative electroencephalography in the ... [https://www.frontiersin.org/journals/human-neuroscience/articles/10.3389/fnhum.2025.1624434/full]
- 10. In older adults resting-state alpha power is associated with ... [https://www.sciencedirect.com/science/article/pii/S0966636225006769]
- 11. Parieto‐occipital sources account for the increase in alpha ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6871495/]
- 12. A framework for analyzing EEG data using high ... [https://pubmed.ncbi.nlm.nih.gov/40100304/]
- 13. Prestimulus α/β power in temporal-order judgments [https://www.frontiersin.org/journals/computational-neuroscience/articles/10.3389/fncom.2023.1145267/full]
- 14. The Journey of the Default Mode Network - PubMed Central [https://pmc.ncbi.nlm.nih.gov/articles/PMC12025022/]
- 15. 20 years of the default mode network: A review and synthesis [https://www.sciencedirect.com/science/article/pii/S0896627323003082]
- 16. Increase in slow frequency and decrease in alpha and beta ... [https://www.sciencedirect.com/science/article/pii/S0010945224003253]
- 17. Discourse Focus and Memory Encoding: The Role of Trial ... [https://pubmed.ncbi.nlm.nih.gov/40631241/]
- 18. Temporal dynamics of attention during encoding vs ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4678590/]
- 19. Alpha activity during verbal working memory retention ... [https://sciety-discovery.elifesciences.org/articles/by?article_doi=10.1101/2025.05.04.652111]
- 20. Does rehearsal matter? Left anterior temporal alpha and theta ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8102380/]
- 21. Blocking of irrelevant memories by posterior alpha activity ... [https://pubmed.ncbi.nlm.nih.gov/24522937/]
- 22. The involvement of alpha oscillations in voluntary attention ... [https://www.sciencedirect.com/science/article/abs/pii/S105381191730900X]
- 23. Electroencephalography source localization - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC10167408/]
- 24. Functional Connectivity of the Dorsal and Ventral Attention ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12476866/]
- 25. Resting-State Functional Connectivity in the Dorsal ... [https://www.frontiersin.org/journals/human-neuroscience/articles/10.3389/fnhum.2021.757128/full]
- 26. Emerging panorama of functional near-infrared spectroscopy ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12426634/]
- 27. fNIRS reproducibility varies with data quality, analysis ... [https://www.nature.com/articles/s42003-025-08412-1]
- 28. Fine Structure of Posterior Alpha Rhythm in Human EEG [https://www.nature.com/articles/s41598-017-08421-z]
- 29. Qualitative and Quantitative Comparative Analysis of ... [https://pubmed.ncbi.nlm.nih.gov/41042404/]
- 30. Precise spatial tuning of visually driven alpha oscillations ... [https://elifesciences.org/articles/90387]
- 31. The applied principles of EEG analysis methods in ... [https://mmrjournal.biomedcentral.com/articles/10.1186/s40779-023-00502-7]
- 32. November 2025 Newsletter [https://www.xylo.bio/blog-posts/november-2025-newsletter]
- 33. Resting-state EEG gamma power predicts immediate and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12381311/]
- 34. Computational methods of EEG signals analysis for ... [https://www.nature.com/articles/s41598-023-32664-8]
- 35. Subsequent memory effect in intracranial and scalp EEG - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC3849113/]
- 36. EEG decoders track memory dynamics [https://www.nature.com/articles/s41467-024-46926-0]
- 37. Decoding memory with explainable AI: A large-scale EEG- ... [https://www.sciencedirect.com/science/article/pii/S2772528625000421]
- 38. Decoding covert visual attention of ... [https://www.nature.com/articles/s41598-025-21635-w]
- 39. A Deep Learning Approach to Alzheimer's Diagnosis Using ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12383294/]
- 40. The roles of alpha oscillation in working memory retention - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC6456781/]
- 41. Visual Working Memory Recruits Two Functionally Distinct ... [https://www.eneuro.org/content/9/5/ENEURO.0159-22.2022]
- 42. Successful memory encoding is associated with increased ... [https://www.sciencedirect.com/science/article/abs/pii/S105381191201097X]
- 43. Prediction of Alpha Power Using Multiple Subjective ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11898570/]
- 44. Biomarkers and Neuropsychological Tools in Attention ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12300379/]
- 45. Electroencephalographic Biomarkers for Neuropsychiatric ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11939446/]
- 46. Can biomarkers be used to diagnose attention deficit ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10030688/]
- 47. Towards predicting posttraumatic stress symptom severity ... [https://www.nature.com/articles/s41598-025-88426-1]
- 48. Multimodal biomarker based on temporal complexity of eye ... [https://journals.plos.org/mentalhealth/article?id=10.1371/journal.pmen.0000456]
- 49. Evidence for Attentional Gradient in the Serial Position ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2748728/]
- 50. Cognitive load detection through EEG lead wise feature ... [https://www.nature.com/articles/s41598-024-84429-6]
- 51. Multiple-Time-Scale Analysis of Attention as Revealed by ... [https://www.frontiersin.org/journals/neuroscience/articles/10.3389/fnins.2019.01307/full]
- 52. EEG decoders track memory dynamics - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC10998865/]
- 53. Identifying causal subsequent memory effects - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC10068819/]
- 54. How to control for confounds in decoding analyses of ... [https://www.sciencedirect.com/science/article/pii/S1053811918319463]
- 55. EEG Alpha Power Is Modulated by Attentional Changes ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6614966/]
- 56. EEG alpha power during maintenance of information in ... [https://www.sciencedirect.com/science/article/abs/pii/S1388245715009888]
- 57. EEG alpha power during maintenance of information in ... [https://pubmed.ncbi.nlm.nih.gov/26541307/]
- 58. Alpha neurofeedback training improves visual working ... [https://www.nature.com/articles/s41539-024-00242-w]
- 59. Working Memory Training and Cortical Arousal in Healthy ... [https://www.frontiersin.org/journals/aging-neuroscience/articles/10.3389/fnagi.2021.718965/full]
- 60. The Effects of Electroencephalogram Feature-Based ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8961031/]
- 61. Working memory processes and intrinsic motivation [https://www.sciencedirect.com/science/article/pii/S016787602400059X]
- 62. Modulation of aperiodic EEG activity provides sensitive ... [https://elifesciences.org/articles/101071]
- 63. Editorial: EEG rhythms: decoding the evolutionary enigma ... [https://www.frontiersin.org/journals/human-neuroscience/articles/10.3389/fnhum.2025.1560294/full]
- 64. Dissociating the roles of alpha oscillation sub-bands in ... [https://www.sciencedirect.com/science/article/pii/S1053811925000308]
- 65. The Role of Quantitative EEG in the Diagnosis of Alzheimer's ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12346855/]
- 66. Research progress of quantitative electroencephalography ... [https://www.frontiersin.org/journals/neurology/articles/10.3389/fneur.2025.1445962/full]
- 67. Lateralised modulation of posterior alpha oscillations by ... [https://pubmed.ncbi.nlm.nih.gov/40928755/]
- 68. Study protocol for evaluating EEG-based predictive model ... [https://www.medrxiv.org/content/10.1101/2025.04.26.25326496v1.full-text]
- 69. Individual stability of single-channel EEG measures over ... [https://www.nature.com/articles/s41598-025-13614-y]
- 70. A systematic review of EEG-based machine learning ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12421762/]
- 71. Alpha-band oscillations, attention, and controlled access to ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3507158/]
- 72. Alpha Wave - an overview | ScienceDirect Topics [https://www.sciencedirect.com/topics/neuroscience/alpha-wave]
- 73. Theta-gamma-coupling as predictor of working memory ... [https://molecularbrain.biomedcentral.com/articles/10.1186/s13041-024-01149-8]
- 74. leveraging theta-gamma coupling for cognitive rehabilitation ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12075380/]
- 75. Alpha modulation during working memory encoding predicts ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6640106/]
- 76. Modulation of alpha power at encoding and retrieval tracks ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4254886/]
- 77. Dysfunctional Alpha Modulation as a Mechanism of ... [https://www.sciencedirect.com/science/article/pii/S2451902224002106]
- 78. Essential 2025 QEEG Guidelines IQCB Technical ... [https://neurofeedback-luxembourg.com/understanding-2025-qeeg-iqcb-technical-requirements-reporting-guidelines-for-effective-neurofeedback/]
- 79. Posterior EEG alpha at rest and during task performance [https://pmc.ncbi.nlm.nih.gov/articles/PMC4537800/]
- 80. The role of post-learning EEG theta/beta ratio in long-term ... [https://www.sciencedirect.com/science/article/abs/pii/S1074742725000577]
- 81. Frontal Theta Modulation in Sequential Working Memory [https://pubmed.ncbi.nlm.nih.gov/41042451/?utm_source=SimplePie&utm_medium=rss&utm_campaign=pubmed-2&utm_content=1pq-4TZ0w1pIimA2HHLJZYTkSjaU335U-aRSFRxJ31Blahz9sN&fc=20220524061305&ff=20251003163345&v=2.18.0.post9+e462414]
- 82. Neural signatures of recollection are sensitive to memory ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11956928/]
- 83. The Role of Alpha-Band Brain Oscillations as a Sensory ... [https://www.frontiersin.org/journals/psychology/articles/10.3389/fpsyg.2011.00154/full]
- 84. Relationships Between Spontaneous Alpha Oscillation and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12134718/]
- 85. Decoding the details of visual working memory ... [https://www.sciencedirect.com/science/article/pii/S105381192500388X]
- 86. Layer-specific entrainment of gamma-band neural activity ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3528834/]
- 87. Spatial Working Memory in Humans Depends on Theta ... [https://www.sciencedirect.com/science/article/pii/S096098221630358X]
- 88. Mystery of gamma wave stimulation in brain disorders [https://molecularneurodegeneration.biomedcentral.com/articles/10.1186/s13024-024-00785-x]
- 89. A Survey on Event-Related Potential Visualization Practices [https://apertureneuro.org/article/116386-the-art-of-brainwaves-a-survey-on-event-related-potential-visualization-practices]
- 90. Deep learning with convolutional neural networks for EEG ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5655781/]
- 91. An Alpha and Theta Intensive and Short Neurofeedback ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4936375/]