Temporal Resolution in Neuroimaging: A Comparative Analysis of EEG, fNIRS, and fMRI for Research and Drug Development
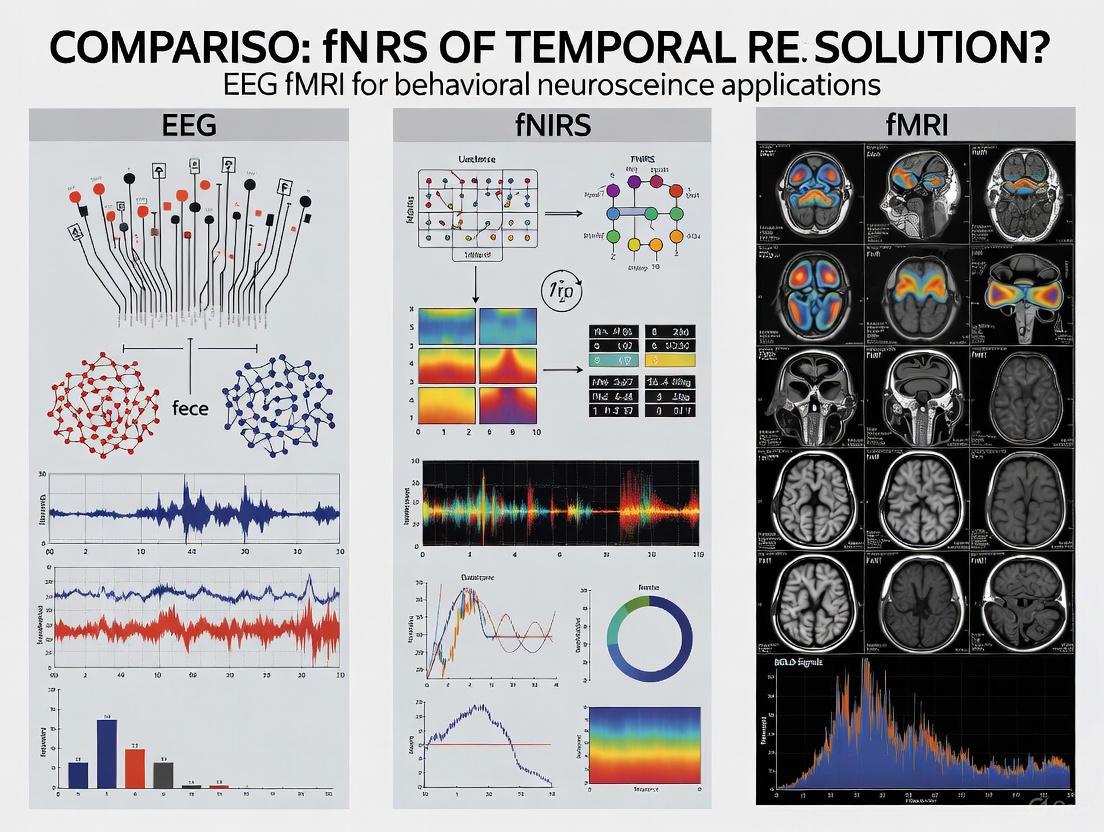
This article provides a comprehensive analysis of the temporal resolution of three cornerstone non-invasive neuroimaging techniques—EEG, fNIRS, and fMRI—tailored for researchers and drug development professionals.
Temporal Resolution in Neuroimaging: A Comparative Analysis of EEG, fNIRS, and fMRI for Research and Drug Development
Abstract
This article provides a comprehensive analysis of the temporal resolution of three cornerstone non-invasive neuroimaging techniques—EEG, fNIRS, and fMRI—tailored for researchers and drug development professionals. It explores the foundational principles defining their speed of measurement, from EEG's millisecond precision to the slower hemodynamic responses captured by fNIRS and fMRI. The review details methodological applications where temporal resolution is critical, such as in brain-computer interfaces and clinical monitoring, addresses key troubleshooting and optimization strategies for data quality, and validates findings through multimodal integration approaches. By synthesizing these aspects, the article serves as a strategic guide for selecting the appropriate imaging modality to accelerate neuroscience research and therapeutic development.
Understanding Temporal Resolution: The Fundamental Principles of EEG, fNIRS, and fMRI
In the quest to decipher the complex workings of the human brain, researchers rely on a suite of non-invasive neuroimaging technologies. Each technique offers a unique window into neural activity, with temporal resolution—the ability to precisely track changes in brain activity over time—serving as a critical differentiator. This parameter determines whether a technology can capture the brain's rapid, millisecond-scale electrical bursts or its slower, metabolic processes. For researchers and drug development professionals, selecting the appropriate tool is paramount, as it directly impacts the observable neural phenomena, from fast sensory processing to sustained cognitive states.
This guide provides a detailed, evidence-based comparison of the temporal resolution capabilities of three dominant neuroimaging modalities: electroencephalography (EEG), functional near-infrared spectroscopy (fNIRS), and functional magnetic resonance imaging (fMRI). By synthesizing current experimental data and methodologies, we aim to equip scientists with the knowledge needed to align their neuroimaging tools with specific research questions in cognitive neuroscience and clinical trials.
Temporal Resolution Defined: A Technical Comparison
Temporal resolution refers to the precision with which a measurement system can track changes in a signal over time. In neuroimaging, it defines the smallest distinguishable time interval between two distinct neural events.
The table below provides a quantitative comparison of the temporal and spatial characteristics of EEG, fNIRS, and fMRI.
Table 1: Technical Specifications of Major Neuroimaging Modalities
| Feature | EEG | fNIRS | fMRI |
|---|---|---|---|
| Temporal Resolution | Millisecond scale (ms) [1] | Seconds (1-10s) [1] [2] | Seconds (4-6s hemodynamic lag) [2] |
| Sampling Rate | Not specified in search results | Superior to fMRI [3] | 0.33 - 2 Hz (BOLD signal) [2] |
| Spatial Resolution | Low (centimeter-level) [1] | Moderate (better than EEG) [1] | High (millimeter-level) [2] |
| Primary Signal Source | Electrical potentials from cortical neurons [1] | Hemodynamic response (HbO/HbR) [1] | Hemodynamic response (BOLD) [2] |
| Key Strength | Capturing rapid neural dynamics [4] | Tolerates movement, portable [1] [3] | Whole-brain coverage, deep structures [2] |
Experimental Evidence: How Temporal Resolution Shapes Research
The theoretical differences in temporal resolution have profound implications for experimental design and data interpretation. The following protocols from peer-reviewed studies illustrate how these modalities are applied to capture distinct neural events.
Protocol 1: EEG for Event-Related Potentials (ERPs)
- Objective: To capture the brain's rapid electrical response to a specific sensory, cognitive, or motor event with millisecond precision [5].
- Task Example: Participants view images of animals or tools and perform silent naming or sensory-based imagery tasks. The neural response time-locked to the stimulus onset is analyzed [6].
- Methodology: EEG electrodes are placed on the scalp according to the international 10–20 system. During a task, hundreds of stimulus-locked trials are recorded and averaged to extract Event-Related Potentials (ERPs), which are voltage fluctuations characterized by specific components (e.g., N300, P300) occurring within hundreds of milliseconds post-stimulus [5].
- Data Interpretation: The high temporal resolution of EEG allows researchers to dissect the precise sequence of neural processes. For instance, studies have shown enhanced ERP amplitudes around 300 ms post-stimulus in parietal and occipital channels during intentional memory tasks, highlighting the rapid engagement of cognitive processes [5].
Protocol 2: fNIRS for Hemodynamic Monitoring During Sustained Tasks
- Objective: To measure changes in cortical blood oxygenation (hemodynamic response) associated with sustained neural activity during extended cognitive or motor tasks [1] [7].
- Task Example: The n-back task, a working memory paradigm where the load (value of 'n') is parametrically varied to manipulate mental workload [7].
- Methodology: fNIRS optodes are placed over the brain region of interest, such as the prefrontal cortex for working memory tasks. The system emits near-infrared light and detects its attenuation after passing through brain tissue, calculating changes in oxygenated (HbO) and deoxygenated hemoglobin (HbR) concentrations over time. Unlike EEG, the signal reflects a slow hemodynamic response that peaks 2-6 seconds after neural activity [1] [2].
- Data Interpretation: Grand block averaging of fNIRS signals reveals a characteristic increase in HbO with higher cognitive load (e.g., 2-back vs. 0-back tasks). This makes fNIRS ideal for studying sustained states like workload, attention, or emotional processing but unsuitable for pinpointing the exact onset of a brief neural event [7].
Protocol 3: fMRI for Spatially-Precise Hemodynamic Mapping
- Objective: To localize brain activity with high spatial resolution across the entire brain, including deep structures, by measuring the Blood-Oxygen-Level-Dependent (BOLD) signal [2] [3].
- Task Example: Block-design or event-related paradigms involving motor execution (e.g., wrist movement), sensory stimulation, or cognitive tasks [2] [3].
- Methodology: Participants perform tasks while inside an MRI scanner. The BOLD signal, an indirect measure of neural activity based on changes in blood oxygenation and flow, is recorded. The hemodynamic response function (HRF) in fMRI is slow, with a typical lag of 4-6 seconds behind the initiating neural event, fundamentally limiting its temporal resolution [2].
- Data Interpretation: The high spatial resolution allows for precise mapping of active regions. However, the slow BOLD response means that fMRI cannot resolve the fine-grained temporal sequence of brain activation, conflating rapid, successive neural events into a single hemodynamic peak [2].
Figure 1: Temporal Sequence of Neuroimaging Signals. This diagram illustrates the fundamental timing differences between direct electrical measurements (EEG) and indirect hemodynamic measurements (fNIRS, fMRI) following a neural event.
The Synergistic Approach: Multimodal Integration
Recognizing that no single modality is perfect, the field is increasingly moving toward multimodal integration, combining EEG and fNIRS to leverage their complementary strengths [1] [7] [8].
- Hybrid EEG-fNIRS for Brain-Computer Interfaces (BCIs): A study on motor imagery classification used an end-to-end deep learning model to fuse EEG and fNIRS signals. EEG provided fine temporal features of the motor intention, while fNIRS contributed complementary spatial information about the activated brain region. This fusion led to an average classification accuracy of 83.26%, a significant 3.78% improvement over using a single modality [9].
- Passive Monitoring of Mental Workload: In a study measuring mental workload during an n-back task, a hybrid EEG-fNIRS system achieved significantly higher accuracy in discriminating between different workload levels than either system alone. The combination provided a richer set of biomarkers, encompassing both the fast electrophysiological changes (EEG) and the slower hemodynamic correlates (fNIRS) of cognitive effort [7].
- Protocol for Simultaneous EEG-fNIRS Recording:
- Sensor Placement: Use an integrated cap with pre-defined openings for both EEG electrodes and fNIRS optodes, often based on the international 10–20 system [1].
- Hardware Synchronization: Employ external hardware triggers (e.g., TTL pulses) or shared acquisition software to synchronize the clocks of the EEG and fNIRS systems [1].
- Data Preprocessing: Process the two signals through separate, modality-specific pipelines to remove artifacts (e.g., motion, cardiac) before data fusion [1] [8].
- Data Fusion: Apply joint analysis techniques such as machine learning, joint Independent Component Analysis (jICA), or canonical correlation analysis (CCA) to integrate the temporal features from EEG with the spatial features from fNIRS [1] [9] [8].
Figure 2: Multimodal EEG-fNIRS Data Fusion Workflow. Combining the complementary strengths of EEG and fNIRS through data fusion techniques yields a more comprehensive picture of brain activity than either modality can provide alone.
Essential Research Reagent Solutions
The following table details key materials and software solutions essential for conducting neuroimaging studies, particularly those involving multimodal setups.
Table 2: Key Reagents and Solutions for Neuroimaging Research
| Item Name | Function/Application | Specification Notes |
|---|---|---|
| Integrated EEG-fNIRS Caps | Provides stable, co-registered placement of electrodes and optodes [1]. | Often based on the 10-20 system; should offer compatibility with various head sizes. |
| Conductive EEG Gel | Ensures low impedance between scalp and EEG electrodes for high-quality signal acquisition [1]. | |
| fNIRS Optode Holders | Precisely positions light sources and detectors on the scalp at fixed distances [1]. | Critical for maintaining consistent light penetration depth. |
| Synchronization Hardware | Generates TTL pulses to synchronize data acquisition across separate EEG and fNIRS systems [1]. | Vital for millisecond-precision multimodal analysis. |
| Motion Correction Algorithms | Software tools to minimize artifacts from participant movement, crucial for both EEG and fNIRS data [1]. | |
| Brain Mapping Software (e.g., AtlasViewer) | Determines anatomical locations of fNIRS channels since fNIRS lacks inherent anatomical information [3]. | Uses 3D digitalization and standard brain atlases. |
| Source-Decomposition Fusion Software | For advanced analysis of multimodal data using methods like joint ICA [1] [8]. | Enables unsupervised, symmetric fusion of EEG and fNIRS signals. |
Temporal resolution is a fundamental property that defines the scope and application of any neuroimaging technology. The choice between EEG, fNIRS, and fMRI is not a search for a superior tool, but for the right tool for the scientific question.
- EEG is the modality of choice when the research demands millisecond precision to dissect the rapid dynamics of brain function, such as in sensory processing or ERP studies [1] [5].
- fNIRS offers an optimal balance for investigating sustained cortical activity in real-world or mobile settings where fMRI is impractical and EEG's sensitivity to motion is problematic [1] [3] [7].
- fMRI remains the gold standard for whole-brain mapping with high spatial resolution, ideal for localizing function and investigating deep brain structures, provided the slow hemodynamic response is compatible with the experimental design [2] [3].
For the most comprehensive insights, particularly in applied fields like drug development where a complete picture of a treatment's effect on brain dynamics is crucial, a multimodal approach that combines the temporal prowess of EEG with the spatial and practical advantages of fNIRS is proving to be a powerfully synergistic path forward [1] [9] [7].
Electroencephalography (EEG) is a cornerstone non-invasive technique for measuring brain activity, renowned for its exceptional temporal resolution. This guide objectively compares its performance against functional near-infrared spectroscopy (fNIRS) and functional magnetic resonance imaging (fMRI), framing the comparison within the critical thesis of temporal resolution in neuroimaging.
Neuroimaging Modalities at a Glance
The selection of a neuroimaging technique is often a trade-off between temporal and spatial resolution. The table below provides a quantitative comparison of EEG, fNIRS, and fMRI.
Table 1: Key Performance Indicators of Non-Invasive Neuroimaging Techniques
| Feature | EEG (Electroencephalography) | fNIRS (Functional Near-Infrared Spectroscopy) | fMRI (Functional Magnetic Resonance Imaging) |
|---|---|---|---|
| What It Measures | Electrical activity from postsynaptic potentials of cortical neurons [10] [11] | Hemodynamic response (changes in oxygenated and deoxygenated hemoglobin) [12] [11] | Blood-oxygen-level-dependent (BOLD) signal, reflecting blood flow and oxygenation [13] |
| Temporal Resolution | High (milliseconds) [13] [11] | Low (seconds) [11] | Low (seconds) [13] |
| Spatial Resolution | Low (centimeter-level) [13] [11] | Moderate (better than EEG, cortical surface) [11] | High (millimeters) [13] |
| Invasiveness | Non-invasive [10] | Non-invasive [12] | Non-invasive [13] |
| Key Strength | Direct measurement of neural dynamics with millisecond precision [11] | Good balance of portability and spatial resolution for cortical areas [12] [11] | Precise spatial localization of brain activity [13] |
| Primary Limitation | Poor spatial resolution; sensitive to movement and environmental artifacts [13] [11] | Limited depth of measurement; indirect, slow hemodynamic response [11] | Very expensive; restrictive environment; low temporal resolution [13] |
Experimental Protocols: Unlocking Brain Dynamics with EEG
The following section details standard methodologies for EEG experimentation, from data collection to analysis, which are critical for generating the robust data used in comparative studies.
Experimental Setup and Data Collection
A meticulously prepared experiment is the foundation of clean EEG data.
- Pilot Testing: Before a full study, pilot sessions are essential to verify that stimuli are presented correctly, equipment functions properly, and participants understand the task [14].
- Signal Quality Control: Researchers aim for low electrode impedance (resistance), which indicates a strong connection and high signal quality. As emphasized by Prof. Steve Luck, "There is no substitute for clean data" – no algorithm can fully salvage poorly recorded signals [14].
- Stimulus Presentation: The choice of visual stimuli, such as text and background colors, can be optimized for participant comfort and to minimize visual strain, especially in dimly lit labs. Grey backgrounds with black text are often recommended over high-contrast combinations (e.g., white on black) for prolonged viewing [15].
Data Pre-processing and Artifact Handling
Raw EEG data contains both relevant neural signals and irrelevant artifacts (e.g., from eye blinks, muscle movement, or heartbeats). Pre-processing is crucial to attenuate these contaminants [14] [16].
- Artifact Rejection vs. Attenuation: Researchers must choose between rejecting contaminated data segments entirely or correcting them using statistical procedures.
- Multimodal Assistance: Combining EEG with other sensors like eye trackers or electrocardiograms (ECG) makes it easier to identify and remove physiological artifacts [14].
The following diagram illustrates a generalized workflow for EEG signal processing, from acquisition to analysis, highlighting the parallel paths for different types of information that can be extracted.
EEG Data Processing Pathway
Data Analysis and Feature Extraction
EEG data can be analyzed to extract various features depending on the research question. The choice of analysis method is an informed decision that should be based on established literature and the specific goals of the study [14].
- Power Spectrum Analysis: This method analyzes energy changes in different frequency bands (e.g., Alpha, Beta, Theta). Common computational methods include:
- Fast Fourier Transform (FFT): A fast algorithm for converting a signal from the time domain to the frequency domain. It has high frequency resolution but can be susceptible to noise [10] [16].
- Welch's Method: An improvement over FFT that involves splitting the signal into overlapping segments, windowing them, and averaging the periodograms. This reduces the variance of the power spectral density estimate [16].
- Time-Frequency Analysis: For non-stationary signals where frequency content changes over time, methods like Wavelet Transform (WT) are more suitable. WT uses variable-sized windows, providing a flexible time-frequency representation [16].
- Event-Related Potentials (ERPs): When analyzing brain responses time-locked to a specific stimulus, researchers examine the latencies and amplitudes of characteristic peaks in the averaged EEG waveform [14] [10].
The Scientist's Toolkit: Essential Reagents and Materials
This table details key materials and software solutions used in typical EEG research protocols.
Table 2: Key Research Reagent Solutions for EEG Experiments
| Item | Function / Description |
|---|---|
| EEG Cap with Electrodes | A head cap embedded with electrodes according to the international 10-20 system for standardized scalp positioning. |
| Electrolyte Gel | Applied to reduce impedance between the scalp and electrodes, ensuring high-quality signal acquisition [14]. |
| Amplifier & Data Acquisition System | Hardware that amplifies the microvolt-level brain signals and converts them from analog to digital for analysis. |
| Stimulus Presentation Software | Software (e.g., Presentation, PsychoPy) used to display visual or auditory stimuli with precise timing. |
| Artifact Processing Tools | Algorithms (e.g., ICA, regression) implemented in software to identify and remove ocular, muscular, and cardiac artifacts [14]. |
| Statistical Analysis Package | Software (e.g., SPSS, R, Python with SciPy) for performing t-tests, ANOVAs, and other statistical tests on EEG metrics [14]. |
The Complementary Nature of Modalities: fNIRS-EEG Fusion
Recognizing that no single modality is perfect, a powerful trend in neuroscience is the development of dual-modality systems. The integration of EEG and fNIRS is particularly promising because their strengths are complementary [12]. EEG provides the high-temporal-resolution electrical data, while fNIRS provides better-spatially-localized hemodynamic data.
Such a system is ideal for probing the relationship between electrical brain activity and the subsequent hemodynamic response (neurovascular coupling). Integrated helmets are designed to house both EEG electrodes and fNIRS optodes, and data fusion techniques like joint Independent Component Analysis (jICA) are used to analyze the combined datasets [12] [11]. This approach is being applied in clinical domains such as epilepsy, attention-deficit hyperactivity disorder (ADHD), and monitoring the depth of anesthesia [12].
In the pursuit of understanding brain function, neuroscientists employ various neuroimaging technologies, each with distinct strengths and limitations. Temporal resolution—the ability to measure changes in brain activity over time—represents a fundamental parameter distinguishing these techniques. Functional Near-Infrared Spectroscopy (fNIRS) occupies a unique position in the temporal landscape of neuroimaging, operating on a seconds-scale timeframe that bridges the gap between the millisecond resolution of electroencephalography (EEG) and the minute-scale resolution of functional magnetic resonance imaging (fMRI). This characteristic directly stems from fNIRS's measurement target: the hemodynamic response associated with neural activity through neurovascular coupling. When brain regions become active, a cascade of vascular changes unfolds, characterized by increased blood flow, blood volume, and oxygen metabolism. fNIRS detects these changes by measuring concentration variations in oxygenated hemoglobin (HbO) and deoxygenated hemoglobin (HbR) using near-infrared light, capturing a physiological process that evolves over several seconds [17]. This review situates fNIRS within the broader neuroimaging toolkit, providing a detailed comparison of its temporal characteristics alongside EEG and fMRI to guide researchers in selecting appropriate methodologies for investigating brain function.
Quantitative Comparison of Temporal Resolution Across Techniques
The temporal capabilities of EEG, fNIRS, and fMRI differ substantially, reflecting their measurement of distinct neural correlates. The following table summarizes key comparative metrics:
Table 1: Temporal Resolution and Characteristics Comparison of EEG, fNIRS, and fMRI
| Feature | EEG | fNIRS | fMRI |
|---|---|---|---|
| Temporal Resolution | Milliseconds (∼1-100 ms) [12] [18] | Seconds (∼1-10 s) [18] [17] [2] | Seconds (∼1-6 s) [2] |
| Measured Signal | Electrical potentials from synaptic activity [18] | Hemodynamic changes (HbO/HbR) [17] | Blood-oxygen-level-dependent (BOLD) signal [2] |
| Hemodynamic Delay | Not applicable | 2-3 second onset, 5-10 second peak [17] | 4-6 second lag [2] |
| Primary Strength | Capturing rapid neural dynamics | Localizing sustained cortical activity | Whole-brain spatial mapping |
| Ideal Use Cases | Event-related potentials, brain-state monitoring [18] | Naturalistic studies, rehabilitation, developmental science [18] | Deep brain structure mapping, detailed spatial localization [2] |
These temporal differences directly translate to varied experimental applications. EEG's millisecond precision makes it ideal for studying the timing of cognitive processes, such as sensory perception and rapid decision-making [18]. In contrast, fNIRS's seconds-scale resolution is well-suited for investigating sustained cognitive states, workload, and affective processing localized in the cortex [18]. fMRI provides comprehensive spatial mapping but its slower temporal resolution and confinement to restrictive scanning environments limit its utility for studying naturalistic behaviors or rapid neural changes [2].
The Physiological Basis of fNIRS's Temporal Profile
The seconds-scale temporal resolution of fNIRS is not a technical limitation but a reflection of its underlying physiological measurement target. fNIRS detects neural activity indirectly via neurovascular coupling, the process where neural firing triggers changes in local blood flow and oxygenation. This hemodynamic response unfolds with a characteristic delay and shape. Following neural activation, the hemodynamic response typically begins after 2-3 seconds, peaks around 5-10 seconds, and then returns to baseline, often followed by a slight undershoot [17]. This slow time course is intrinsic to the biological process of vascular regulation and is therefore a fundamental property shared by all hemodynamic-based neuroimaging methods, including fMRI.
The fNIRS signal comprises changes in both oxygenated hemoglobin (HbO) and deoxygenated hemoglobin (HbR). During cortical activation, local HbO concentration typically increases due to elevated blood flow exceeding oxygen consumption, while HbR concentration shows a slight decrease as oxygen is extracted from the blood [19] [17]. This relationship is central to interpreting fNIRS data, with the HbO signal generally considered more sensitive and robust for detecting brain activity.
Figure 1: The fNIRS Signaling Pathway. This diagram illustrates the temporal sequence from neural activity to the measurable fNIRS signal, highlighting the neurovascular coupling process that introduces a characteristic delay.
Experimental Evidence: Protocol and Data
Semantic Decoding with Simultaneous EEG-fNIRS
A 2025 study investigating semantic neural decoding provides a clear example of fNIRS implementation in cognitive neuroscience. The research aimed to differentiate between imagined categories (animals vs. tools) using silent naming and sensory-based imagery tasks [6].
- Experimental Protocol: Participants were shown images representing animals or tools and performed four mental tasks: silent naming, visual imagery, auditory imagery, and tactile imagery. Each trial involved a cue presentation followed by a 3-second mental task period. Simultaneous EEG and fNIRS signals were recorded from 12 participants, allowing direct comparison of electrical and hemodynamic responses [6].
- Temporal Considerations: The 3-second task duration was strategically chosen to accommodate the slow hemodynamic response measured by fNIRS. This design contrasts with typical EEG paradigms, which can utilize much shorter trial durations due to the modality's millisecond-scale resolution.
- Data Interpretation: The fNIRS data captured the build-up and peak of the hemodynamic response during the sustained cognitive effort of mental imagery. This study exemplifies how experimental designs must be tailored to the temporal characteristics of fNIRS, with sufficiently long task periods and inter-stimulus intervals to allow the hemodynamic response to evolve and return to baseline [6] [17].
Motor Execution, Observation, and Imagery Study
A 2023 multimodal investigation further highlights fNIRS's application in mapping cortical activity during different motor states, demonstrating its utility in ecological paradigms [20].
- Experimental Protocol: Researchers collected simultaneous fNIRS-EEG data from 21 participants during motor execution, observation, and imagery conditions using a live-action paradigm with a cup-moving task. A 24-channel fNIRS system measured HbO and HbR changes over sensorimotor and parietal cortices at a 10 Hz sampling rate, while EEG recorded electrical activity [20].
- Methodology and Analysis: The fNIRS data revealed activated regions in the left angular gyrus, right supramarginal gyrus, and right superior/inferior parietal lobes. The differential activation patterns between conditions emerged gradually over seconds, consistent with the hemodynamic response timecourse. The study employed structured sparse multiset Canonical Correlation Analysis (ssmCCA) to fuse fNIRS and EEG data, leveraging the complementary temporal and spatial information from both modalities [20].
- Significance: This research demonstrates fNIRS's particular strength in capturing the sustained, localized cortical activation patterns during ecologically valid tasks that involve natural movement and social interaction—contexts where fMRI is impractical due to motion constraints [20].
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 2: Essential Materials for fNIRS Research
| Item | Function/Purpose | Example/Notes |
|---|---|---|
| fNIRS System | Measures HbO/HbR concentration changes using near-infrared light. | Continuous-wave systems (e.g., NIRScout, Hitachi ETG-4100) are common [21] [20]. |
| Optodes | Emit (sources) and detect (detectors) near-infrared light through tissue. | Typically 16-32 sources/detectors forming multiple channels [21] [20]. |
| Probe Cap/Helmet | Holds optodes in standardized positions on the scalp. | Often uses International 10-20 system for placement; can be integrated with EEG electrodes [12] [20]. |
| Digitization System | Records precise 3D locations of optodes on the head. | Magnetic digitizers (e.g., Fastrak) co-register measurement positions with brain anatomy [21] [20]. |
| Synchronization Hardware | Aligns fNIRS data with other modalities (EEG, fMRI) and task stimuli. | TTL pulses, parallel ports, or shared clock systems ensure temporal alignment [12]. |
| Processing Software | Converts raw light intensity to HbO/HbR and removes artifacts. | Homer2, AtlasViewer, in-house MATLAB scripts [21]. |
Integrated Analysis: Placing fNIRS in the Broader Temporal Hierarchy
When evaluating neuroimaging techniques, temporal resolution exists in a fundamental trade-off with spatial resolution. The following diagram illustrates this relationship and situates fNIRS within the broader hierarchy of common neuroimaging methods:
Figure 2: Neuroimaging Modalities ranked by Temporal Resolution. This hierarchy shows fNIRS positioned between the high temporal resolution of electrical recording techniques (MEA, EEG) and the lower temporal resolution of metabolic imaging methods (fMRI, PET). Adapted from [12].
This hierarchy reveals fNIRS's strategic position. While it cannot capture the millisecond-scale electrical events detectable by EEG, it provides significantly better temporal resolution than fMRI for tracking the evolving hemodynamic response [12] [2]. This advantage, combined with its portability and motion tolerance, makes fNIRS particularly valuable for studying brain function in naturalistic settings, with populations prone to movement (e.g., infants, clinical patients), and during rehabilitation protocols where ecological validity is paramount [19] [4] [18].
The combination of fNIRS with EEG in multimodal approaches represents a powerful trend in neuroscience, as it simultaneously captures both the fast electrical dynamics (via EEG) and the localized hemodynamic changes (via fNIRS) associated with neural processing [12] [20]. This integration allows researchers to overcome the limitations of either technique used in isolation, providing a more comprehensive picture of brain function across multiple temporal and spatial scales.
Functional Near-Infrared Spectroscopy (fNIRS) provides a unique window into brain function by tracking hemodynamic responses on a seconds scale. This temporal profile, while slower than electrical recording techniques like EEG, is ideally suited for investigating sustained cognitive processes, localized cortical activation, and brain function in real-world contexts. The experimental evidence demonstrates that fNIRS can effectively discriminate between different cognitive states—from semantic categorization to motor imagery—when appropriate experimental designs accommodate the characteristic hemodynamic delay. When selecting neuroimaging methods for research or clinical applications, understanding the temporal attributes of fNIRS and how they complement other modalities enables more informed choices. For studies requiring both temporal dynamics and spatial localization, combined fNIRS-EEG approaches offer a particularly promising path forward in unraveling the complexities of human brain function.
Functional Magnetic Resonance Imaging (fMRI) has become a cornerstone technique for non-invasive mapping of human brain function. Unlike methods that directly measure neural electrical activity, fMRI detects changes in blood oxygenation, making it an indirect marker of brain activity through the mechanism known as neurovascular coupling. The dominant contrast mechanism in fMRI is the Blood-Oxygen-Level-Dependent (BOLD) signal, which originates from changes in the paramagnetic properties of deoxygenated hemoglobin in the blood surrounding active neurons [22] [23].
Understanding the temporal characteristics of the BOLD signal is crucial for interpreting fMRI data, especially when compared to other major neuroimaging modalities. While fMRI provides excellent spatial resolution, its temporal resolution is fundamentally limited by the sluggish nature of the hemodynamic response, which typically lags behind the initiating neural activity by 4–6 seconds [2]. This article provides a detailed comparison of fMRI's temporal resolution against electroencephalography (EEG) and functional near-infrared spectroscopy (fNIRS), situating the BOLD signal within the broader context of temporal resolution in brain imaging research.
Quantitative Comparison of Neuroimaging Modalities
The following tables summarize the key technical and performance characteristics of the three major non-invasive neuroimaging modalities, with a specific focus on their temporal attributes.
Table 1: Technical Specifications and Performance Metrics of fMRI, EEG, and fNIRS
| Feature | fMRI (BOLD) | EEG | fNIRS |
|---|---|---|---|
| What is Measured | Changes in blood oxygenation (BOLD signal) [22] | Electrical potentials from synchronized neuronal firing [13] | Changes in oxy/deoxy-hemoglobin concentrations using near-infrared light [2] [13] |
| Temporal Resolution | Low (0.3-2 Hz sampling rate); hemodynamic lag of 4-6 s [2] | Very High (milliseconds) [13] [24] | Moderate (milliseconds to seconds) [2] [19] |
| Spatial Resolution | High (millimeters) [2] [13] | Low (centimeters) [13] | Moderate (1-3 centimeters) [2] |
| Portability & Cost | Low portability, very high cost [2] [13] | High portability, low-moderate cost [24] | High portability, moderate cost [2] [24] |
| Key Temporal Limitation | Slow hemodynamic response limits study of rapid neural dynamics [2] [22] | Poor at localizing deep sources; signal is a blurred mixture [13] [23] | Faster than fMRI but slower than EEG; still an indirect metabolic measure [2] [19] |
Table 2: Practical Considerations for Research and Clinical Applications
| Consideration | fMRI (BOLD) | EEG | fNIRS |
|---|---|---|---|
| Best Suited For | Precise spatial localization of activity; deep brain structures [2] [23] | Tracking very fast neural oscillations and event-related potentials [13] [24] | Naturalistic studies, bedside monitoring, populations prone to movement (infants, patients) [2] [19] |
| Experimental Environment | Restrictive scanner environment; sensitive to motion [2] | Flexible; can be used in more naturalistic settings [24] | Highly flexible; ideal for real-world and clinical settings [2] [24] |
| Main Artifact Sources | Motion, cardiac/pulse, respiratory noise [25] | Muscle, eye movement, environmental electrical noise [13] | Scalp blood flow, hair color, movement [2] [13] |
The Physiological Basis of the BOLD Signal and Its Temporal Dynamics
The Neurovascular Coupling Pathway
The BOLD signal is an indirect and complex reflection of neuronal activity. During neural activation, there is a local increase in energy consumption. This triggers a compensatory increase in cerebral blood flow (CBF) that overcompensates for the oxygen demand, leading to a net decrease in the concentration of deoxygenated hemoglobin in the local vasculature [22] [23]. Because deoxygenated hemoglobin is paramagnetic, this decrease alters the local magnetic field homogeneity, which is detected as the BOLD signal by the MRI scanner [23] [26].
The temporal dynamics of this response are not instantaneous. The canonical hemodynamic response function (HRF) is characterized by a 2-second onset delay, a peak that occurs 6-12 seconds after stimulus onset, and a slow return to baseline that often includes a prolonged "post-stimulus undershoot" [23] [26]. This slow timescale fundamentally limits the ability of standard BOLD fMRI to track rapid changes in neural information processing.
Diagram 1: The BOLD Signal Pathway.
Neural Correlates of the BOLD Signal
A foundational question is what specific aspect of neural activity the BOLD signal reflects. Converging evidence from simultaneous fMRI and electrophysiological recordings, primarily in sensory cortices, indicates that the BOLD signal correlates more strongly with the local field potential (LFP) than with the spiking activity (action potentials) of individual neurons [22] [23].
The LFP largely reflects integrative post-synaptic activity—the summed excitatory and inhibitory inputs to a neural population—as well as dendritic processing and intrinsic membrane oscillations [22]. This is distinct from the output spiking activity of neurons. This distinction is critical because it means the BOLD signal is a better proxy for the input and local processing within a brain area than for its final output firing. In some brain regions and under certain conditions, this coupling can dissociate, highlighting that the BOLD-neural relationship is not always one-to-one [22].
Experimental Protocols for Characterizing and Comparing Temporal Resolution
Synchronous Multimodal fMRI-fNIRS Validation
Objective: To validate fNIRS signals against the fMRI BOLD response and leverage their complementary temporal and spatial strengths [2].
Protocol Summary:
- Setup: A custom cap integrating fNIRS optodes is used inside the MRI scanner. The system must be MRI-compatible to avoid electromagnetic interference [2] [24].
- Stimulus Presentation: Participants perform motor, cognitive, or sensory tasks in a block-based or event-related design while data is acquired simultaneously from both modalities [2].
- Data Acquisition: fMRI acquires whole-brain BOLD data with high spatial resolution. fNIRS acquires time-series data of HbO and HbR concentrations from the cortical surface with high temporal sampling [2].
- Analysis: The fNIRS-derived hemoglobin signals are convolved with a hemodynamic response function and coregistered with the fMRI BOLD data. Temporal correlation and spatial overlap of activation maps are quantified to validate fNIRS and create a spatiotemporally enriched dataset [2].
Motor Imagery Neurofeedback with EEG-fNIRS
Objective: To investigate the benefits of combining the high temporal resolution of EEG with the hemodynamic measures of fNIRS for brain-computer interfaces and rehabilitation, contrasting with the impracticality of real-time fMRI for such ecological applications [24].
Protocol Summary:
- Setup: Participants wear a custom cap that integrates both EEG electrodes and fNIRS optodes over the sensorimotor cortices [24].
- Task: Participants perform left-hand motor imagery (mental rehearsal of movement without execution). A visual neurofeedback signal (e.g., a moving ball) is provided in real-time [24].
- Real-Time Signal Processing: The system calculates a neurofeedback score from three distinct conditions:
- EEG-only: Based on event-related desynchronization in the mu/beta rhythm over the motor cortex.
- fNIRS-only: Based on the increase of HbO in the contralateral motor cortex.
- EEG-fNIRS combined: A multimodal score integrating features from both signals to provide more specific feedback [24].
- Comparison: The efficacy, participant control, and specificity of brain activation are compared across the three neurofeedback conditions [24].
The Scientist's Toolkit: Key Research Reagents and Materials
Table 3: Essential Materials and Equipment for Multimodal Neuroimaging Research
| Item | Function in Research |
|---|---|
| High-Density EEG System (e.g., 32+ channels) | Records electrical brain activity with millisecond temporal resolution. Essential for capturing event-related potentials and neural oscillations [24]. |
| fNIRS System (Continuous Wave or Time-Domain) | Measures cortical hemodynamic activity with greater portability and motion tolerance than fMRI. Ideal for naturalistic settings and infant studies [2] [19]. |
| MRI-Compatible fNIRS Cap | Enables simultaneous acquisition of fMRI and fNIRS data without causing interference or safety issues inside the MRI scanner [2] [24]. |
| Custom Neurofeedback Software Platform | Integrates real-time signal processing from one or more modalities, computes a feedback metric, and presents it to the participant in a closed-loop paradigm [24]. |
| Biophysical Signal Models (e.g., Hemodynamic Response Function) | Used to model and predict the slow BOLD response and to disentangle neural signals from hemodynamic confounds in multimodal data fusion [2] [26]. |
The BOLD signal measured by fMRI provides an invaluable, albeit slow, window into brain function, offering unmatched spatial localization for a non-invasive technique. Its temporal resolution is fundamentally constrained by the hemodynamic response, limiting its ability to track the brain's rapid dynamics. As the field of cognitive neuroscience progresses toward studying the brain in more naturalistic and interactive contexts, the strategic combination of fMRI with electrophysiological techniques like EEG and fNIRS is becoming increasingly important. These multimodal approaches synergistically combine fMRI's spatial precision with the superior temporal resolution of other methods, paving the way for a more comprehensive and temporally accurate understanding of human brain function in health and disease.
Understanding the brain's dynamic functions requires neuroimaging techniques that can capture its rapid and complex activities. Temporal resolution—the ability to measure changes in brain activity over time—serves as a fundamental differentiator among major non-invasive neuroimaging technologies. Within cognitive neuroscience and clinical research, electroencephalography (EEG), functional near-infrared spectroscopy (fNIRS), and functional magnetic resonance imaging (fMRI) represent three widely used modalities that operate on dramatically different temporal scales. This hierarchy directly influences their applicability for studying various neural processes, from millisecond-scale electrical firing to second-level hemodynamic responses.
Each technique captures distinct physiological phenomena: EEG directly measures electrical activity from synchronized neuronal firing, fNIRS monitors hemodynamic changes through near-infrared light, and fMRI detects blood oxygenation-level dependent (BOLD) signals via magnetic properties. These fundamental measurement principles dictate not only their temporal characteristics but also their spatial resolution, portability, and suitability for different experimental environments. This guide provides a direct comparative analysis of these modalities, with a focused examination of their temporal capabilities, supported by experimental data and methodological protocols to inform researcher selection and application.
Fundamental Principles and Measurement Bases
The temporal characteristics of EEG, fNIRS, and fMRI are intrinsically linked to their underlying biophysical measurement principles. EEG records postsynaptic electrical potentials generated by cortical pyramidal neurons directly at the scalp surface, enabling direct monitoring of neural activity with minimal physiological latency [27]. This direct measurement approach allows EEG to capture neural dynamics on a millisecond scale, matching the speed at which neural information processing occurs [27].
In contrast, both fNIRS and fMRI measure hemodynamic responses—metabolic consequences of neural activity rather than the activity itself. fNIRS employs near-infrared light (650-950 nm) to measure concentration changes in oxygenated (HbO) and deoxygenated hemoglobin (HbR) in cortical blood flow [27] [2]. fMRI detects these same hemodynamic changes indirectly through the Blood Oxygen Level Dependent (BOLD) signal, which exploits the different magnetic properties of oxygenated versus deoxygenated hemoglobin [3]. This shared dependence on neurovascular coupling introduces an inherent physiological delay—the hemodynamic response typically lags behind neural activity by 2-6 seconds [27] [2].
The fundamental difference in measurement targets creates a temporal hierarchy: EEG captures neural activity directly and almost instantaneously, while fNIRS and fMRI track the slower, metabolic consequences of that activity. This distinction is crucial for experimental design and data interpretation across neuroscience research and clinical applications.
Quantitative Comparison of Temporal and Spatial Characteristics
The temporal and spatial capabilities of EEG, fNIRS, and fMRI demonstrate a well-characterized inverse relationship, where techniques with superior temporal resolution typically exhibit limited spatial resolution, and vice versa. [27]
Table 1: Comprehensive Comparison of EEG, fNIRS, and fMRI Technical Specifications
| Feature | EEG | fNIRS | fMRI |
|---|---|---|---|
| Temporal Resolution | High (millisecond scale) [27] | Low (seconds) [27] | Low (seconds); BOLD signal sampling typically 0.33-2 Hz [2] |
| Spatial Resolution | Low (centimeter-level) [27] | Moderate (better than EEG); ~1-3 cm [2] | High (millimeter-level) [2] [3] |
| Depth of Measurement | Cortical surface [27] | Outer cortex (~1-2.5 cm deep) [27] [2] | Whole brain (cortical and subcortical) [2] |
| Measurement Basis | Electrical activity of neurons (postsynaptic potentials) [27] | Hemodynamic response (blood oxygenation levels) [27] | Hemodynamic response (BOLD signal) [2] [3] |
| Physiological Lag | Minimal (direct neural measurement) | 2-6 seconds (hemodynamic response) [27] | 4-6 seconds (hemodynamic response) [2] |
| Portability | High (lightweight, wireless systems available) [27] | High (portable, wearable formats) [27] [3] | Low (requires MRI scanner environment) [3] |
| Motion Tolerance | Low (susceptible to movement artifacts) [27] | High (relatively robust to movement) [27] [3] | Low (highly sensitive to motion artifacts) [2] [3] |
The temporal hierarchy is unequivocal: EEG provides the highest temporal resolution (milliseconds), enabling tracking of rapid neural dynamics such as those occurring during sensory processing or transient cognitive events. fNIRS occupies an intermediate position, with temporal resolution on the scale of seconds, limited by the hemodynamic response latency. fMRI shares similar temporal constraints due to its identical dependence on neurovascular coupling, though its sampling rate is often technically limited by scanner capabilities [2].
Spatially, this relationship reverses: fMRI provides the highest spatial resolution (millimeter-level) and whole-brain coverage, including subcortical structures. fNIRS offers moderate spatial resolution but is restricted to superficial cortical regions due to limited light penetration depth. EEG provides the most limited spatial resolution due to signal dispersion through the skull and scalp [27] [2] [3].
Table 2: Optimal Application Domains Based on Resolution Characteristics
| Research Application | Recommended Modality | Rationale |
|---|---|---|
| Event-Related Potentials (ERPs) | EEG [27] [5] | Millisecond resolution captures rapid neural dynamics |
| Brain-Computer Interfaces (BCIs) | EEG or EEG-fNIRS hybrid [27] [6] [9] | Fast responses (EEG) or improved classification (hybrid) |
| Sensory/Motor Timing Studies | EEG [27] | Precise timing of stimulus processing and response |
| Sustained Cognitive Tasks | fNIRS [27] [28] | Tracks hemodynamic changes during extended tasks |
| Naturalistic Environment Studies | fNIRS [27] [3] | Motion tolerance and portability for real-world settings |
| Deep Brain Structure Mapping | fMRI [2] [3] | Whole-brain coverage including subcortical regions |
| Anatomical-Functional Correlation | fMRI or multimodal [2] [29] | High spatial precision for localization |
Experimental Protocols and Methodological Approaches
Simultaneous EEG-fNIRS Protocol for Semantic Decoding
A recent study investigating semantic neural decoding of imagined animals and tools employed simultaneous EEG-fNIRS recordings to leverage their complementary temporal and spatial strengths [6]. The experimental protocol involved:
- Participants: 12 right-handed native English speakers (20-57 years) performed silent naming and sensory-based imagery tasks [6].
- Stimuli: 18 animals and 18 tools presented as gray-scale images against white background [6].
- Task Design: Four distinct mental tasks:
- Silent naming: Participants silently named displayed objects
- Visual imagery: Participants visualized the object in their minds
- Auditory imagery: Participants imagined sounds associated with the object
- Tactile imagery: Participants imagined the feeling of touching the object [6]
- Temporal Parameters: Mental tasks lasted 3 seconds each, with randomized block ordering [6].
- Data Acquisition: EEG recorded electrical activity at the scalp surface while fNIRS monitored hemodynamic responses in cortical regions, with synchronization between modalities [6].
This protocol capitalized on EEG's high temporal resolution to capture rapid neural dynamics during imagery onset while utilizing fNIRS to localize sustained cortical activation patterns throughout the several-second task duration.
Integrated fMRI-fNIRS Methodologies
Multimodal integration of fMRI and fNIRS has been systematically reviewed across 63 studies, revealing two primary integration modes [2]:
- Synchronous Acquisition: Simultaneous data collection capitalizes on fMRI's high spatial resolution and fNIRS's superior temporal resolution and portability [2].
- Asynchronous Acquisition: Sequential data collection uses each modality under its optimal conditions, with subsequent temporal alignment [2].
Key methodological considerations include addressing hardware incompatibilities (electromagnetic interference in MRI environments), motion constraints, and developing sophisticated data fusion pipelines often enhanced by machine learning approaches [2].
Multimodal Experimental Workflow
Advanced Data Fusion and Analytical Techniques
Joint EEG-fNIRS Source Reconstruction
Advanced computational approaches have been developed to overcome the inherent limitations of individual modalities. One promising algorithm utilizes fNIRS (more specifically, diffuse optical tomography or DOT) reconstruction as spatial priors for EEG source localization [30]. This method demonstrates that:
- Neuronal sources separated by only 2.3-3.3 cm and 50 ms can be accurately recovered using joint EEG-DOT, but not by either modality in isolation [30].
- The spatial precision of DOT reconstruction (millimeter-scale) significantly enhances EEG's centimeter-scale spatial resolution when properly integrated [30].
- Performance can be further optimized through strategic placement of EEG electrodes and DOT optodes based on target brain regions [30].
This fusion approach effectively marries the millisecond temporal resolution of EEG with the millimeter spatial resolution achievable with high-density DOT, creating a synergistic analytical framework that transcends individual modality limitations.
Deep Learning Integration for Brain-Computer Interfaces
Motor imagery classification for BCIs has demonstrated significant improvements through EEG-fNIRS fusion using deep learning architectures. Recent implementations include:
- EEG Feature Extraction: Dual-scale temporal convolution and depthwise separable convolution capture spatiotemporal patterns, enhanced with hybrid attention modules to emphasize salient neural features [9].
- fNIRS Feature Extraction: Spatial convolution across channels explores regional activation differences, while parallel temporal convolution with gated recurrent units (GRUs) captures hemodynamic temporal dynamics [9].
- Decision Fusion: Dirichlet distribution parameter estimation models uncertainty, with Dempster-Shafer Theory implementing a two-layer reasoning process to fuse evidence from both modalities [9].
This approach achieved an average classification accuracy of 83.26% for motor imagery tasks—a 3.78% improvement over state-of-the-art unimodal methods [9], demonstrating the practical advantage of leveraging complementary temporal and hemodynamic information.
Temporal Hierarchy of Neural Signals
The Scientist's Toolkit: Essential Research Solutions
Table 3: Essential Research Materials and Analytical Tools
| Tool/Solution | Function/Purpose | Application Context |
|---|---|---|
| High-Density EEG Systems | Record electrical activity with millisecond temporal resolution [27] | ERP studies, rapid cognitive processing, seizure monitoring |
| Portable fNIRS Systems | Monitor hemodynamic responses in naturalistic settings [27] [3] | Ecological studies, child development, rehabilitation monitoring |
| Integrated EEG-fNIRS Caps | Enable simultaneous data collection with coordinated placement [27] | Multimodal studies requiring both temporal and spatial information |
| fMRI-Compatible fNIRS Probes | Allow simultaneous fMRI-fNIRS data acquisition [2] | Validation studies, enhanced spatial mapping |
| 3D Digitization Systems | Precisely localize EEG electrode/fNIRS optode positions [3] | Accurate spatial registration and source localization |
| Motion Correction Algorithms | Minimize movement artifacts during data collection [27] | Studies with children, clinical populations, or natural movement |
| Data Synchronization Interfaces | Temporally align data streams from multiple modalities [27] | All multimodal study designs |
| Advanced Fusion Algorithms | Integrate heterogeneous neural data (e.g., jICA, CCA) [27] | Multimodal feature extraction and pattern recognition |
| Deep Learning Frameworks | Implement complex fusion architectures for classification [9] | BCI applications, cognitive state decoding |
| Source Reconstruction Tools | Localize neural activity from scalp recordings [30] | Precise spatial mapping from EEG/fNIRS data |
The temporal hierarchy among EEG, fNIRS, and fMRI is unequivocal: EEG operates at the millisecond scale essential for capturing direct neural dynamics, while both fNIRS and fMRI track the slower hemodynamic consequences of neural activity, with fNIRS offering practical advantages for naturalistic settings and fMRI providing superior spatial resolution for deep brain structures. This hierarchy fundamentally guides modality selection based on specific research questions—whether the priority lies in capturing rapid neural transitions or precisely localizing sustained brain activity.
Future advancements are increasingly focused on multimodal integration, leveraging complementary strengths to overcome individual limitations. Emerging methodologies in hardware integration, synchronization protocols, and machine learning-based data fusion are creating new possibilities for comprehensive brain mapping [27] [2] [9]. Particularly promising are portable integrated systems that combine EEG and fNIRS, enabling researchers to capture both millisecond electrical dynamics and localized hemodynamic responses in real-world environments [30].
These technological innovations continue to blur the traditional boundaries between modalities, offering neuroscientists and clinical researchers an expanding toolkit for decoding brain function across multiple spatiotemporal scales. As these integrated approaches mature, they promise to transform our understanding of brain dynamics in health and disease, enabling increasingly sophisticated investigation of the human brain's remarkable capabilities.
Leveraging Temporal Strengths: Methodological and Application-Based Selection
Understanding the brain's dynamic processes requires neuroimaging techniques that can keep pace with neural activity. While various modalities offer insights into brain function, their value is often determined by a key parameter: temporal resolution, or the ability to measure changes in brain activity over time. Electroencephalography (EEG) stands apart for its millisecond-scale temporal precision, enabling researchers to capture high-speed neural phenomena inaccessible to slower hemodynamic-based methods. This capability makes EEG indispensable for studying event-related potentials (ERPs), seizure dynamics, and sleep architecture, where timing is paramount.
Functional magnetic resonance imaging (fMRI) and functional near-infrared spectroscopy (fNIRS) have revolutionized brain mapping but operate on a fundamentally different time scale. Both fMRI and fNIRS measure the hemodynamic response—changes in blood oxygenation that follow neural activity—which unfolds over seconds [31] [2]. In contrast, EEG directly measures the electrical activity of neuronal populations with millisecond precision, providing a direct view of neural dynamics rather than an indirect metabolic correlate [31]. This temporal advantage positions EEG as the modality of choice for investigating the brain's high-speed processes, though each technique offers complementary strengths that can be leveraged in multimodal approaches.
Technical Comparison of Neuroimaging Modalities
Fundamental Principles and Measurement Techniques
EEG records electrical potentials generated by the synchronized firing of cortical neurons, primarily pyramidal cells, via electrodes placed on the scalp. These measurements represent postsynaptic potentials rather than individual action potentials, reflecting the summed electrical activity of large neural populations [31] [32]. The exceptional temporal resolution of EEG—capable of capturing neural dynamics on a millisecond scale—makes it ideal for analyzing rapid cognitive processes like sensory perception, attention, and motor planning [31].
fNIRS monitors cerebral hemodynamic responses by measuring changes in oxygenated (HbO) and deoxygenated hemoglobin (HbR) using near-infrared light. This technique provides an indirect measure of neural activity through neurovascular coupling, with temporal resolution limited by the delay of the hemodynamic response (typically 2-6 seconds) [31] [2]. fNIRS offers better spatial resolution than EEG for surface cortical areas and greater tolerance for movement artifacts, making it suitable for studies in naturalistic settings [31].
fMRI detects brain activity by measuring the Blood Oxygen Level Dependent (BOLD) signal, which reflects changes in deoxygenated hemoglobin due to increased blood flow when brain regions become active. While fMRI provides high spatial resolution (millimeter-level) and whole-brain coverage including subcortical structures, its temporal resolution is constrained by the hemodynamic response, typically sampling at 0.33-2 Hz [2] [3].
Table 1: Technical Specifications of Major Neuroimaging Modalities
| Feature | EEG | fNIRS | fMRI |
|---|---|---|---|
| What It Measures | Electrical activity of neurons | Hemodynamic response (blood oxygenation) | Blood Oxygen Level Dependent (BOLD) signal |
| Temporal Resolution | High (milliseconds) | Low (seconds) | Low (seconds) |
| Spatial Resolution | Low (centimeter-level) | Moderate (better than EEG) | High (millimeter-level) |
| Depth of Measurement | Cortical surface | Outer cortex (~1-2.5 cm deep) | Whole brain (cortical & subcortical) |
| Portability | High (lightweight, wireless systems available) | High (portable, wearable formats) | Low (immobile scanner) |
| Best Use Cases | Fast cognitive tasks, ERPs, seizure detection, sleep research | Naturalistic studies, child development, motor rehabilitation | Spatial mapping, deep brain structures, network connectivity |
Comparative Advantages and Limitations for High-Speed Processes
EEG's unparalleled temporal resolution makes it uniquely suited for capturing neural events that occur in milliseconds, such as the sequential processing stages reflected in ERP components [32]. The direct measurement of electrical activity bypasses the physiological delay inherent in hemodynamic methods, allowing researchers to observe neural processing in real time.
However, this temporal advantage comes with spatial limitations. EEG signals are attenuated and distorted as they pass through the skull and scalp, resulting in limited spatial accuracy [31]. Additionally, EEG is highly susceptible to movement artifacts, requiring controlled environments for optimal data quality [31].
In contrast, fNIRS provides a balance between reasonable spatial resolution for cortical areas and greater tolerance for movement, enabling studies in more ecologically valid settings [31] [3]. fMRI offers the highest spatial resolution and access to subcortical structures but requires complete immobility in a loud, confined scanner environment [2] [3].
For investigating high-speed processes, EEG remains the undisputed gold standard for temporal precision, while fNIRS and fMRI provide complementary spatial information that can be integrated through multimodal approaches.
Event-Related Potentials (ERPs): Millisecond-Scale Brain Dynamics
Fundamentals of the ERP Technique
Event-related potentials (ERPs) are voltage fluctuations in the EEG that are time-locked to specific sensory, cognitive, or motor events [33] [32]. These signals represent complex activity of neural networks responsible for discriminative behavior and novel stimulus recognition [32]. The ERP technique relies on signal averaging to extract these small voltage changes from the ongoing EEG activity, which typically has a much larger amplitude than the ERP components of interest.
The averaging process assumes that the brain's response to repeated similar events is consistent, while background EEG activity and noise are random. By averaging across many trials, the random activity cancels out, leaving the time-locked ERP components [33]. This requires precise temporal records of events, synchronization with EEG data acquisition, strong experimental design, and typically large numbers of trials per experimental condition (approximately 100) to achieve adequate signal-to-noise ratio [33].
ERP components are typically labeled according to their polarity (P for positive, N for negative) and their ordinal position or latency in milliseconds. For example, the N100 is a negative component peaking around 100 milliseconds post-stimulus, while P300 is a positive component peaking around 300 milliseconds [32].
Key ERP Components and Their Functional Significance
Table 2: Major ERP Components and Their Functional Correlates
| Component | Latency (ms) | Polarity | Functional Significance | Clinical Research Applications |
|---|---|---|---|---|
| N100/N1 | 80-120 | Negative | Early sensory processing, attention modulation | Sensory gating in schizophrenia |
| P200/P2 | 150-275 | Positive | Early perceptual analysis, stimulus classification | Information processing deficits |
| N200/N2 | 180-325 | Negative | Deviance detection, conflict monitoring, cognitive control | Cognitive control deficits in ADHD |
| P300/P3 | 250-500 | Positive | Context updating, attention allocation, memory processes | Cognitive impairment in depression, dementia |
| N400 | ~400 | Negative | Semantic processing, language comprehension | Language processing in aphasia |
| CNV | Variable | Negative | Expectation, preparation, motor planning | Attention deficits |
The P300 component, perhaps the most studied ERP, is typically elicited using the "oddball" paradigm, where participants detect infrequent target stimuli among frequent standard stimuli [32]. The P300 reflects cognitive processes such as attention allocation, context updating, and memory activation [33] [32]. It is divided into two subcomponents: P3a, associated with novelty detection and involuntary attention shifting, and P3b, linked to conscious stimulus evaluation and memory operations [32].
The N400 component, first reported by Kutas and Hillyard in 1980, is sensitive to semantic processing and is typically larger for semantically incongruous words in a sentence context [33]. This component has provided valuable insights into the timing of semantic processing during language comprehension.
The Contingent Negative Variation (CNV) is a slow negative wave that develops between a warning stimulus and an imperative stimulus requiring a response, reflecting expectation, preparation, and motor planning [33] [32].
Experimental Paradigms and Protocol Details
Classic Oddball Paradigm:
- Stimuli: Series of frequent "standard" stimuli (e.g., 80%) and rare "target" stimuli (e.g., 20%)
- Task: Participants respond (button press) to target stimuli
- Recording Parameters: 19+ scalp electrodes (10-20 system), sampling rate ≥250 Hz, filter settings (0.1-30 Hz)
- Trials: Minimum 40-60 artifact-free target trials for averaging
- Measures: P300 amplitude and latency at parietal sites (Pz)
Semantic Violation Paradigm:
- Stimuli: Sentences ending with either expected or semantically incongruous words
- Task: Silent reading or semantic judgment
- Measures: N400 amplitude and latency at central sites (Cz)
CNV Paradigm:
- Stimuli: Warning stimulus (S1) followed by imperative stimulus (S1) after fixed interval (1-2 s)
- Task: Speeded response to S2
- Measures: Slow negative wave development between S1 and S2 at central-frontal sites
Seizure Dynamics: Capturing Epileptiform Activity
Interictal Epileptiform Discharges (IEDs) and High-Frequency Oscillations
EEG's millisecond temporal resolution is crucial for detecting and characterizing the brief, transient events that define epileptiform activity. Interictal epileptiform discharges (IEDs)—including spikes (20-70 ms), poly-spikes, sharp waves (70-200 ms), and spike-wave complexes—represent pathological electrophysiological events occurring between seizures in epilepsy patients [34]. These events occur preferentially during non-rapid eye movement (NREM) sleep and are associated with impaired memory and cognition [34].
The high temporal precision of EEG allows for precise identification of IED morphology, which varies between subjects due to different IED sources and brain pathologies [34]. While visual annotation by trained neurologists remains the gold standard for IED identification, automated detection algorithms leveraging machine learning approaches have achieved high precision (94.4%) and sensitivity (94.3%) in intracranial EEG recordings [34].
Intracranial EEG (iEEG) for Seizure Focus Localization
In drug-resistant epilepsy patients being evaluated for surgical treatment, intracranial EEG (iEEG) provides direct recordings from depth electrodes implanted in target regions. iEEG offers superior spatial specificity compared to scalp EEG and can detect high-frequency oscillations (80-500 Hz) that are potential biomarkers of the seizure onset zone [34].
Typical iEEG Protocol for Epilepsy Monitoring:
- Electrode Placement: Depth electrodes with platinum contacts along the shaft, implanted in medial temporal lobe structures (hippocampus, amygdala, entorhinal cortex) and suspected seizure onset zones
- Recording Parameters: Continuous recording sampled at 2000 Hz, referenced to a central scalp electrode
- Annotation: Expert neurologists review montages of intracranial channels, identifying IEDs based on established morphological criteria
- Co-registration: Pre-implantation MRI integrated with post-implantation CT scans for precise electrode localization
Sleep Research: Tracking Neural Oscillations Across Sleep Stages
Sleep Architecture and Phasic Events
EEG is the fundamental tool for sleep staging and monitoring sleep architecture, based on characteristic patterns of neural oscillations that define different sleep stages. The traditional method for sleep scoring follows the Rechtschaffen and Kales (R&K) rules, which involve examination of polysomnograms using combined EEG, electrooculogram (EOG), and electromyogram (EMG) measures [35].
Key EEG signatures in sleep include:
- Sleep spindles: Brief (0.5-2.0 s) bursts of 11-16 Hz activity during Stage II sleep
- K-complexes: High-amplitude negative sharp waves followed by positive components in Stage II sleep
- Slow-wave activity: High-amplitude, low-frequency (<4 Hz) oscillations during deep sleep (N3)
- Rapid eye movements (REMs) and low-voltage mixed-frequency EEG during REM sleep
ERPs During Sleep and Methodological Considerations
ERPs can be recorded during sleep to investigate sensory processing and cognitive function across different vigilance states. However, sleep ERP research requires careful methodological control:
Sleep Stage Monitoring:
- Use Rechtschaffen and Kales rules with experienced scorers
- Supplement with quantitative measures like alpha slow-wave index (ASI) to distinguish wakefulness from sleep
- Employ time-frequency analysis to validate sleep stage classification
First Night Effect (FNE) Control:
- FNE characteristics: increased sleep fragmentation, increased sleep latencies, decreased REM sleep, decreased slow wave sleep
- Recommendations: Use two consecutive nights with testing on the second night, or test healthy young participants in comfortable settings during the second half of the night
Sleep Disorder Screening:
- Pre-recording interview exploring sleep habits and behaviors
- Application of subjective sleep abnormality scales
- Polysomnographic recording with infrared-video monitoring to rule out sleep disorders
Multimodal Integration: Leveraging Complementary Strengths
EEG-fNIRS Integration
Combining EEG with fNIRS provides a powerful multimodal approach that captures both the electrical neural activity and the accompanying hemodynamic response. This integration is particularly valuable for brain-computer interfaces (BCIs), where EEG provides millisecond temporal resolution for rapid classification, while fNIRS offers better spatial localization and resistance to motion artifacts [6] [9].
In motor imagery BCIs, simultaneous EEG-fNIRS recording has demonstrated improved classification accuracy (83.26% in recent studies) compared to unimodal approaches [9]. The complementary nature of these signals allows for more robust system performance, with EEG capturing transient task-related dynamics and fNIRS providing sustained activation patterns.
Implementation Considerations for EEG-fNIRS Integration:
- Sensor Placement Compatibility: Both systems often use the international 10-20 system for placement
- Hardware Integration: Synchronization via triggers or shared clock systems
- Motion Artifact Management: fNIRS is more motion-tolerant, while EEG requires stricter movement control
- Data Fusion: Separate preprocessing pipelines followed by integration using techniques like joint Independent Component Analysis (jICA) or machine learning approaches
EEG-fMRI Integration
While simultaneous EEG-fMRI recording presents technical challenges due to potential electromagnetic interference, the combination offers unique insights by linking EEG's temporal precision with fMRI's spatial specificity. This approach is particularly valuable for localizing the generators of EEG phenomena and investigating neurovascular coupling.
In epilepsy monitoring, simultaneous EEG-fMRI can help localize the seizure onset zone by capturing interictal discharges on EEG and correlating them with BOLD signal changes. In cognitive neuroscience, this integration helps bridge the gap between the millisecond dynamics of information processing (ERPs) and the underlying distributed neural networks.
Essential Research Reagents and Materials
Table 3: Key Research Reagents and Solutions for EEG Research
| Item | Function/Application | Specifications |
|---|---|---|
| EEG Electrodes | Electrical signal acquisition from scalp | Ag/AgCl sintered electrodes; impedance <5 kΩ |
| Electrode Gel/Cream | Ensuring conductive connection between scalp and electrode | Electrolyte gel with chloride composition |
| Abrading Paste | Light skin abrasion to reduce impedance | Mild abrasive with pumice or similar material |
| Conductive Adhesive | Securing electrodes, particularly for long-term recordings | Medical-grade adhesive with conductive properties |
| Electrode Caps/Headsets | Standardized electrode positioning | Compatible with 10-20, 10-10, or 10-5 systems |
| Impedance Checker | Verifying electrode-skin contact quality | Range: 1-100 kΩ; portable for bedside use |
| Event Marker System | Synchronizing external events with EEG recording | Optical, electrical, or serial port triggers |
| Faraday Cage/Shielded Room | Minimizing environmental electromagnetic interference | Copper mesh shielding; electrical grounding |
EEG's millisecond temporal resolution establishes it as the indispensable tool for investigating high-speed neural processes, particularly event-related potentials, seizure dynamics, and sleep architecture. While fNIRS and fMRI offer complementary strengths in spatial localization and access to deeper brain structures, neither can match EEG's ability to capture the rapid temporal dynamics of neural communication.
The continuing evolution of multimodal integration approaches promises even greater insights, combining EEG's temporal precision with the spatial strengths of other modalities. For researchers focused on the timing of neural events—from the 100-millisecond scale of early sensory processing to the brief paroxysmal discharges of epileptic tissue—EEG remains the cornerstone technology for illuminating the brain's high-speed operations.
Functional near-infrared spectroscopy (fNIRS) has emerged as a pivotal neuroimaging technology for monitoring sustained cortical activity, occupying a unique niche between the high temporal resolution of electroencephalography (EEG) and the high spatial resolution of functional magnetic resonance imaging (fMRI). This guide provides a comprehensive comparison of fNIRS against EEG and fMRI, detailing its specific advantages for applications in cognitive workload assessment, emotional state monitoring, and motor rehabilitation. We present quantitative performance data, detailed experimental protocols, and essential methodological considerations to guide researchers in selecting and implementing appropriate neuroimaging modalities for their specific research objectives.
Understanding brain function in real-world scenarios requires neuroimaging technologies that can capture neural activity over sustained periods with practical feasibility. While EEG measures electrical activity directly with millisecond precision, its spatial resolution is limited due to the skull's blurring effect on electrical signals [36]. Conversely, fMRI provides exquisite spatial detail of deep brain structures but is constrained by its immobility, low temporal resolution, and sensitivity to motion artifacts [3] [2]. fNIRS occupies a crucial middle ground, measuring hemodynamic responses associated with neural activity with several distinct advantages: superior motion tolerance, portability for real-world studies, and moderate spatial resolution specifically for cortical surface regions [36] [3]. This combination makes fNIRS particularly suited for investigating sustained cognitive processes, emotional processing, and rehabilitation paradigms where ecological validity and participant mobility are prioritized.
Technical Comparison of Neuroimaging Modalities
Core Performance Metrics
Table 1: Comprehensive comparison of fNIRS, EEG, and fMRI across key technical parameters.
| Feature | fNIRS | EEG | fMRI |
|---|---|---|---|
| What It Measures | Hemodynamic response (HbO, HbR) [36] | Electrical activity of neurons [36] | Blood-oxygen-level-dependent (BOLD) signal [3] |
| Temporal Resolution | Low (seconds) [36] | High (milliseconds) [36] | Low (seconds) [2] |
| Spatial Resolution | Moderate (cortical surface) [36] | Low (centimeter-level) [36] | High (millimeter-level) [2] |
| Depth of Measurement | Outer cortex (~1-2.5 cm deep) [36] | Cortical surface [36] | Whole brain (cortical & subcortical) [2] |
| Portability | High (wearable systems available) [36] [3] | High (lightweight, wireless systems) [36] | None (requires scanner environment) [3] |
| Motion Tolerance | Relatively robust [36] [3] | Susceptible to movement artifacts [36] | Highly sensitive to motion [3] |
| Setup Complexity | Moderate (optode placement) [36] | Moderate (electrode gel application) [36] | High (requires specialized facility) [3] |
| Approximate Cost | Moderate [3] | Generally lower [36] | Very high [3] |
| Best Use Cases | Naturalistic studies, child development, motor rehab [36] | Fast cognitive tasks, ERP studies, sleep research [36] | Deep brain structures, precise spatial localization [2] |
Signal Characteristics and Physiological Basis
fNIRS measures neural activity indirectly through neurovascular coupling, detecting changes in oxygenated (HbO) and deoxygenated hemoglobin (HbR) concentrations in the blood using near-infrared light (650-1000 nm) [3]. This hemodynamic response typically unfolds over 2-6 seconds, similar to the fMRI BOLD response but with direct measurement of both HbO and HbR [36] [37]. In contrast, EEG directly measures postsynaptic electrical potentials from synchronized neuronal firing with millisecond precision, providing immediate temporal markers of neural processing but with limited spatial specificity [36]. fMRI detects magnetic susceptibility changes associated with blood oxygenation, offering whole-brain coverage including subcortical structures, but with inherent temporal delays due to the sluggish hemodynamic response [2].
Figure 1: Signaling pathways for major neuroimaging modalities. fNIRS and fMRI both measure the hemodynamic response to neural activity but through different physical mechanisms, while EEG measures electrical activity directly.
Experimental Applications and Protocols
Cognitive Workload Assessment
Experimental Protocol: fNIRS excels at measuring sustained prefrontal cortex activity during cognitive workload tasks. A standard protocol involves administering the N-back task (varying difficulty levels from 1-back to 3-back) while placing fNIRS optodes over the prefrontal cortex [36]. Participants are typically presented with a sequence of stimuli and must indicate when the current stimulus matches the one presented N steps earlier. Each block lasts 2-3 minutes with resting baselines between conditions.
Key Findings: Studies consistently show dose-dependent increases in HbO concentration in the dorsolateral and frontopolar prefrontal regions with increasing cognitive load [36]. fNIRS-based workload classification using support vector machines (SVM) has achieved accuracies exceeding 91% in discriminating between high and low mental workload states [38]. The portability of fNIRS enables workload assessment in real-world environments such as driving simulators, classroom settings, and clinical monitoring scenarios where EEG would suffer from motion artifacts and fMRI is impractical [36].
Motor Rehabilitation and Imagery
Experimental Protocol: For motor rehabilitation applications, fNIRS is typically deployed over the primary motor cortex (M1) and supplementary motor area. Participants perform either actual movements or motor imagery tasks (imagining movements without physical execution) [38] [39]. A typical session involves alternating blocks of rest and activity (20-30 seconds each), with multiple trials per condition. For patients with severe motor impairments, the decoded motor intention can control assistive devices like robotic exoskeleton hands [38].
Key Findings: fNIRS reliably detects hemodynamic changes in the contralateral motor cortex during both actual movement and motor imagery [39]. In hybrid fNIRS-EEG systems, this capability has been leveraged for brain-computer interfaces (BCIs) that provide neurofeedback during rehabilitation training [39]. Studies have demonstrated classification accuracies of 67-92% for discriminating between different motor states, with higher accuracies typically achieved through multimodal fNIRS-EEG approaches [38] [39]. The technology's relative robustness to movement makes it particularly valuable for rehabilitation settings where complete stillness is impractical.
Emotional State Monitoring
Experimental Protocol: Emotional processing studies with fNIRS typically focus on the prefrontal cortex, which is critically involved in emotion regulation [36]. Experimental paradigms include viewing emotionally valenced images (from standardized sets like IAPS), recalling autobiographical emotional memories, or exposure to social stress tests. Blocked designs with 1-2 minute conditions alternating between different emotional states (e.g., neutral, positive, negative) are commonly employed.
Key Findings: fNIRS studies have identified distinct prefrontal activation patterns associated with different emotional valences, though the spatial specificity is more limited than fMRI [36] [40]. Lateralization effects (differential left vs. right prefrontal engagement) have been observed during emotion processing, consistent with the fMRI literature [36]. In clinical applications, fNIRS has shown potential as an objective marker in psychiatric disorders, though diagnostic specificity remains challenging with reported concordance rates of 38-44% for distinguishing disorders like depression and bipolar disorder [40].
Multimodal Integration Approaches
fNIRS-EEG Synergy
The combination of fNIRS and EEG creates a powerful multimodal approach that compensates for the limitations of each individual modality [30] [39]. EEG provides millisecond-scale temporal resolution for capturing rapid neural dynamics, while fNIRS offers better spatial localization of the underlying cortical activity [36] [30].
Implementation Protocol: Simultaneous fNIRS-EEG recording typically uses integrated caps with pre-defined openings for both optodes and electrodes, often following the international 10-20 system for standardized placement [36]. Hardware synchronization is achieved via TTL pulses or shared clock systems. Data processing requires separate preprocessing pipelines followed by integrated analysis approaches such as joint Independent Component Analysis (jICA) or machine learning models that combine features from both modalities [36].
Performance Gains: Research demonstrates that joint fNIRS-EEG source reconstruction can resolve neuronal sources separated by as little as 2.3-3.3 cm and 50 ms—a feat neither modality could achieve independently [30]. This enhanced spatiotemporal resolution is particularly valuable for tracking information flow in neural circuits and developing more responsive BCIs [30] [39].
Figure 2: Workflow for simultaneous fNIRS-EEG data acquisition and analysis. The multimodal approach leverages complementary signals through specialized fusion algorithms to achieve enhanced spatiotemporal resolution.
fNIRS-fMRI Validation and Complementarity
While fNIRS and fMRI both measure hemodynamic responses, their integration provides unique opportunities for validation and extended application [3] [2] [37]. fMRI serves as a gold standard for spatial localization, while fNIRS enables follow-up studies in naturalistic settings.
Implementation Protocol: Simultaneous fNIRS-fMRI recording requires MRI-compatible fNIRS systems with specialized optodes and long optical fibers to operate within the scanner environment [37]. Careful positioning is needed to avoid interference with the head coil. Temporal synchronization is typically achieved using the scanner's trigger pulses. Concurrent recording during motor tasks (e.g., finger tapping) has shown strong spatial correlation between fNIRS activations and fMRI BOLD signals in the motor cortex [37].
Applications: This multimodal approach is particularly valuable for validating fNIRS localization accuracy and studying neurovascular coupling dynamics [2]. Once validated, fNIRS can be deployed independently for longitudinal monitoring, pediatric studies, or ecological assessments where fMRI is impractical [3] [2].
Practical Implementation Guide
The Researcher's Toolkit
Table 2: Essential research reagents and solutions for fNIRS experimentation.
| Item | Function | Application Notes |
|---|---|---|
| fNIRS System | Measures light absorption to compute HbO/HbR concentrations | Choose portable/wearable systems for ecological studies; MRI-compatible versions for simultaneous fMRI [37] |
| Optodes & Holders | Deliver light to scalp and detect returning light | Use spring-loaded holders for consistent pressure; MRI-compatible versions for multimodal studies [36] |
| EEG Integration Cap | Enables simultaneous fNIRS-EEG recording | Prefer caps with predefined openings for both modalities following 10-20 system [36] |
| 3D Digitizer | Records precise optode/electrode locations | Enables accurate co-registration with anatomical brain atlas [3] |
| Synchronization Interface | Aligns fNIRS with other modality timestamps | TTL pulses or shared clock systems for multimodal studies [36] |
| Prefrontal Cortex Array | Specific optode configuration for cognitive studies | Dense coverage over frontal regions for workload/emotion studies [36] |
| Motor Cortex Array | Specific optode configuration for motor studies | Coverage over primary motor and supplementary motor areas [39] |
Selection Guidelines Based on Research Goals
Choose fNIRS when: Your research requires monitoring sustained cortical activity in naturalistic settings, involves populations prone to movement (children, patients), prioritizes portability and cost-effectiveness, or focuses specifically on surface cortical regions [36] [3].
Choose EEG when: Your research questions involve millisecond-scale temporal dynamics, require tracking rapid neural oscillations, or involve environments where head-mounted systems are impractical [36].
Choose fMRI when: Your research requires whole-brain coverage including subcortical structures, demands millimeter-scale spatial precision, or involves detailed anatomical localization [2].
Opt for multimodal fNIRS-EEG when: You need both high temporal resolution and improved spatial localization, particularly for BCIs, rehabilitation monitoring, or studying complex cognitive processes [30] [39].
fNIRS represents an optimal balance of portability, spatial specificity, and practical implementation for studying sustained cortical activity across diverse research domains. While no single neuroimaging modality excels across all parameters, fNIRS occupies a crucial niche that enables researchers to investigate brain function in ecologically valid contexts that closely mirror real-world conditions. The technology's particular strengths in monitoring cognitive workload, emotional states, and motor rehabilitation processes—especially when combined with complementary modalities like EEG—make it an indispensable tool in the modern neuroimaging toolkit. As methodological advances continue to enhance its spatial resolution and analytical sophistication, fNIRS is poised to expand further into clinical applications and naturalistic research environments where traditional neuroimaging technologies face inherent limitations.
Functional Magnetic Resonance Imaging (fMRI) remains the preeminent non-invasive tool for high-resolution spatial mapping of brain function. While techniques like EEG and fNIRS offer superior temporal resolution or portability, fMRI provides unparalleled access to the brain's deep structures and detailed network organization. This guide objectively compares fMRI's capabilities against other modalities, framed within the critical trade-off between spatial and temporal resolution in neuroimaging.
Comparative Analysis of Neuroimaging Modalities
The choice of neuroimaging technique is governed by a fundamental trade-off: methods that excel at localizing brain activity in space (fMRI) typically do so at the cost of capturing its rapid millisecond-scale timing, and vice-versa [41] [42]. The table below summarizes the core technical specifications of fMRI, fNIRS, and EEG.
Table 1: Technical Comparison of Key Neuroimaging Modalities
| Feature | fMRI | fNIRS | EEG |
|---|---|---|---|
| What It Measures | Blood Oxygenation Level Dependent (BOLD) signal (hemodynamic response) [43] | Changes in oxygenated (HbO) & deoxygenated hemoglobin (HbR) [41] | Electrical potentials from synchronized neuronal firing [41] [13] |
| Spatial Resolution | High (Millimeter-level) [13] [43] | Moderate (Centimeter-level), limited to cortex [41] [13] | Low (Centimeter-level) [41] [13] |
| Temporal Resolution | Low (Seconds), limited by hemodynamic lag [43] | Moderate (Seconds) [13] | Very High (Millisecond-level) [41] [44] |
| Depth of Measurement | Whole brain, including deep structures (e.g., thalamus, hippocampus) [43] | Superficial cortex (1-2 cm depth) [13] [43] | Cortical surface [41] |
| Portability | Low, requires restrictive scanner environment [13] [43] | High, wearable systems for naturalistic settings [41] [43] | High, lightweight wireless systems available [41] |
Experimental Protocols for Spatial Mapping and Connectivity
High-Resolution Functional Localization of Thalamic Nuclei
Objective: To investigate task-dependent functional connectivity of specific thalamic nuclei using high-field fMRI [45].
Protocol:
- Participants: Healthy adults.
- Imaging: A 9.4 Tesla MRI scanner for high-resolution T1-weighted anatomical images and task-based fMRI (GE-EPI sequence, TR=2s) [45].
- Tasks: Block-design paradigms:
- Motor Task: Visually guided finger-tapping with the right hand.
- Sensory Task: Passive tactile stimulation of the fingertips via air pulses [45].
- Analysis: General Linear Model (GLM) analysis identified activation in thalamic nucleus groups (lateral and pulvinar) during tasks. Functional connectivity of these activated nuclei with cortical, subcortical, and cerebellar regions was then assessed [45].
Supporting Data: This protocol demonstrated that thalamic nuclei are not static relays but flexible hubs. They showed distinct, task-specific functional connectivity patterns with various brain regions during motor vs. sensory tasks, which could only be delineated with high spatial resolution [45].
Precision Mapping of Individual Brain Networks
Objective: To determine if whole-brain, within-individual functional networks can be estimated using task-based fMRI data alone, rather than the standard resting-state data [46].
Protocol:
- Data Acquisition: fMRI data is collected from individuals performing one or more cognitive or motor tasks.
- Network Estimation: Correlation matrices are estimated from the task-based fMRI time series.
- Validation: The resulting networks are compared to those derived from traditional resting-state data from the same individual for similarity and spatial overlap [46].
Supporting Data: This study found that correlation matrices from task data were similar to those from rest, with the amount of data being the most critical factor. Precision networks from task fMRI showed strong spatial overlap with resting-state networks and predicted the same triple functional dissociations in independent data [46]. This confirms a stable, idiosyncratic network architecture within an individual that persists across task states.
Multimodal Integration for High Spatiotemporal Resolution
Objective: To overcome the spatiotemporal trade-off by integrating MEG and fMRI data to estimate latent cortical source activity with high resolution in both space and time [42].
Protocol:
- Stimuli: Narrative stories presented over several hours.
- Data Collection: Whole-head MEG data is collected. fMRI data from the same stimuli is sourced from a separate, open dataset [42].
- Model Architecture: A transformer-based encoding model is trained to predict both MEG and fMRI signals simultaneously. The model incorporates anatomical information and biophysical forward models.
- Output: The model produces estimates of source activity on the cortical surface with high spatial and temporal fidelity [42].
Supporting Data: The model predicted MEG data better than single-modality encoding models. Crucially, the estimated source activity generalized effectively, predicting electrocorticography (ECoG) signals in a new dataset better than a model trained directly on the ECoG data, validating its physiological accuracy [42].
Signaling Pathways and Experimental Workflows
From Neural Activity to Hemodynamic Signal
The following diagram illustrates the neurovascular coupling pathway, the biological basis for fMRI and fNIRS signals, and contrasts it with the direct electrical measurement of EEG.
Multimodal Data Fusion Experimental Workflow
This workflow outlines the process for integrating data from multiple neuroimaging modalities, such as MEG and fMRI, to achieve high spatiotemporal resolution.
The Scientist's Toolkit: Key Research Reagents & Materials
Table 2: Essential Materials for High-Resolution fMRI and Multimodal Studies
| Item | Function / Description | Example Use Case |
|---|---|---|
| High-Field MRI Scanner (e.g., 9.4T) | Provides superior signal-to-noise ratio for imaging small subcortical structures [45]. | High-resolution mapping of thalamic nuclei [45]. |
| Biophysical Forward Models | Mathematical models predicting sensor signals from neural source activity (e.g., lead-field matrix for MEG) [42]. | Core component in MEG-fMRI fusion models for source estimation [42]. |
| Structural Decoupling Index (SDI) | A graph-based metric quantifying the (dis)alignment between structural and functional networks in a region [29]. | Studying structure-function coupling across modalities (EEG/fNIRS) and brain states [29]. |
| Transformer-Based Encoding Model | A deep learning architecture that learns to predict brain activity from stimulus features [42]. | Integrating MEG and fMRI to estimate latent source dynamics [42]. |
| International 10-20/10-5 System | Standardized scheme for placing EEG electrodes or fNIRS optodes on the scalp [44] [29]. | Ensures consistent and replicable sensor placement across subjects and studies [29]. |
fMRI's dominance in spatial mapping of deep brain structures and network connectivity is well-established, providing irreplaceable millimeter-scale resolution for localization. However, the evolving landscape of neuroimaging emphasizes integration rather than substitution. The future lies in sophisticated multimodal approaches that combine fMRI's spatial precision with the high temporal resolution of EEG or the portability of fNIRS. These hybrid frameworks, powered by advanced computational models, are paving a practical route toward a long-sought goal in neuroscience: millisecond-and-millimeter resolution mapping of human brain function.
In the quest to map human brain activity, the neuroimaging community has long sought a single modality that offers both the exquisite temporal resolution to capture neural events in real-time and the spatial stability to localize them precisely. This ideal tool remains elusive. Instead, a powerful paradigm has emerged: the combination of complementary technologies. Electroencephalography (EEG) and functional Near-Infrared Spectroscopy (fNIRS) represent two such complementary approaches. EEG measures the brain's electrical activity directly from the scalp surface, providing millisecond-level temporal tracking of neural events [47] [48]. In contrast, fNIRS uses near-infrared light to measure hemodynamic changes—shifts in blood oxygenation that are an indirect metabolic correlate of neural activity—offering superior resistance to movement artifacts and better spatial localization than EEG, though with inherently slower response times [49] [2]. Framing this within broader neuroimaging research, functional Magnetic Resonance Imaging (fMRI) provides high spatial resolution for deep brain structures but is constrained by low temporal resolution, high cost, and lack of portability, making it unsuitable for many real-world BCI applications [2]. This guide objectively compares the performance of EEG and fNIRS BCI technologies, providing the experimental data and methodological context to help researchers select the optimal tool for their specific neuroscientific or clinical questions.
Technical Showdown: A Quantitative Comparison of EEG and fNIRS
The core strengths and limitations of EEG and fNIRS stem from the fundamental physiological phenomena they measure. The following table provides a direct, data-driven comparison of these two dominant non-invasive BCI modalities.
Table 1: Technical Performance Comparison of EEG and fNIRS BCIs
| Performance Metric | Electroencephalography (EEG) | Functional Near-Infrared Spectroscopy (fNIRS) |
|---|---|---|
| Primary Signal Source | Electrical potentials from synaptic activity [48] | Hemodynamic changes (HbO/HbR concentration) [49] [2] |
| Temporal Resolution | Millisecond-range (high) [44] | Slower, hemodynamic delay of 4-6s [2] |
| Spatial Resolution | ~2 cm at best (low) [44] | 1-3 cm (moderate) [2] |
| Key Artifact Vulnerability | Sensitive to electrical and motion noise [47] [49] | Robust to electrical noise; sensitive to scalp blood flow [49] [2] |
| Portability & Cost | High portability, cost-effective [47] | Portable, cost-effective [44] |
| Typical BCI Applications | Real-time control, communication spellers, neurogadgets [50] [48] | Motor imagery, prosthetic control, clinical monitoring [49] [51] |
This quantitative profile reveals the inherent trade-offs. EEG's supreme temporal resolution makes it the preferred choice for applications requiring rapid, real-time control, such as moving a cursor or selecting a letter on a screen. However, its susceptibility to noise can be a significant barrier in real-world settings. fNIRS sacrifices speed for signal stability and better spatial definition, which is advantageous for tasks like controlling a prosthetic limb or in rehabilitation settings where patient movement is expected [49].
The Synergistic Frontier: Hybrid EEG-fNIRS BCIs
Recognizing that neither modality is perfect, the field is increasingly moving toward hybrid BCIs that integrate EEG and fNIRS to leverage their complementary strengths [52] [53]. The synergy is logical: EEG provides the high-temporal-resolution "when," and fNIRS provides the more stable, spatially resolved "where."
Advanced data fusion methodologies are key to unlocking this synergy. A leading approach is the Spatial–Temporal Alignment Network (STA-Net), an end-to-end deep learning framework designed to overcome the inherent temporal misalignment between fast EEG signals and slower fNIRS hemodynamics [52]. The STA-Net architecture uses an fNIRS-guided spatial alignment layer to identify task-sensitive brain regions and an EEG-guided temporal alignment layer to dynamically adjust the fNIRS delay, which can vary by subject and task. This sophisticated fusion has yielded remarkable performance, with one study reporting classification accuracies of 69.65% for motor imagery (MI), 85.14% for mental arithmetic (MA), and 79.03% for word generation (WG) tasks, outperforming state-of-the-art single-modality algorithms [52].
Table 2: Experimental Performance of BCI Modalities in Mental Task Classification
| Mental Task | EEG-Only Accuracy | fNIRS-Only Accuracy | Hybrid EEG-fNIRS Accuracy |
|---|---|---|---|
| Motor Imagery (MI) | Information Missing | Information Missing | 69.65% [52] |
| Mental Arithmetic (MA) | Information Missing | Information Missing | 85.14% [52] |
| Word Generation (WG) | Information Missing | Information Missing | 79.03% [52] |
| 2-Class Hand Gripping | Information Missing | ~85-90% [51] | Information Missing |
The hybrid advantage is further validated in clinical populations. The HEFMI-ICH dataset, which includes data from intracerebral hemorrhage (ICH) patients, shows that hybrid systems can achieve 5-10% improvement in classification accuracy compared to unimodal systems, a critical enhancement for precision rehabilitation [53].
Experimental Protocols and Decoding Methodologies
Protocol for Real-Time EEG Decoding of Individual Finger Movements
A groundbreaking 2025 study demonstrated real-time, non-invasive robotic hand control at the individual finger level using EEG [50].
- Objective: To decode movement execution (ME) and motor imagery (MI) of individual fingers and translate the signals into real-time robotic finger motions.
- Participants: 21 able-bodied, experienced BCI users.
- Task Paradigm: Participants executed or imagined movements of their right-hand thumb, index, and pinky fingers in response to visual cues. The paradigm included both binary (thumb vs. pinky) and ternary (thumb vs. index vs. pinky) classification tasks.
- Data Acquisition: EEG signals were recorded using a multi-electrode cap following standard placement systems.
- Signal Processing & Decoding: A deep neural network (EEGNet-8.2) was implemented for real-time decoding. A "fine-tuning" mechanism was used, where a base model trained on initial data was updated with data from the same session to adapt to inter-session variability.
- Real-Time Feedback: Participants received two forms of feedback: visual (the target finger on a screen changed color) and physical (a robotic hand moved the corresponding finger).
- Results: The system achieved real-time decoding accuracies of 80.56% for two-finger MI tasks and 60.61% for three-finger MI tasks, demonstrating the feasibility of naturalistic, dexterous non-invasive BCI control [50].
Protocol for fNIRS-Based Classification of Hand Gripping Motions
A 2025 study showcased the power of advanced deep learning for improving fNIRS-BCI classification of motor tasks [51].
- Objective: To classify fNIRS signals associated with a two-class hand-gripping motor activity using novel deep learning-derived feature stacking.
- Participants: 20 healthy participants.
- Task Paradigm: Participants performed repetitive hand gripping motions, with data structured into distinct trials for each class.
- Data Acquisition: fNIRS signals were collected using a commercial neuroimaging system, measuring changes in oxygenated and deoxygenated hemoglobin.
- Signal Preprocessing: Data was preprocessed using nirsLAB software to filter out physiological artifacts and noise.
- Feature Extraction & Classification: The study proposed two novel methods: a "stack" method using CNNs, LSTMs, and Bi-LSTMs to extract features classified by a stack model; and an "fft" method that enhanced these features using Fast Fourier Transformation before stacking.
- Results: The proposed fft and stack methods achieved classification accuracies of 90.11% and 87.00%, respectively, significantly outperforming conventional CNN (85.16%), LSTM (79.46%), and Bi-LSTM (81.88%) models [51].
Visualizing the Signaling Pathways and Experimental Workflow
Neural Signaling and BCI Measurement Pathways
Neural Signaling and BCI Measurement Pathways
Hybrid EEG-fNIRS Experimental Data Workflow
Hybrid EEG-fNIRS Experimental Data Workflow
The Scientist's Toolkit: Essential Research Reagents and Materials
Translating BCI research from concept to reality requires a suite of specialized hardware and software. The following table details key components used in state-of-the-art experiments, providing a resource for laboratory setup and experimental design.
Table 3: Essential Research Tools for EEG and fNIRS BCI Experiments
| Tool Name / Category | Specification / Type | Primary Function in BCI Research |
|---|---|---|
| g.HIamp Amplifier [53] | EEG Signal Amplifier | High-quality, multi-channel acquisition of electrical brain signals at high sampling rates (e.g., 256 Hz). |
| NirScan System [53] | fNIRS Acquisition Unit | A continuous-wave system for measuring hemodynamic responses via near-infrared light at specified wavelengths. |
| Hybrid EEG-fNIRS Cap [53] | Integrated Sensor Cap | A custom-designed headgear co-locating EEG electrodes and fNIRS optodes according to the 10-20 system for synchronized data collection. |
| Dry Electrodes [54] | EEG Sensor | Enable quicker setup and improved user comfort by eliminating the need for conductive gel, favorable for consumer applications. |
| EEGNet [50] [52] | Deep Learning Algorithm | A compact convolutional neural network architecture specifically designed for EEG-based BCI classification tasks. |
| STA-Net [52] | Deep Learning Fusion Model | An end-to-end network for spatially and temporally aligning hybrid EEG-fNIRS signals to improve classification accuracy. |
| E-Prime / PsychToolbox [53] | Experimental Control Software | Presents visual/auditory cues and sends synchronization triggers to ensure temporal alignment between task paradigms and data recording. |
The comparison between EEG and fNIRS is not a contest with a single winner but a guide for strategic selection. EEG remains the undisputed champion for BCI applications demanding the fastest possible real-time response, such as controlling a robotic hand's individual fingers [50]. fNIRS offers a robust alternative where signal stability, user movement, and superior spatial localization are prioritized over raw speed, such as in prolonged rehabilitation sessions or for monitoring specific cortical regions [49] [51]. The most promising future, however, lies in integration, not isolation. The emergence of sophisticated hybrid systems and fusion algorithms like STA-Net demonstrates that leveraging the complementary strengths of EEG's speed and fNIRS's stability can create a whole that is greater than the sum of its parts, pushing the boundaries of what is possible in non-invasive brain-computer interfacing [52] [53].
In the quest to understand brain function and diagnose neurological disorders, researchers and clinicians rely on a suite of non-invasive neuroimaging technologies. Among these, electroencephalography (EEG), functional near-infrared spectroscopy (fNIRS), and functional magnetic resonance imaging (fMRI) stand as pillars of modern neuroscience. Each technique offers a unique window into brain activity by measuring different physiological phenomena with distinct temporal and spatial characteristics. The choice of technique is often a trade-off, particularly regarding temporal resolution—the ability to precisely track the timing of neural events. This comparative guide examines the performance of EEG, fNIRS, and fMRI through the critical lens of temporal resolution, focusing on their applications in epilepsy diagnosis and therapy response monitoring. Understanding these fundamental differences enables researchers to select optimal methodologies and interpret neuroimaging data within the constraints of each technology's temporal capabilities.
Fundamental Technical Comparison
EEG, fNIRS, and fMRI measure fundamentally different physiological processes associated with brain activity. EEG records the brain's electrical activity directly from the scalp surface, detecting voltage fluctuations resulting from ionic current flows within neurons. This direct measurement provides millisecond-level temporal resolution, allowing it to capture rapidly changing neural dynamics such as epileptic spikes and neural oscillations [55] [56]. However, as electrical signals pass through the skull and scalp, they blur, resulting in limited spatial resolution at the centimeter level [55].
In contrast, fNIRS measures hemodynamic responses, specifically changes in oxygenated (HbO) and deoxygenated hemoglobin (HbR) concentrations in the blood, using near-infrared light. This indirect measure of neural activity, governed by neurovascular coupling, provides temporal resolution on the scale of seconds [55] [2]. While slower than EEG, fNIRS offers better spatial resolution for cortical surface areas and is more tolerant of movement artifacts [55].
fMRI also measures hemodynamic activity but does so by detecting blood-oxygen-level-dependent (BOLD) signals through magnetic resonance imaging. Although it provides excellent spatial resolution (millimeter-level) and whole-brain coverage including subcortical structures, its temporal resolution is the slowest among the three, typically ranging from 1-4 seconds, as it tracks the slow hemodynamic response that lags 4-6 seconds behind neural activity [2] [3].
Table 1: Technical Specifications and Performance Comparison of EEG, fNIRS, and fMRI
| Feature | EEG | fNIRS | fMRI |
|---|---|---|---|
| What It Measures | Electrical activity from neurons | Hemodynamic response (HbO, HbR) | Hemodynamic response (BOLD signal) |
| Temporal Resolution | High (milliseconds) [55] | Moderate (seconds) [55] | Low (seconds) [2] |
| Spatial Resolution | Low (centimeter-level) [55] | Moderate (better than EEG) [55] | High (millimeter-level) [2] |
| Depth of Measurement | Cortical surface [55] | Outer cortex (1-2.5 cm deep) [55] | Whole brain (cortical & subcortical) [2] |
| Key Strength | Captures rapid neural dynamics | Good balance of portability & spatial resolution | Gold standard for spatial localization |
| Primary Limitation | Poor spatial localization | Limited to cortical surface; indirect measure | Slow temporal resolution; expensive, immobile |
The following diagram illustrates the relationship between temporal and spatial resolution for these three modalities, highlighting their complementary nature:
Figure 1: Temporal vs. Spatial Resolution Trade-offs in Neuroimaging. EEG excels in temporal resolution but has poor spatial resolution, while fMRI provides excellent spatial resolution with limited temporal precision. fNIRS offers a middle ground with moderate capabilities in both dimensions [55] [2].
Experimental Data and Clinical Performance
Epilepsy Diagnosis and Monitoring
In epilepsy diagnosis, where capturing rapid, transient neural events is crucial, the high temporal resolution of EEG makes it the gold standard for detecting interictal spikes and seizure onset [57]. However, fNIRS provides complementary information through its hemodynamic measurements, revealing ictal and postictal hemodynamic changes in the epileptic focus with better spatial resolution than EEG [57]. Simultaneous EEG-fNIRS recordings have demonstrated correlated electrical and hemodynamic activities during epileptic events, offering a more comprehensive picture of epileptogenesis [58] [57]. Specifically, fNIRS can detect changes in cytochrome c oxidase (CcO), a crucial enzyme in cellular respiration, providing insight into the metabolic dimension of epilepsy related to mitochondrial dysfunction [57]. fMRI's utility in epilepsy is primarily for presurgical mapping due to its excellent spatial localization, but its poor temporal resolution limits its use for capturing dynamic seizure activity [2].
Therapy Response Monitoring
For monitoring treatment response in psychiatric disorders such as depression and schizophrenia, fNIRS has emerged as a promising tool due to its portability, cost-effectiveness, and tolerance of movement [59]. Studies utilizing verbal fluency tasks and dorsolateral prefrontal cortex (DLPFC)-targeted paradigms have shown that fNIRS can detect hemodynamic changes associated with pharmacological, psychotherapeutic, and neuromodulatory treatments [59]. While EEG can track rapid neurophysiological changes with millisecond precision, fNIRS provides more localized information about cortical activation patterns relevant to treatment efficacy. fMRI remains valuable for detailed baseline assessments and whole-brain connectivity analyses but is less practical for frequent monitoring due to cost and logistical constraints [59].
Table 2: Clinical Applications and Experimental Evidence for EEG, fNIRS, and fMRI
| Clinical Domain | EEG Evidence & Protocols | fNIRS Evidence & Protocols | fMRI Evidence & Protocols |
|---|---|---|---|
| Epilepsy | Gold standard for seizure detection [57].Protocol: Long-term monitoring with scalp or intracranial electrodes. | Detects hemodynamic changes in epileptic focus [57].Protocol: Simultaneous with EEG during seizure tasks. | Presurgical mapping of epileptogenic zones [2].Protocol: Ictal and interictal BOLD imaging. |
| Psychiatric Therapy Monitoring | Tracks neurophysiological changes with high temporal resolution [59]. | Prefrontal cortex activation during cognitive tasks (e.g., verbal fluency) predicts treatment response [59]. | Whole-brain functional connectivity patterns associated with treatment efficacy [59]. |
| Motor Rehabilitation | Movement-related cortical potentials and sensorimotor rhythms [20]. | Hemodynamic responses in motor cortex during execution, observation, and imagery [20]. | Detailed mapping of motor network plasticity [2]. |
| Cognitive Studies | Event-related potentials (ERPs) for sensory and decision processes [55]. | Prefrontal cortex activation during sustained attention and problem-solving [55]. | Whole-brain activation patterns during complex cognitive tasks [2]. |
Integrated Experimental Approaches
Multimodal Integration Methodologies
Recognizing that no single modality perfectly captures the brain's complexity, researchers increasingly combine EEG, fNIRS, and fMRI in multimodal approaches. These integrated designs leverage the complementary strengths of each technique, with high-temporal resolution modalities (EEG) informing the analysis of high-spatial resolution modalities (fMRI, fNIRS) [2] [56]. Two primary integration methods have emerged: synchronous and asynchronous detection modes. Synchronous acquisition uses hardware synchronization to simultaneously collect data from multiple modalities, while asynchronous approaches combine data collected in separate sessions through co-registration and normalization [2].
A prominent example is the simultaneous recording of fNIRS and EEG, which has been successfully implemented in studies of motor execution, observation, and imagery [20]. In these experiments, researchers used a 24-channel fNIRS system embedded within a 128-electrode EEG cap, with optodes placed over sensorimotor and parietal cortices to index action observation network activity [20]. The integration of these complementary data streams enables researchers to correlate electrical and hemodynamic activities, providing a more complete picture of neural dynamics than either modality could offer alone.
Experimental Workflow for Multimodal Studies
The following diagram illustrates a typical workflow for a simultaneous fNIRS-EEG study, demonstrating how these modalities are integrated in practice:
Figure 2: Experimental Workflow for Simultaneous fNIRS-EEG Studies. This workflow demonstrates the process from study design to integrated interpretation, highlighting key stages including hardware setup with integrated caps, synchronized data recording, separate preprocessing pipelines, and multimodal analysis techniques [55] [56] [20].
For studies integrating fMRI with fNIRS or EEG, additional considerations include managing electromagnetic interference in the MRI environment and addressing the limited movement tolerance of fMRI [2]. Despite these challenges, simultaneous fMRI-fNIRS recordings have been successfully implemented, allowing researchers to leverage fMRI's whole-brain coverage to compensate for fNIRS's limited penetration depth [2].
The Scientist's Toolkit: Essential Research Reagents and Materials
Implementing EEG, fNIRS, and fMRI research requires specialized equipment and software. The following table details essential solutions for researchers designing neuroimaging studies:
Table 3: Essential Research Solutions for Neuroimaging Studies
| Tool Category | Specific Examples | Function & Application |
|---|---|---|
| fNIRS Systems | Continuous-wave systems (e.g., NIRScout) [56] | Measure relative changes in HbO and HbR concentrations using constant light intensity. |
| EEG Systems | Amplifier systems (e.g., BrainAMP) [58] | Record electrical brain activity with high temporal resolution. |
| Integrated Systems | Combined fNIRS-EEG caps [58] [20] | Enable simultaneous acquisition of hemodynamic and electrical signals. |
| Co-registration Tools | 3D magnetic space digitizers (e.g., Fastrak) [20] | Precisely localize fNIRS optodes and EEG electrodes relative to head landmarks. |
| Analysis Software | AtlasViewer, Homer2, NIRS-KIT [3] | Process fNIRS signals, map channels to brain regions, and perform statistical analyses. |
| Multimodal Fusion Algorithms | Structured Sparse Multiset CCA (ssmCCA) [20] | Identify brain regions consistently activated across fNIRS and EEG modalities. |
| Experimental Paradigms | Verbal Fluency Task, Motor Execution/Observation/Imagery [59] [20] | Standardized protocols to elicit neural responses in specific cognitive domains. |
The comparative analysis of EEG, fNIRS, and fMRI reveals a clear trade-off between temporal and spatial resolution that fundamentally influences their application in epilepsy diagnosis and therapy response monitoring. EEG remains unsurpassed for capturing rapid neural dynamics with millisecond precision, making it indispensable for epilepsy diagnosis where timing is critical. fNIRS offers a favorable balance of moderate temporal resolution, improved spatial localization compared to EEG, and practical advantages of portability and motion tolerance, positioning it as a valuable tool for therapy monitoring in naturalistic settings. fMRI provides the gold standard for spatial localization but is limited by its slow temporal resolution. Rather than viewing these modalities as competitors, researchers should consider them as complementary tools that, when integrated through multimodal designs, can provide a more comprehensive understanding of brain function in health and disease. The future of neuroimaging lies not in identifying a single superior technology, but in strategically combining these complementary modalities to overcome their individual limitations and leverage their unique strengths.
Navigating Limitations and Enhancing Data Quality in Temporal Analysis
This guide objectively compares the performance of electroencephalography (EEG), functional near-infrared spectroscopy (fNIRS), and functional magnetic resonance imaging (fMRI) in managing common artifacts and confounds, framed within the critical thesis of temporal resolution in neuroimaging.
The selection of a neuroimaging modality is a fundamental decision in experimental design, heavily influenced by the trade-offs between temporal and spatial resolution. EEG measures electrical activity from populations of cortical neurons with a millisecond-scale temporal resolution, capturing the brain's rapid dynamics directly [60] [56]. In contrast, fNIRS and fMRI measure the slow hemodynamic response (changes in blood oxygenation), an indirect marker of neural activity, with resolutions on the scale of seconds [60] [3]. While fNIRS and fMRI provide better spatial localization than EEG, their inherent temporal lag, dictated by neurovascular coupling, makes them blind to the fastest neural processes [56]. This hierarchy of temporal fidelity directly impacts each modality's susceptibility and response to various artifacts, which are the focus of this comparison.
Table 1: Susceptibility of EEG, fNIRS, and fMRI to Common Artifacts and Confounds
| Artifact/Confound Type | EEG (High Temporal Resolution) | fNIRS (Moderate Temporal Resolution) | fMRI (Low Temporal Resolution) |
|---|---|---|---|
| Motion Artifacts | High susceptibility; causes large signal spikes and shifts [60] [56]. | Moderate tolerance; more robust to movement than EEG [60] [61]. | Very high susceptibility; requires complete stillness [3]. |
| Physiological Confounds (Cardiac, Respiration) | Significant high-frequency confounds; can be filtered [29]. | Significant low-frequency confounds; correlates with hemodynamic noise [29]. | Significant confounds (BOLD signal); requires specific filtering models. |
| Environmental Noise | Highly sensitive to electromagnetic interference [62]. | Largely insensitive to electromagnetic fields [62]. | Highly sensitive to metallic objects; generates loud acoustic noise [3]. |
| Spatial Confounds (Scalp Blood Flow) | Not a primary confound for electrical signals. | Major confound; requires short-separation channels to regress out [62] [29]. | Less of a confound; global signal can be regressed. |
| Typical Motion Correction Methods | Filtering, blind source separation (e.g., ICA) [56]. | Wavelet, moving average, spline interpolation [63]. | Volume realignment, prospective motion correction. |
Motion Artifacts: Mechanisms and Correction Protocols
Motion artifacts present a fundamental challenge in neuroimaging, but their nature and impact vary dramatically across modalities due to differences in the underlying signal being measured.
EEG and Motion
EEG's exceptional sensitivity to motion stems from its measurement of microvolt-scale electrical potentials. Head movements can cause electrode-skin impedance changes, muscle activation generates electromyographic (EMG) noise, and even eye movements create large ocular artifacts that overwhelm cortical signals [56]. This makes EEG poorly suited for highly mobile or naturalistic studies without sophisticated correction.
Experimental Protocol: Motion Correction in Pediatric fNIRS Motion is a predominant challenge in pediatric populations. A systematic study compared six software-based motion correction techniques on fNIRS data from children (ages 6-9.9 years) performing a language task [63].
- Methodology: fNIRS data was collected over the left inferior frontal gyrus. Artifacts were categorized into four types: brief spikes (Type A), longer peaks (Type B), gentle slopes (Type C), and slow baseline shifts (Type D).
- Corrective Workflow: The following diagram illustrates the processing and motion correction pipeline for such a dataset.
Figure 1: fNIRS motion artifact correction workflow.
- Key Findings: The evaluation revealed that the Moving Average (MA) and Wavelet-based methods yielded the best outcomes for retaining signal quality in pediatric data, which is often noisier than adult data [63].
fNIRS and Motion
fNIRS is more tolerant of movement than EEG, but motion remains a significant noise source. Artifacts typically manifest as abrupt shifts in the light intensity signal when an optode is temporarily displaced from the scalp, changing the light coupling [63].
fMRI and Motion
fMRI is exquisitely sensitive to motion because movement alters the magnetic field homogeneity. Even millimeter-scale head movements can cause spin history effects and misalignment between image volumes, leading to severe reconstruction artifacts. This necessitates extreme subject compliance and limits its use in populations like infants and children [3].
Physiological Confounds: Inherent Biological Noise
Physiological processes create rhythmic, pervasive noise that can be confounded with neural signals of interest.
- EEG: The electrical activity of the heart (ECG) and scalp muscles (EMG) are major confounds. Blinks and eye movements generate large electrical fields that propagate to frontal EEG electrodes. These are often removed using techniques like Independent Component Analysis (ICA) [56].
- fNIRS: The hemodynamic signal is contaminated by systemic blood pressure oscillations (∼1 Hz) from the cardiac cycle, respiratory cycles (∼0.3 Hz), and very low-frequency Mayer waves (∼0.1 Hz) related to blood pressure regulation [29]. These signals are spectrally overlapping with the task-evoked hemodynamic response, making them a fundamental confound.
- fMRI: Similar to fNIRS, fMRI's BOLD signal is contaminated by cardiac and respiratory cycles. These are typically addressed using recording of physiological waveforms and subsequent modeling and removal during preprocessing.
Environmental Noise and Experimental Constraints
The physical environment imposes distinct constraints on each modality.
- EEG is highly susceptible to electromagnetic interference from power lines (50/60 Hz), lighting, and other electronic equipment, requiring shielded rooms often [62].
- fNIRS, as an optical technique, is largely immune to electromagnetic interference. This allows it to be used in a wide range of real-world environments, including alongside other equipment like hearing aids, which would cripple EEG recordings [62].
- fMRI has the most restrictive environment. The high-strength static magnetic field prohibits most metallic objects. Furthermore, the intense acoustic noise (over 100 dB SPL) generated by gradient coils during scanning is a profound environmental confound for any auditory or resting-state study and can cause patient discomfort [3].
The Multimodal Approach: Integrating EEG and fNIRS
To overcome the limitations of any single modality, a powerful strategy is the simultaneous acquisition of EEG and fNIRS [56] [20]. This approach harnesses their complementary strengths: EEG provides the millisecond temporal dynamics of neural processing, while fNIRS provides the improved spatial localization of the associated hemodynamic response [60] [56].
Experimental Protocol: Investigating the Action Observation Network A 2023 study used simultaneous fNIRS-EEG to elucidate neural activity during motor execution, observation, and imagery [20].
- Methodology: 21 participants were fitted with a 128-electrode EEG cap integrated with a 24-channel fNIRS system. In a live-action paradigm, participants performed, observed, or imagined a cup-moving task.
- Data Fusion Analysis: The study employed structured sparse multiset Canonical Correlation Analysis (ssmCCA) to fuse the electrical (EEG) and hemodynamic (fNIRS) data. This advanced statistical technique identifies components that are maximally correlated across the two modalities, pinpointing brain regions consistently active in both.
- Key Findings: Unimodal analyses showed differentiated activation but with incomplete overlap in the regions identified by fNIRS and EEG. However, the fused ssmCCA analysis consistently identified a shared neural signature in the left inferior parietal lobe across all three conditions, a key node of the Action Observation Network that was more robustly characterized by the multimodal approach [20].
Figure 2: Multimodal EEG-fNIRS data fusion workflow.
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 2: Key Materials and Solutions for EEG-fNIRS Research
| Item / Solution | Function / Purpose | Considerations |
|---|---|---|
| Conductive EEG Gel / Paste | Reduces impedance between scalp and electrode for high-quality electrical signal acquisition. | Gel typically offers lower impedance; paste is easier for cleanup. Crucial for signal-to-noise ratio. |
| fNIRS Optode Holders / Probe Caps | Securely positions light sources and detectors on the scalp at fixed distances. | Inter-optode distance (e.g., 3 cm) determines penetration depth. Custom caps ensure consistent placement across subjects [20]. |
| 3D Digitizer | Records the precise 3D locations of EEG electrodes and fNIRS optodes relative to scalp landmarks (nasion, inion). | Essential for coregistering measurement channels to an anatomical brain atlas for accurate source modeling [29]. |
| Short-Separation fNIRS Channels | Detectors placed very close (e.g., 8 mm) to a source to measure systemic physiological noise from the scalp. | Used as a regressor in analysis to separate cortical fNIRS signals from superficial confounds [62]. |
| Synchronization Trigger Box | Generates a common electrical pulse (TTL) to mark event onsets in both EEG and fNIRS data streams. | Vital for temporal alignment of data from separate acquisition systems in a multimodal setup [56] [6]. |
In cognitive neuroscience and drug development, the integrity of neural data is paramount. Advanced preprocessing pipelines serve as the critical foundation for cleaning and refining raw brain signals, directly impacting the validity of subsequent analysis and interpretation. The choice of preprocessing technique is intrinsically linked to the fundamental characteristics of the neuroimaging modality employed, particularly its temporal resolution. Electroencephalography (EEG) and functional Near-Infrared Spectroscopy (fNIRS) represent two widely used non-invasive methods that operate on different temporal and physiological scales [64]. EEG measures the brain's electrical activity with millisecond precision, capturing direct neural firing, while fNIRS monitors hemodynamic responses through changes in blood oxygenation, a slower process measured on a scale of seconds [64]. This comparison is often framed within a broader context that includes functional Magnetic Resonance Imaging (fMRI), which also measures hemodynamics but with higher spatial resolution and lower portability than fNIRS [3]. This guide provides an objective comparison of advanced preprocessing techniques for EEG and fNIRS, detailing specialized methodologies for handling motion artifacts, physiological noise, and other contaminants to ensure data quality for research and clinical applications.
Core Modality Comparison: EEG vs. fNIRS
Understanding the inherent strengths and limitations of EEG and fNIRS is essential for selecting appropriate preprocessing strategies. The following table outlines their core characteristics.
Table 1: Fundamental Comparison of EEG and fNIRS Neuroimaging Modalities
| Feature | EEG (Electroencephalography) | fNIRS (Functional Near-Infrared Spectroscopy) |
|---|---|---|
| What It Measures | Electrical activity from postsynaptic potentials in cortical neurons [64] | Hemodynamic response (changes in oxygenated and deoxygenated hemoglobin) [64] |
| Temporal Resolution | Very High (milliseconds) [64] | Moderate (seconds) [64] |
| Spatial Resolution | Low (centimeter-level) [64] | Moderate (better than EEG, but limited to cortical surface) [64] |
| Primary Preprocessing Challenges | Motion artifacts, ocular and muscle activity, line noise [65] [16] [66] | Systemic physiological noise (cardiac, respiratory), motion artifacts, low signal-to-noise ratio [67] [68] [69] |
| Ideal Application Context | Fast cognitive tasks, event-related potentials (ERPs), brain-computer interfaces (BCIs) [64] [65] | Sustained cognitive states, naturalistic studies, populations prone to movement (infants, patients) [64] [67] |
The physiological basis of the signals dictates the nature of the artifacts and the corresponding correction approaches. EEG's exceptional temporal resolution makes it ideal for tracking fast neural dynamics but also renders it highly susceptible to electrical and motion-based interference [64] [65]. In contrast, fNIRS is more tolerant of movement but is profoundly contaminated by systemic physiological noise from the scalp and skull, which can mimic or obscure the true cerebral hemodynamic response [69].
Advanced fNIRS Preprocessing Pipelines
Tackling Physiological Noise and Motion in fNIRS
The fNIRS signal is a composite of cerebral activity and confounding noise from physiological processes (e.g., heart rate, blood pressure, respiration) and subject motion. Effective preprocessing is critical to isolate the brain's functional response.
Table 2: Comparison of Advanced fNIRS Preprocessing Techniques for Noise Removal
| Method | Core Principle | Key Experimental Findings | Performance Data |
|---|---|---|---|
| Short-Separation Regression | Uses signals from short source-detector distances (~8 mm) to regress out superficial scalp hemodynamics [69] | Receiver Operating Characteristic (ROC) simulation on experimental breath-hold data found it the best-performing method [69] | Highest sensitivity and specificity (Area Under Curve, AUC). Performance improved using all available short-separation channels [69] |
| Baseline-Derived PCA (bPCA) | Calculates principal components from a resting-state baseline and removes them from task data [69] | Effectively reduces global physiological noise; performance is inferior to short-separation regression but a good alternative when short channels are unavailable [69] | Removes components explaining up to 80% of spatial covariance from baseline [69] |
| Cumulative Curve Fitting Approximation (CCFA) | A non-stationary filtering procedure for detrending and removing high-frequency noise [68] | Comparison with DCT and Band-Pass Filtering (BPF) on synthetic and real data demonstrated a higher Signal-to-Noise Ratio (SNR) [68] | Greater SNR improvement than conventional Discrete Cosine Transform (DCT) and BPF methods [68] |
| Wavelet-Based Motion Correction | Uses wavelet transforms to identify and correct transient motion artifacts in the signal [67] | In infant data, it retains a larger number of trials but may reduce the amplitude of the recovered Hemodynamic Response Function (HRF) [67] | Performs well with moderately noisy datasets; more motion-tolerant than EEG [64] [67] |
Experimental Protocol: fNIRS Noise Removal
A seminal study by [69] provides a robust experimental framework for comparing fNIRS noise correction techniques:
- Data Acquisition: Experimental background physiological signals were recorded during a resting-state scan and a breath-hold task, which induces systemic physiological changes.
- Signal Simulation: To enable quantitative performance evaluation, synthetic "brain" responses at varying amplitudes were added to the real physiological data. This created a ground truth for comparison.
- Pipeline Application: Multiple analysis pipelines were applied, including short-separation regression, bPCA, standard PCA, and various filtering methods.
- Performance Quantification: Receiver Operating Characteristic (ROC) analysis was used to compute the sensitivity and specificity of each method in detecting the known synthetic brain signal. The Area Under the Curve (AUC) of the ROC served as the primary performance metric [69].
The workflow for evaluating these methods, from data preparation to performance assessment, can be visualized as follows:
Figure 1: fNIRS Method Evaluation Workflow. This diagram outlines the process for quantitatively comparing the performance of different fNIRS preprocessing techniques, as implemented in [69].
Advanced EEG Preprocessing Pipelines
Motion and Artifact Correction in EEG
EEG preprocessing confronts a wide array of artifacts, with motion and ocular movements posing significant challenges, especially in mobile or developmental studies. The following table compares modern correction methods.
Table 3: Comparison of Advanced EEG Preprocessing Techniques for Motion and Artifact Correction
| Method | Core Principle | Key Experimental Findings | Impact on Decoding |
|---|---|---|---|
| iCanClean | Uses Canonical Correlation Analysis (CCA) with dual-layer or pseudo-reference noise signals to identify and subtract motion-correlated subspaces [65] | In human running data, it effectively reduced gait-frequency power, recovered dipolar brain ICs, and enabled P300 ERP component identification [65] | N/A (Method focused on signal fidelity) |
| Artifact Subspace Reconstruction (ASR) | Uses a sliding-window PCA to identify and remove high-variance components exceeding a threshold ("k") based on a clean calibration period [65] | Improved ICA decomposition quality during running; a "k" parameter that is too low can "overclean" and distort neural signals [65] | N/A (Method focused on signal fidelity) |
| Independent Component Analysis (ICA) | Blind source separation that decomposes data into maximally independent components, which are then classified and removed as artifacts [66] | In infant EEG, ICA demonstrated higher sensitivity (better artifact removal) but lower specificity (more distortion of clean neural signals) [66] | Generally decreases decoding performance by removing signal predictive of the task condition [70] |
| Artifact Blocking (AB) | An alternative to ICA that uses spatial filtering to block the transmission of artifacts from specific channels [66] | In infant EEG, AB demonstrated lower sensitivity but higher specificity, causing less distortion to clean neural segments than ICA [66] | N/A (Method focused on signal fidelity) |
Experimental Protocol: EEG during Human Locomotion
A 2025 study [65] established a rigorous protocol for evaluating motion artifact removal during ecologically valid tasks like running:
- Task Design: Participants performed a Flanker task (a cognitive conflict paradigm) under two conditions: while jogging on a treadmill and while standing still. The static condition provided a motion-free baseline.
- Data Acquisition & Preprocessing: EEG was recorded during both tasks. The data were then preprocessed using different methods: iCanClean (with pseudo-reference signals) and ASR (with an aggressive "k" parameter of 3).
- Performance Evaluation: The effectiveness of each preprocessing method was assessed based on multiple metrics:
- ICA Component Dipolarity: The number of brain-like independent components recovered.
- Spectral Power: Reduction in power at the gait frequency and its harmonics.
- Event-Related Potential (ERP) Recovery: The ability to capture the expected P300 ERP component and its "congruency effect" (amplitude difference between congruent and incongruent Flanker stimuli) during running compared to standing [65].
The following diagram illustrates the logical relationship between the preprocessing methods and the metrics used for their validation:
Figure 2: EEG Method Validation Logic. This diagram shows the key metrics used to validate the success of advanced EEG preprocessing pipelines, particularly in motion-heavy contexts [65].
The Impact of Preprocessing on Decoding Performance
Beyond cleaning the signal, preprocessing choices profoundly impact the performance of machine learning models in decoding neural states. A large-scale multiverse analysis of seven EEG experiments revealed that preprocessing steps can dramatically shape decoding outcomes [70].
- Artifact Correction: Steps like ICA and the use of the Autoreject package generally decreased decoding performance across experiments. This counterintuitive result suggests that artifacts can be systematically correlated with task conditions, providing the decoder with predictive (but non-neural) information. Removing them reduces this spurious accuracy, which is desirable for valid neuro-scientific interpretation [70].
- Filtering: Using a higher high-pass filter (HPF) cutoff consistently increased decoding performance. This is likely because it removes slow drifts that are not informative for the classification task. For time-resolved decoding, a lower low-pass filter (LPF) cutoff was also beneficial, as it reduces high-frequency noise [70].
- Baseline Correction & Detrending: These steps also increased decoding performance, likely by normalizing trials and removing non-stationary trends, thereby helping the model focus on task-relevant signal changes [70].
The Scientist's Toolkit: Key Research Reagents and Solutions
The implementation of advanced preprocessing pipelines often relies on specialized software tools and methodological components.
Table 4: Essential Reagents and Computational Tools for Advanced Preprocessing
| Tool / Solution | Function | Application Context |
|---|---|---|
| Short-Separation fNIRS Channels | Optodes placed 8-15 mm from a source to measure systemic physiological noise in the scalp [69] | Critical regressor for General Linear Models (GLM) to separate cerebral from extra-cerebral hemodynamic signals [69] |
| Dual-Layer EEG Electrodes | Specialized sensors with a dedicated noise-capture layer mechanically coupled to the scalp electrode [65] | Provides a pure noise reference for iCanClean processing, dramatically improving motion artifact removal in mobile EEG [65] |
| iCanClean Algorithm | A signal processing routine using Canonical Correlation Analysis (CCA) to remove noise subspaces [65] | Method of choice for cleaning EEG data during high-motion studies (e.g., walking, running) when dual-layer electrodes are used [65] |
| Artifact Subspace Reconstruction (ASR) | An adaptive, data-driven method for removing high-amplitude artifacts from continuous EEG [65] | Effective for cleaning mobile EEG; performance depends critically on the calibration data and the "k" threshold parameter [65] |
| HAPPE Pipeline | The Harvard Automated Processing Pipeline for Electroencephalography, includes wavelet-enhanced ICA [66] | Standardized automated pipeline for EEG preprocessing, designed to be effective with shorter data segments, as in infant studies [66] |
| MADE Pipeline | The Maryland Analysis of Developmental EEG pipeline [66] | A standardized automated pipeline optimized for developmental (infant) EEG data, incorporating EOG thresholding and robust referencing [66] |
The selection of an advanced preprocessing pipeline is a decisive step in neuroimaging research that must be aligned with the specific modality, the experimental paradigm, and the target population. For fNIRS, methods leveraging short-separation channels as regressors currently provide the highest sensitivity and specificity in removing confounding physiological noise [69]. For EEG in mobile or high-motion contexts, iCanClean has demonstrated superior performance in recovering neural signals, though ASR remains a viable option when parameters are carefully tuned [65]. It is crucial to recognize that preprocessing choices are not merely technical—they directly influence analytical outcomes, as evidenced by their significant impact on decoding performance [70]. Researchers must therefore justify their selected pipelines based on empirical comparisons and a clear understanding of the trade-offs between signal preservation and artifact removal.
The human brain operates across multiple temporal and spatial scales, making no single neuroimaging modality sufficient to capture its full complexity. Multimodal integration is the simultaneous or combined use of different neuroimaging techniques to leverage their complementary strengths, thereby providing a more comprehensive understanding of brain function [71]. This approach is particularly valuable for investigating the relationship between neural electrical activity and the subsequent hemodynamic responses that supply energy to active neurons. Electroencephalography (EEG), functional near-infrared spectroscopy (fNIRS), and functional magnetic resonance imaging (fMRI) represent three cornerstone techniques in non-invasive human brain imaging, each offering unique windows into brain function [71] [56] [72].
The theoretical basis for combining these modalities lies in the phenomenon of neurovascular coupling, the intimate relationship between neural activity and cerebral blood flow [56]. When neurons become active, they trigger a complex cascade of events that ultimately increases local blood flow to deliver oxygen and glucose, a process measured as the hemodynamic response. EEG captures the direct, fast electrical consequences of neural signaling—primarily postsynaptic potentials from synchronized assemblies of pyramidal neurons [56] [73]. In contrast, fNIRS and fMRI measure the indirect, slower hemodynamic consequences of this activity: fNIRS detects changes in oxygenated and deoxygenated hemoglobin concentrations [56] [74], while fMRI measures the blood-oxygenation-level-dependent (BOLD) signal [72]. By designing experimental paradigms that simultaneously capture both electrical and hemodynamic signals, researchers can investigate the complete sequence of neural events from initial activation to metabolic supply.
Technical Comparison of Neuroimaging Modalities
Each major neuroimaging modality offers a distinct balance of temporal resolution (ability to track rapid changes over time) and spatial resolution (ability to pinpoint the location of activity). Understanding these fundamental characteristics is essential for selecting the appropriate tool for specific research questions and for designing effective multimodal experiments.
Table 1: Technical Specifications of EEG, fNIRS, and fMRI
| Feature | EEG | fNIRS | fMRI |
|---|---|---|---|
| What It Measures | Electrical activity from postsynaptic potentials [56] [72] | Concentration changes in oxygenated (HbO) & deoxygenated hemoglobin (HbR) [56] [74] | Blood-Oxygenation-Level Dependent (BOLD) signal [72] |
| Temporal Resolution | Very High (milliseconds) [71] [73] | Low (seconds) [56] [73] | Low (seconds); Effective Temporal Resolution (ETR) depends on CNR and parameters [75] |
| Spatial Resolution | Low (centimeter-level) [71] [73] | Moderate (1-3 cm), limited to cortex [74] | High (millimeters), can image deeper structures [71] [72] |
| Depth of Measurement | Cortical surface [73] | Outer cortex (1-2.5 cm deep) [73] [74] | Whole brain |
| Key Strength | Capturing rapid neural dynamics, event-related potentials [73] | Good spatial resolution for cortex, motion-tolerant, portable [56] [74] | Detailed whole-brain mapping, excellent spatial resolution [71] [72] |
| Primary Limitation | Poor spatial resolution, sensitive to motion artifacts [71] [73] | Limited penetration depth, slow hemodynamic response [56] [73] | Low temporal resolution, expensive, not portable [72] [75] |
The comparative analysis reveals a clear complementarity. EEG's millisecond-scale temporal resolution is ideal for tracking the fast dynamics of brain networks, such as those involved in motor and working memory tasks [76]. However, its spatial resolution is poor due to the blurring effect of the skull and scalp [71]. Conversely, fMRI provides high spatial resolution, allowing for precise anatomical localization of active brain regions, but its temporal resolution is limited by the sluggish nature of the hemodynamic response, typically on the order of seconds [72] [75]. fNIRS occupies a middle ground, offering better spatial resolution than EEG for cortical areas and greater portability and motion tolerance than fMRI, though it cannot probe deep brain structures [73] [74].
A critical concept for fMRI is the Effective Temporal Resolution (ETR). While the sampling rate (Repetition Time, TR) is often cited as its temporal resolution, the actual ability to discern the timing of neural events is influenced by the contrast-to-noise ratio (CNR) and other scanning parameters. Research using dynamic phantoms has shown that with optimized multi-echo sequences at ultra-high field (7T), ETR can be as low as 151 ms for cortex-mimicking setups, demonstrating that under ideal conditions, fMRI can achieve better temporal precision than traditionally assumed, though this is highly dependent on specific experimental parameters [75].
Task Paradigm Design for Multimodal Experiments
Designing effective task paradigms for simultaneous multimodal recording requires careful consideration of the physiological and temporal characteristics of each signal. The goal is to create a protocol that robustly engages the cognitive or neural process of interest while being compatible with the technical constraints of all modalities involved.
Fundamental Principles of Paradigm Design
The most important principle is accounting for the differential latency of the signals. An EEG response to a stimulus is virtually instantaneous, while the corresponding hemodynamic response measured by fNIRS or fMRI peaks 4-6 seconds after stimulus onset [75]. This has direct implications for trial structure and inter-stimulus interval (ISI). For block designs, periods of task engagement ("ON" blocks) must be long enough—typically 20-30 seconds—for the hemodynamic response to robustly rise and fall, allowing for a stable baseline [77] [75]. For event-related designs, the ISI must be jittered and sufficiently long to allow the hemodynamic response to return to baseline before the next trial, avoiding overlapping responses that are difficult to disentangle [72].
Another key principle is signal provenance. EEG is highly susceptible to artifacts from muscle activity (e.g., jaw clenching, talking) and eye movements [73]. fNIRS, while more motion-tolerant, can be affected by systemic physiological noise such as blood pressure fluctuations and heart rate [56] [74]. Therefore, tasks should be designed to minimize confounding movements. For example, "silent naming" paradigms can be used to study language processing without generating speech-related muscle artifacts in EEG [77]. Furthermore, tasks should be selected to target brain regions accessible to all modalities; for instance, prefrontal cortex tasks are well-suited for EEG-fNIRS studies due to fNIRS's sensitivity to cortical surfaces [74].
Exemplar Experimental Protocols
Motor Execution Task (for EEG-fNIRS): This is a classic paradigm for validating hybrid systems. Participants perform repetitive self-paced finger or thumb tapping [78].
- Protocol: A block design consists of 20-second blocks of rest alternating with 20-second blocks of motor activity (e.g., right thumb tapping). The task should be simple and reproducible.
- Measured Signals: EEG is used to detect the event-related desynchronization (ERD) in the mu/beta rhythms over the contralateral motor cortex, providing a precise electrical signature of motor planning and execution with high temporal resolution. Concurrently, fNIRS measures the increase in oxygenated hemoglobin (HbO) and decrease in deoxygenated hemoglobin (HbR) in the same motor cortex area, reflecting the hemodynamic response to the neural activity [78].
- Rationale: This combination allows researchers to study the neurovascular coupling mechanism directly. The EEG signal pinpoints the exact timing of neural activation, while fNIRS provides better spatial localization of the active motor region than EEG alone.
Working Memory N-Back Task (for EEG-fNIRS/fMRI): This paradigm is excellent for investigating higher-order cognition and is widely used in clinical populations, including schizophrenia research [74].
- Protocol: Participants are shown a sequence of stimuli (e.g., letters) and must indicate whether the current stimulus matches the one presented 'n' trials back. A common block design alternates between a control condition (0-back) and a high-load condition (2-back or 3-back), with each block lasting 20-30 seconds.
- Measured Signals: EEG captures theta (4-7 Hz) and gamma (>25 Hz) band power changes, which are associated with working memory maintenance and processing, as well as the P300 event-related potential related to context updating. fNIRS or fMRI simultaneously records hemodynamic activity, typically revealing increased activation in the dorsolateral prefrontal cortex (DLPFC) and parietal areas during the high-load condition [74].
- Rationale: The combination reveals both the fast oscillatory dynamics of memory operations (EEG) and the sustained, localized cortical engagement (fNIRS/fMRI). In clinical settings, this can identify specific deficits, such as reduced prefrontal HbO in patients, which may serve as a biomarker [74].
Picture Naming Task (for MEG-fMRI): While involving MEG instead of EEG, this paradigm from the search results illustrates the powerful fusion of temporal and spatial data to dissociate cognitive processes [77].
- Protocol: Participants silently name objects or actions from presented line drawings. The experiment uses a blocked design with different task conditions, such as naming objects from object images, naming actions from action images, and naming objects from action images [77].
- Measured Signals: MEG provides millisecond-resolution data on the sequence of neural activation from visual processing to speech preparation. fMRI provides precise anatomical localization of the brain networks involved in each task condition.
- Rationale: By examining the correlation patterns between MEG and fMRI signals across different tasks, researchers can identify how neural engagement varies with cognitive demand. This approach can reveal task-dependent neural engagement that might not be detectable using either modality in isolation [77].
Advanced Data Integration and Analysis Methods
The true power of multimodal imaging is realized through sophisticated data integration techniques that transform parallel data streams into a unified model of brain function. Analysis strategies can be broadly categorized into three main approaches.
1. Parallel Analysis: This is the most straightforward approach, where EEG and hemodynamic (fNIRS/fMRI) data are processed and analyzed independently using modality-specific pipelines [56]. The results are then compared or interpreted together at the group or results level. For instance, one might examine whether the source-localized EEG activity in the prefrontal cortex during a decision-making task co-occurs with the significant fNIRS-based HbO increases in the same region. This approach is methodologically simpler but may miss more nuanced interactions between the electrical and hemodynamic signals.
2. Asymmetric (Informed) Analysis: This is a more powerful approach where data from one modality is used to inform or constrain the analysis of the other. A prominent example is EEG-informed fMRI analysis, where specific EEG features (e.g., the power of a particular oscillation or the amplitude of an ERP component) are extracted and used as regressors in the general linear model (GLM) analysis of the fMRI data [56] [77]. This allows researchers to identify brain regions whose BOLD signal fluctuations are directly correlated with variations in specific electrophysiological phenomena. Conversely, fMRI data can be used to spatially constrain EEG source localization algorithms, significantly improving the accuracy of pinpointing the origins of electrical brain activity [71] [77].
3. Fusion-Based Analysis: The most integrated approach involves jointly decomposing the data from both modalities to find common underlying components. Techniques such as Joint Independent Component Analysis (jICA) or Canonical Correlation Analysis (CCA) are used to identify coupled patterns of electrical and hemodynamic activity [56] [73]. For example, a study might use jICA to find a component that simultaneously expresses a specific EEG theta rhythm and a specific pattern of prefrontal fNIRS activation during a working memory task. This component would represent a unified "neurovascular signature" of the cognitive process. A benchmarking study highlights that source separation methods like ICA are particularly promising for tracking the fast reconfiguration of electrophysiological brain networks during tasks [76].
Table 2: Key Reagents and Computational Tools for Multimodal Research
| Category | Item | Primary Function |
|---|---|---|
| Hardware & Acquisition | MR-Compatible EEG System [71] | Allows safe and simultaneous recording of EEG inside the MRI scanner, designed to minimize interference. |
| Integrated EEG-fNIRS Cap [73] | Ensures precise and stable co-registration of EEG electrodes and fNIRS optodes on the scalp according to the 10-20 system. | |
| Synchronization Trigger Box [73] | Sends TTL pulses to all recording devices to align data streams from different modalities with millisecond precision. | |
| Software & Algorithms | Advanced Source Separation (e.g., ICA) [76] | Decomposes mixed signals from EEG/MEG to identify underlying brain and artifact sources. |
| Joint ICA (jICA) Toolboxes [73] | Statistically identifies components that are mutually independent across two different types of data (e.g., EEG and fNIRS). | |
| Boundary Element Method (BEM) / Finite Element Method (FEM) [71] | Creates realistic head models from structural MRI to solve the forward problem for accurate EEG source localization. | |
| Motion Correction Algorithms (for fNIRS) [74] | Removes motion artifacts from fNIRS signals using methods like wavelet decomposition. | |
| Experimental Materials | Dynamic Phantom ("Brain Dancer") [75] | Provides a ground-truth physical model to validate and quantify the effective temporal resolution of an fMRI sequence. |
Optimizing experimental designs for simultaneous acquisition of hemodynamic and electrical signals is a critical frontier in cognitive neuroscience and neuropharmacology. The complementary nature of EEG, fNIRS, and fMRI provides an unprecedented opportunity to create spatiotemporally rich models of brain function, but harnessing this potential requires meticulous paradigm design. The key lies in respecting the distinct temporal dynamics and physiological origins of each signal—designing tasks with appropriate block lengths and jittered trials to disentangle the slow hemodynamic response, while minimizing artifacts for the sensitive electrical recordings.
The future of this field points toward deeper integration, moving beyond parallel recording to truly fused analysis. Machine learning approaches applied to hybrid feature sets, real-time analysis for neurofeedback and closed-loop neuromodulation, and the systematic use of physical phantoms to quantify and optimize system performance [75] will drive the next wave of innovations. For researchers and drug development professionals, mastering these multimodal principles is no longer optional but essential for developing sensitive biomarkers, evaluating targeted therapeutics, and ultimately achieving a more holistic and mechanistic understanding of the human brain in health and disease.
Strategies for Improving Signal-to-Noise Ratio in Each Modality
Improving the Signal-to-Noise Ratio (SNR) is a fundamental challenge in neuroimaging that directly impacts the quality and reliability of brain activity measurements. Within the context of comparing the temporal resolution of EEG, fNIRS, and fMRI, the strategies for SNR enhancement are uniquely tailored to the specific strengths and limitations of each modality. This guide objectively compares the experimental protocols and performance outcomes associated with state-of-the-art SNR improvement techniques across these three key non-invasive brain imaging technologies.
SNR Fundamentals and Modality Comparison
The signals of interest in neuroimaging are often obscured by noise, which can originate from physiological processes (e.g., heartbeat, respiration), subject motion, or the instrumentation itself. The chosen strategy for SNR improvement is heavily influenced by a modality's inherent temporal resolution.
- EEG, with its millisecond-scale temporal resolution, is ideal for tracking fast neural dynamics but is highly susceptible to electrical noise and motion artifacts [79] [49].
- fNIRS measures hemodynamic changes with a temporal resolution on the scale of seconds, offering a better balance, but its signals are contaminated by systemic physiological noise [79] [80].
- fMRI, also tracking the hemodynamic response, provides superior spatial resolution but has the slowest temporal response (on the scale of seconds) and is highly sensitive to motion [2] [3].
The table below summarizes the core SNR challenges and primary improvement strategies for each modality.
| Modality | Temporal Resolution | Primary SNR Challenges | Core SNR Improvement Strategies |
|---|---|---|---|
| EEG | High (Milliseconds) [79] | Low spatial resolution; high susceptibility to electrical and motion artifacts [79] [49] | Advanced multielectrode lead designs; hybrid attention modules in deep learning [81] [9] |
| fNIRS | Moderate (Seconds) [79] | Contamination by systemic physiological noise (cardiac, respiratory); motion artifacts [80] [82] | General Linear Model (GLM) with short-separation regression; spatial convolution across channels [80] [9] |
| fMRI | Slow (Seconds) [2] | Sensitivity to motion; physiological noise; arbitrary signal units [2] [83] | Multi-echo acquisition with advanced denoising (e.g., Total Variation minimization); T2* mapping [83] |
SNR Enhancement in Electroencephalography (EEG)
EEG's exceptional temporal resolution is counterbalanced by a low signal-to-noise ratio, necessitating sophisticated processing to extract meaningful neural patterns.
Experimental Protocol: Dual-Scale Spatiotemporal Feature Extraction
A prominent deep learning-based method for improving EEG SNR in Motor Imagery classification involves a structured pipeline to enhance spatiotemporal features [9].
- Signal Acquisition: EEG data is collected using a standard cap with electrodes placed according to the international 10-20 system.
- Temporal Feature Extraction: Raw EEG signals are processed using a dual-scale temporal convolution network. This architecture captures neural patterns across different temporal scales, isolating relevant signals from short- and long-duration noise.
- Spatial Feature Extraction: The features are then passed through a depthwise separable convolution layer. This step efficiently models the spatial relationships between different EEG electrodes, improving the system's ability to localize brain activity.
- Feature Enhancement: A hybrid attention module is applied to the features. This module automatically weights the importance of different temporal segments and spatial channels, making the network more sensitive to salient neural patterns related to the task (e.g., motor imagery) and less sensitive to irrelevant noise [9].
Performance Data
In a direct experimental test on a public dataset (TU-Berlin-A), this deep learning model achieved an average classification accuracy of 83.26% for motor imagery tasks, representing a 3.78% improvement over previous state-of-the-art methods [9]. This demonstrates a significant practical gain in signal decoding performance attributable to improved SNR handling.
SNR Enhancement in Functional Near-Infrared Spectroscopy (fNIRS)
fNIRS signals are plagued by noise from superficial tissues. A powerful method to address this is the use of the General Linear Model (GLM) with short-separation regression, adapted from established fMRI analysis.
Experimental Protocol: GLM with Short-Separation Regression
This protocol uses a processing pipeline that simultaneously models the brain signal and confounding noise, which is especially crucial for single-trial analysis in Brain-Computer Interfaces [80].
- Hardware Setup: A combined fNIRS system is used, featuring standard optodes to measure cortical activation and short-separation optodes (typically placed less than 1 cm from the source). These short-separation channels are predominantly sensitive to systemic physiological changes in the scalp.
- Data Preprocessing: The raw light intensity signals from all channels are converted into concentration changes of oxygenated (HbO) and deoxygenated hemoglobin (HbR).
- GLM Design Matrix Construction: A design matrix for the General Linear Model is created. It contains two key types of regressors:
- Task Regressor: This models the expected hemodynamic response (e.g., a canonical Hemodynamic Response Function aligned with the task timing).
- Nuisance Regressors: These include the signals from the short-separation channels, as well as other parameters like motion artifacts and physiological recordings (e.g., heart rate, respiration). These explicitly model the non-cerebral noise.
- Model Fitting and Signal Extraction: The GLM is fitted to the data from each standard channel. The weight (beta value) of the task regressor provides a clean, SNR-enhanced estimate of the task-evoked brain activity, as the variance explained by the nuisance regressors has been statistically removed [80].
GLM-based fNIRS denoising workflow.
Performance Data
A study using synthetic hemodynamic responses added to resting-state fNIRS data (providing ground truth) compared this GLM approach to conventional preprocessing. The results showed that features extracted from the GLM-cleaned signals led to a statistically significant average improvement of +7.4% in binary classification accuracy across subjects and feature types [80].
SNR Enhancement in Functional Magnetic Resonance Imaging (fMRI)
The qualitative nature and noise susceptibility of the standard fMRI BOLD signal drive the need for quantitative methods like multi-echo T2* mapping.
Experimental Protocol: Multi-Echo fMRI with Total Variation Denoising
This advanced protocol moves beyond qualitative BOLD analysis to produce quantitative, high-SNR T2* maps [83].
- Image Acquisition: Instead of a standard single-echo sequence, a multi-echo gradient-echo (GRE) sequence is used. This sequence acquires multiple echoes (e.g., 3) at different echo times (TEs) after a single radiofrequency excitation within each repetition time (TR).
- Denoising of Echo Time Series: The time series for each individual echo is denoised using an algorithm that minimizes total variation (TV). This state-of-the-art approach enforces temporal smoothness in the signal, based on the physiological property that the true BOLD signal changes smoothly over time, without altering its local mean.
- T2* Map Calculation: At every time point, the signal intensities across the denoised echoes are fitted to a mono-exponential decay model (S = S0 * exp(-TE / T2)) on a voxel-by-voxel basis. This generates a dynamic, time-varying T2 map.
- Quantitative Analysis: The resulting T2* time series, now in quantitative units (milliseconds), are used for subsequent analysis. This signal is more resistant to artifacts and provides a direct measure of blood oxygenation changes [83].
Performance Data
When compared to current standard denoising methods like 3dDespike, tedana, and NORDIC, the TV-minimizing algorithm applied to multi-echo data produced superior distributions of Signal-to-Noise (SNR) and Contrast-to-Noise (CNR) ratios. Crucially, the denoised echoes and calculated T2* time courses matched the shape of the theoretical hemodynamic response function much more closely than results from other methods [83].
The Scientist's Toolkit: Key Reagents & Materials
| Item Name | Function in SNR Improvement |
|---|---|
| Short-Separation fNIRS Optodes | Measures systemic physiological noise from the scalp, used as a nuisance regressor in the GLM to isolate cerebral brain activity [80]. |
| High-Density EEG Caps with fNIRS Openings | Enables precise, co-registered placement of electrodes and optodes for hybrid EEG-fNIRS studies, minimizing cross-modality interference and motion artifacts [79]. |
| Conductive Electrode Gel (e.g., NeuroPrep) | Applied to EEG electrodes to reduce impedance between the scalp and electrode, thereby improving signal quality and strength [49]. |
| Multi-Echo fMRI Pulse Sequence | A specialized MRI acquisition sequence that collects data at multiple echo times, enabling the quantitative calculation of T2* and improved BOLD contrast [83]. |
| 3D Digitization System (e.g., Polhemus) | Used to accurately record the 3D positions of EEG electrodes or fNIRS optodes on a subject's head, allowing for precise co-registration with anatomical MRI data and improved spatial specificity [82] [3]. |
Cross-Modality Integration for Superior SNR
A powerful trend in neuroimaging is the fusion of complementary modalities to overcome the limitations of any single technique. The integration of EEG and fNIRS is a prime example, synergistically combining electrophysiological and hemodynamic information [9] [6] [49].
In one advanced framework, EEG and fNIRS data are processed through separate, optimized deep learning networks to extract their respective spatiotemporal and hemodynamic features. The decisions from each unimodal network are then fused using the Dempster-Shafer Theory (DST). This method goes beyond simple averaging; it first quantifies the uncertainty associated with each modality's decision and then uses a two-layer reasoning process to combine the evidence, yielding a final, more robust classification [9]. This hybrid approach has been shown to outperform unimodal systems, providing a practical pathway to higher SNR and more reliable brain state decoding in real-world applications.
Understanding the human brain requires sophisticated neuroimaging technologies, each presenting a unique balance of benefits and limitations. Researchers and drug development professionals must navigate a critical trade-off: the choice between the high spatial resolution and precise control offered by traditional laboratory equipment and the ecological validity and accessibility provided by portable technologies. Ecological validity refers to the degree to which research findings reflect real-life functioning and experiences in natural environments [84] [85]. This comparative guide objectively analyzes three foundational neuroimaging techniques—functional magnetic resonance imaging (fMRI), electroencephalography (EEG), and functional near-infrared spectroscopy (fNIRS)—focusing on their temporal resolution, portability, and applicability in real-world research settings. As mobile neuroimaging technologies advance, they present new opportunities to study brain function directly in naturalistic learning and social environments, bridging the gap between laboratory-controlled conditions and authentic human experiences [86].
Neuroimaging Technology Comparison
The selection of neuroimaging technology significantly influences experimental design, participant population, and generalizability of findings. Below is a systematic comparison of the core technical specifications for fMRI, EEG, and fNIRS.
Table 1: Technical Specifications and Performance Comparison of Neuroimaging Technologies
| Feature | fMRI | EEG | fNIRS |
|---|---|---|---|
| Spatial Resolution | High (millimeter-level) [2] | Low (approximately 2 cm) [6] | Moderate (1-3 cm) [2] |
| Temporal Resolution | Low (0.33-2 Hz) [2] | High (millisecond-level) [6] | Moderate (up to 10+ Hz) [2] |
| Portability | Non-portable; requires fixed facility [2] | Highly portable systems available [87] | Portable; suitable for field studies [2] [88] |
| Primary Signal Measured | Blood Oxygen Level Dependent (BOLD) signal [2] | Electrical potentials from neuronal firing [87] | Concentration changes in oxygenated/deoxygenated hemoglobin [2] [88] |
| Key Strength | Whole-brain coverage, including subcortical structures [2] | Excellent for capturing rapid neural dynamics [6] | Good balance of spatial resolution and ecological validity [86] |
| Key Limitation | Highly sensitive to motion artifacts; restrictive environment [2] [89] | Susceptible to motion and environmental noise; poor spatial resolution [6] | Limited to cortical surface measurements (~2 cm depth) [2] |
Experimental Evidence: Lab vs. Real-World Performance
Data Quality in Community Settings
Recent studies directly compare traditional and portable methods, providing quantitative evidence for the viability of real-world neuroimaging.
Table 2: Experimental Data Comparing Laboratory and Community Recordings
| Experiment Focus | Methodology | Key Finding | Implication |
|---|---|---|---|
| EEG Data Quality in Children [87] | Direct comparison of lab (129-channel system) vs. home (32-channel portable system) EEG in children under 4. | Comparable data retention rates, noise levels, and spectral power measures at group level. | Portable EEG maintains data integrity comparable to laboratory systems in developmental populations. |
| fNIRS Signal Quality During Motion [88] | Comparison of fNIRS signals during finger tapping with and without head motion. | Motion noise causes oxy-Hb and deoxy-Hb signals to become positively correlated versus the negative correlation during true neural activation. | fNIRS is more motion-tolerant than fMRI, but motion correction algorithms are essential for ecological validity. |
| Semantic Decoding for BCIs [6] | Use of simultaneous EEG-fNIRS to differentiate between imagined semantic categories (animals vs. tools). | Combined approach leverages EEG's temporal resolution and fNIRS's spatial potential for a more practical BCI. | Multimodal integration enhances ecological validity and practicality for real-world applications like brain-computer interfaces. |
A 2025 study demonstrated that portable EEG systems could achieve data quality metrics highly consistent with laboratory systems in young children. Despite different equipment and settings, researchers found no systematic differences in data retention rates, noise levels, or spectral power measures at the group level across lab and home environments [87]. This finding is crucial for developmental and clinical populations for whom lab access is challenging.
Methodological Protocols for Real-World Neuroscience
Implementing neuroimaging outside the laboratory requires standardized protocols to ensure data quality and interpretability.
Protocol for Community-Based EEG Recordings [87]:
- Equipment: 32 active gel-based electrodes with a portable amplifier.
- Setup: Electrode impedances maintained below 100 kΩ, consistent with infant EEG protocols.
- Location: Conducted in homes or community settings chosen by families.
- Procedure: Aim for five minutes of continuous, task-free EEG while the child sits on a caregiver's lap.
- Data Processing: Apply identical processing pipelines (e.g., high-pass filtering, spectral analysis) to both lab and community data for direct comparison.
Protocol for fNIRS in Naturalistic Paradigms [86] [88]:
- Stimuli: Use of real-world educational videos or social interactions instead of abstract laboratory stimuli.
- Task Design: Incorporation of semi-naturalistic and fully naturalistic experiments, such as classroom learning or face-to-face communication.
- Motion Mitigation: Application of correlation-based signal improvement methods that leverage the negative correlation between oxy-Hb and deoxy-Hb to identify and reduce motion noise [88].
Integrated Experimental Workflows
Multimodal approaches combine complementary strengths of different neuroimaging technologies. The following diagram illustrates a typical workflow for a simultaneous EEG-fNIRS experiment.
Simultaneous EEG-fNIRS Experimental Workflow. This workflow captures the parallel data processing streams that converge for a comprehensive analysis of neural activity, combining EEG's temporal precision with fNIRS's hemodynamic information [6] [5].
The decision to use a specific neuroimaging modality or combination depends heavily on the research question and the required balance between control and ecological validity. The framework below visualizes this decision-making process.
Neuroimaging Modality Selection Framework. This decision tree guides the selection of an appropriate neuroimaging method based on the specific requirements of a research question, highlighting the trade-offs involved [86] [2] [90].
Essential Research Reagents and Materials
Successful implementation of neuroimaging studies, especially in real-world settings, relies on specialized equipment and software.
Table 3: Key Research Reagent Solutions for Neuroimaging
| Item Name | Function/Application | Relevance to Ecological Validity |
|---|---|---|
| High-Density EEG HydroCel Geodesic Sensor Net [87] | Dense array of electrodes for lab-based electrical brain activity recording. | Gold standard for lab EEG; serves as a benchmark for validating portable systems. |
| Portable EEG with Active Electrodes (e.g., BrainVision actiCAP) [87] | Mobile system with electrodes that amplify the signal at the source to reduce noise. | Enables high-quality recordings in homes, schools, and other community settings. |
| fNIRS System (e.g., portable continuous-wave) [2] [88] | Emits near-infrared light and detects reflected light to measure cortical hemodynamics. | Allows for brain imaging during active behaviors, social interaction, and in natural environments. |
| Accelerometer [88] | Measures head motion independently. | Can be integrated with fNIRS to provide a reference signal for motion artifact correction. |
| Correlation-Based Signal Improvement Algorithm [88] | Software method that enforces negative correlation between oxy-Hb and deoxy-Hb. | Improves fNIRS signal quality in the presence of motion, crucial for real-world studies. |
| EEGLAB & Custom MATLAB Scripts [87] | Open-source software environment for processing EEG and other neurophysiological data. | Standardizes data processing pipelines, enabling direct comparison between lab and field data. |
The transition of neuroimaging from the laboratory to real-world settings represents a paradigm shift in neuroscience and drug development. No single technology universally outperforms others; instead, each occupies a distinct niche in the research ecosystem. fMRI remains unparalleled for mapping deep brain structures with high spatial resolution but is fundamentally constrained by its immobility and sensitivity to motion. EEG provides millisecond-level temporal resolution to capture rapid neural dynamics and has proven highly adaptable to portable applications. fNIRS offers a compelling middle ground, with reasonable spatial resolution and significant tolerance for movement, making it a leading tool for studying cortical brain function in authentic social and learning environments. The most promising future direction lies in multimodal integration, such as combined EEG-fNIRS, which synergistically leverages the temporal strength of one and the spatial/ecological advantages of the other. As portable technologies and analytical methods continue to advance, they will undoubtedly enhance the scalability, inclusivity, and ecological validity of brain research, providing deeper insights into human cognition and behavior as it naturally occurs.
Validation and Multimodal Fusion: Creating a Comprehensive Picture of Brain Activity
The quest to understand human brain function relies heavily on the ability to accurately measure neural activity across both space and time. No single neuroimaging modality can fully capture the brain's complex dynamics; each technique offers distinct strengths and limitations in temporal resolution, spatial resolution, and practical applicability. Cross-validation studies, which correlate findings across multiple imaging techniques, are therefore essential for verifying results and gaining a more complete understanding of brain function. This guide focuses on two critical pairs for such validation: functional near-infrared spectroscopy (fNIRS) with functional magnetic resonance imaging (fMRI), and electroencephalography (EEG) with magnetoencephalography (MEG). Within the specific context of temporal resolution comparison, these correlations help researchers determine which techniques are optimal for capturing rapid neural processes versus slower hemodynamic responses. For drug development professionals and neuroscientists, understanding these relationships is crucial for selecting appropriate biomarkers and interpreting neurophysiological data across different experimental conditions.
Table: Fundamental Characteristics of Neuroimaging Modalities
| Modality | Temporal Resolution | Spatial Resolution | Measured Signal | Key Advantages | Principal Limitations |
|---|---|---|---|---|---|
| EEG | Millisecond level [56] | ~2 cm [6] | Electrical potentials from synchronized pyramidal neurons [56] | Excellent temporal resolution, portable, cost-effective [71] [56] | Poor spatial resolution, sensitive to motion artifacts and muscle activity [71] [56] |
| MEG | Millisecond level [71] | Several millimeters to centimeters [71] | Magnetic fields from electrical currents in neurons [71] | Excellent temporal resolution, not distorted by skull/skin [71] | Expensive, bulky equipment, limited to tangential currents [71] |
| fNIRS | ~0.1-1 Hz [2] | 1-3 cm [2] | Hemodynamic changes (HbO/HbR concentration) [12] [56] | Good motion tolerance, portable, measures both HbO and HbR [91] [2] [92] | Limited to cortical regions, affected by scalp blood flow [2] [56] |
| fMRI | 0.3-2 Hz (limited by hemodynamic response) [2] | Millimeter level [71] [2] | Hemodynamic changes (BOLD signal) [2] | Whole-brain coverage including subcortical structures, high spatial resolution [71] [2] | Poor temporal resolution, expensive, sensitive to motion, noisy environment [71] [2] |
Technical Bases for Correlation
Neurovascular Coupling: The fNIRS-fMRI Link
The correlation between fNIRS and fMRI is rooted in neurovascular coupling, the physiological process that links neural activity to subsequent changes in cerebral blood flow [56]. When neurons become active, they trigger a hemodynamic response that delivers oxygenated blood to the region. This process underlies the signals detected by both modalities:
- fNIRS measures this coupling directly by quantifying concentration changes in oxygenated hemoglobin (HbO) and deoxygenated hemoglobin (HbR) in cortical blood vessels using near-infrared light [56].
- fMRI detects the Blood Oxygen Level Dependent (BOLD) signal, which reflects the ratio of oxygenated to deoxygenated hemoglobin [2].
The relationship is strengthened because fNIRS measurements have been proven to be similar to the BOLD response obtained by fMRI [56]. Specifically, the fMRI BOLD signal has been shown to correlate negatively with HbR and positively with HbO, creating a direct physiological link between the modalities [2]. This shared basis in hemodynamic response provides a strong foundation for cross-validation, though the temporal characteristics differ due to each technique's specific measurement constraints.
Common Neural Generators: The EEG-MEG Connection
EEG and MEG share a more direct relationship as both measure electrophysiological activity originating from the same underlying neural populations:
- EEG records electrical potentials generated primarily by synchronized post-synaptic potentials at cortical pyramidal neurons [56]. These signals must pass through several layers including the cerebrospinal fluid, skull, and scalp, which blur the electrical potential distribution.
- MEG detects the magnetic fields produced by these same electrical currents [71]. Since magnetic fields are less distorted by the skull and scalp, MEG can provide more accurate spatial localization of the neural sources.
Both techniques capture neural activity with millisecond temporal resolution, making them ideal for studying the rapid dynamics of brain function [71] [56]. Their correlation stems from this shared origin in electrophysiological processes, though each modality has different sensitivity profiles—EEG is more sensitive to radial sources and tangential dipoles, while MEG primarily detects tangential currents.
Diagram Title: Neural Signal Pathways for Major Neuroimaging Modalities
fNIRS-fMRI Correlation
Experimental Protocols for fNIRS-fMRI Correlation
Correlating fNIRS with fMRI requires carefully designed experimental protocols that account for the technical constraints of both systems, particularly when conducted simultaneously. Two primary integration approaches have been established:
Synchronous Acquisition: This method involves collecting fNIRS and fMRI data simultaneously within the MRI scanner environment [2]. This approach requires MR-compatible fNIRS hardware specifically designed to function without interference in high magnetic fields. The synchronization of data acquisition is typically achieved through trigger pulses from the MRI scanner to the fNIRS system to align the datasets precisely. Experimental paradigms for synchronous acquisition often include block designs (alternating periods of task and rest) or event-related designs that can capture the hemodynamic response functions detectable by both modalities.
Asynchronous Acquisition: In this approach, fNIRS and fMRI data are collected in separate sessions but using identical or closely matched experimental paradigms [2]. This method avoids the technical challenges of simultaneous recording but introduces potential variability due to session differences. To ensure valid correlations, researchers must maintain consistency in task design, stimulus presentation, and participant instruction across sessions. Spatial co-registration is achieved using fiduciary markers and individual anatomical scans to align fNIRS probe locations with fMRI voxels.
For both approaches, motor tasks (such as finger tapping) and cognitive paradigms (including working memory or visual processing tasks) are commonly used, as they elicit robust, localized hemodynamic responses measurable by both techniques [2].
Key Correlation Findings and Data
The correlation between fNIRS and fMRI has been extensively validated across multiple studies and brain regions:
Spatial Concordance: Studies consistently show strong spatial correspondence between fNIRS activation maps and fMRI BOLD signals in cortical regions. The primary motor cortex activation during finger-tapping tasks demonstrates a spatial correlation of 0.6-0.9 between fNIRS HbO concentrations and fMRI BOLD signals [2]. This high correlation validates fNIRS as a reliable tool for mapping localized cortical activation.
Temporal Relationships: While both modalities track the hemodynamic response, differences emerge in their temporal profiles. The fNIRS signal typically shows a faster response onset compared to the fMRI BOLD signal, as fNIRS more directly measures blood oxygenation changes without the complex weighting of the BOLD effect [2]. The peak hemodynamic response in both modalities generally occurs 4-6 seconds after stimulus onset, with fNIRS providing better characterization of the initial dip due to its direct measurement of hemoglobin species.
Quantitative Correlations: The table below summarizes correlation coefficients reported across multiple studies:
Table: fNIRS-fMRI Correlation Coefficients by Brain Region and Task
| Brain Region | Task Paradigm | Correlation Coefficient (HbO-BOLD) | Notes | Study References |
|---|---|---|---|---|
| Prefrontal Cortex | Working Memory (n-back) | 0.72-0.89 | Strongest correlation in dorsolateral PFC | [2] |
| Motor Cortex | Finger Tapping | 0.65-0.91 | Consistent across hemisphere activation | [2] |
| Visual Cortex | Checkerboard Stimulation | 0.58-0.79 | Lower correlation potentially due to hair density | [2] |
| Auditory Cortex | Speech Processing | 0.61-0.83 | Moderate correlation across temporal regions | [2] |
The integration of fMRI's high spatial resolution with fNIRS's superior temporal resolution and portability enables robust spatiotemporal mapping of neural activity, validated across motor, cognitive, and clinical tasks [2]. This complementary relationship makes combined fNIRS-fMRI particularly valuable for studies requiring both precise spatial localization and naturalistic testing environments.
EEG-MEG Correlation
Experimental Protocols for EEG-MEG Correlation
Correlating EEG and MEG signals presents unique challenges due to their different physical properties and sensitivity profiles. Established protocols include:
Simultaneous EEG-MEG Recording: This gold-standard approach collects data from both modalities at the same time, ensuring identical brain states and experimental conditions. Successful simultaneous recording requires:
- MRI-compatible EEG systems that do not produce magnetic interference
- Careful preparation to minimize artifacts from eye movements, muscle activity, and cardiac signals
- Sensor co-registration using head position indicator coils and digitization of electrode locations relative to anatomical landmarks
- Shielding of EEG electrodes to reduce interference with MEG sensors
Source Reconstruction Methods: Advanced algorithms are employed to estimate the neural sources underlying both EEG and MEG signals:
- Realistic head models created from individual MRI scans improve accuracy
- Boundary Element Method (BEM) and Finite Element Method (FEM) are used to model electrical conductivity and magnetic field propagation
- Beamforming and Minimum Norm Estimation approaches help localize sources from both modalities
Common Experimental Paradigms:
- Somatosensory evoked responses to electrical median nerve stimulation
- Auditory processing tasks using tone bursts or speech stimuli
- Visual processing with checkerboard patterns or face stimuli
- Cognitive tasks such as oddball paradigms or working memory tasks
A novel method using virtual sensors (VS) that combines EEG and MEG data has been shown to directly capture brain activity while improving accuracy and identifying trial-to-trial variability [71]. This approach offers clearer insights into communication between different brain regions while simplifying computational processes.
Key Correlation Findings and Data
EEG and MEG show strong correlations due to their shared neural origins, with important distinctions:
Temporal Correlation: EEG and MEG signals exhibit nearly identical temporal patterns with millisecond precision, reflecting their common generation by synchronized neural populations [71]. The correlation in time courses typically exceeds 0.9 for evoked responses, confirming their equivalent temporal resolution for capturing neural dynamics.
Spatial Complementarity: While both modalities identify similar activated brain regions, MEG generally provides superior spatial resolution (several millimeters) compared to EEG (~2 cm) due to reduced distortion from skull and scalp tissues [71]. The integration of both signals through source localization algorithms can achieve spatial accuracy approaching 5-10 mm for cortical sources.
Sensitivity Profiles: Each modality has distinct sensitivity to different neural source orientations:
- MEG is primarily sensitive to tangential currents in sulcal walls
- EEG detects both tangential and radial currents but with greater blurring from volume conduction
- Combined EEG-MEG provides more complete sampling of neural activity
Table: EEG-MEG Correlation Metrics by Neural Oscillation Band
| Frequency Band | Spatial Correlation | Temporal Correlation | MEG Advantage | EEG Advantage |
|---|---|---|---|---|
| Theta (4-7 Hz) | 0.75-0.85 | 0.88-0.95 | Better deep source localization | Better frontal lobe coverage |
| Alpha (8-12 Hz) | 0.82-0.91 | 0.91-0.97 | Superior occipital localization | Enhanced sensitivity to posterior rhythms |
| Beta (15-30 Hz) | 0.78-0.87 | 0.85-0.93 | Better spatial precision | Greater amplitude for movement-related activity |
| Gamma (>30 Hz) | 0.65-0.79 | 0.72-0.88 | Less muscle artifact contamination | More clinical applications established |
The combination of EEG and MEG is particularly powerful for studying fast neural dynamics during cognitive processes, with MEG providing more accurate spatial localization while EEG offers broader coverage including radial sources [71]. This synergy makes them ideal for investigating the timing and location of neural events with high precision.
The Scientist's Toolkit
Research Reagent Solutions
Table: Essential Materials and Equipment for Multimodal Neuroimaging Studies
| Item Category | Specific Examples | Function/Purpose | Key Considerations |
|---|---|---|---|
| fNIRS Hardware | Continuous Wave (CW) NIRS systems, time-domain/drequency-domain systems [56] | Measures HbO/HbR concentration changes using near-infrared light | Portability, number of channels, sampling rate, compatibility with other systems [2] |
| EEG Systems | Active/passive electrodes, amplifiers, electrode caps [56] | Records electrical brain activity with high temporal resolution | Number of channels, impedance requirements, compatibility with MEG/fMRI [71] |
| MEG Systems | Whole-head neuromagnetometers, SQUID sensors [71] | Measures magnetic fields from neural currents | Sensitivity, helmet design, environmental shielding [71] |
| fMRI Systems | 3T/7T MRI scanners, head coils [2] | Provides high-spatial-resolution BOLD signals | Magnetic field strength, gradient performance, multi-channel coils [2] |
| Integration Equipment | MR-compatible EEG/fNIRS systems, synchronization interfaces, custom helmets [12] [2] | Enables simultaneous multimodal data acquisition | Hardware compatibility, artifact minimization, patient comfort [12] |
| Auxiliary Monitors | Short-separation fNIRS detectors, EOG/ECG electrodes, motion tracking [8] | Records and removes non-neural confounds | Placement optimization, sampling rate synchronization [8] |
Experimental Workflow Integration
Diagram Title: Multimodal Neuroimaging Experimental Workflow
Cross-validation studies correlating fNIRS with fMRI and EEG with MEG provide essential verification of neuroimaging findings while leveraging the complementary strengths of each modality. The strong correlation between fNIRS and fMRI hemodynamic responses validates fNIRS as a reliable tool for mapping cortical activation with the advantages of portability and higher temporal resolution, while fMRI provides unparalleled spatial precision and deep brain coverage. Similarly, EEG and MEG show remarkable temporal correlation with complementary spatial sensitivity profiles, making their combination powerful for studying fast neural dynamics with improved source localization.
For researchers and drug development professionals, these correlation studies offer critical insights for selecting appropriate imaging biomarkers and interpreting neurophysiological data across different experimental contexts. The continued refinement of multimodal integration approaches, including hardware compatibility, standardized protocols, and advanced data fusion algorithms, will further enhance the validity and utility of these correlations in both basic neuroscience and clinical applications. As these fields evolve, cross-validation will remain fundamental to establishing robust, reproducible neuroimaging findings that advance our understanding of brain function in health and disease.
In cognitive neuroscience and clinical neuroimaging, a fundamental trade-off has long defined technological capabilities: the choice between capturing brain activity with high temporal precision or with clear spatial localization. Electroencephalography (EEG) measures electrical brain activity directly with millisecond-scale temporal resolution, ideal for tracking fast cognitive processes, but suffers from limited spatial accuracy due to signal dispersion through the skull and scalp [93] [13]. Conversely, functional near-infrared spectroscopy (fNIRS) measures hemodynamic responses (changes in blood oxygenation) with superior spatial resolution for cortical areas, but is constrained by the slow nature of blood flow, with temporal resolution on the scale of seconds [93] [82]. Functional magnetic resonance imaging (fMRI), while offering high spatial resolution, lacks portability and has even poorer temporal resolution than fNIRS [30] [6].
This complementary relationship has driven the emergence of integrated EEG-fNIRS systems that simultaneously capture both electrophysiological and hemodynamic aspects of brain activity. By harnessing the temporal strengths of EEG and spatial advantages of fNIRS, researchers can achieve superior spatiotemporal resolution than either modality provides alone [30]. This multimodal approach enables new research possibilities across cognitive neuroscience, brain-computer interfaces (BCIs), and clinical applications from epilepsy monitoring to neurorehabilitation [12] [94]. This guide examines the technical foundations, experimental evidence, and practical implementation of this powerful integrated approach to brain imaging.
Fundamental Technical Comparisons: EEG vs. fNIRS
Core Physiological Principles and Measurement Characteristics
EEG and fNIRS capture fundamentally different physiological processes associated with neural activity. EEG records electrical potentials generated by synchronized firing of cortical neurons, primarily pyramidal cells [93]. These voltage fluctuations are measured directly at the scalp surface but are significantly attenuated and blurred by intervening tissues. fNIRS is an optical technique that uses near-infrared light to measure changes in cerebral blood oxygenation resulting from neural activity via neurovascular coupling [93] [82]. It detects concentration changes in oxygenated hemoglobin (HbO) and deoxygenated hemoglobin (HbR) in the outer layers of the cortex [82].
Table 1: Fundamental Characteristics of EEG and fNIRS
| Feature | EEG (Electroencephalography) | fNIRS (Functional Near-Infrared Spectroscopy) |
|---|---|---|
| What It Measures | Electrical activity of neurons | Hemodynamic response (blood oxygenation levels) |
| Signal Source | Postsynaptic potentials in cortical neurons | Changes in oxygenated and deoxygenated hemoglobin |
| Temporal Resolution | High (milliseconds) [93] | Low (seconds) [93] |
| Spatial Resolution | Low (centimeter-level) [93] | Moderate (better than EEG, limited to cortex) [93] |
| Depth of Measurement | Cortical surface | Outer cortex (~1–2.5 cm deep) [93] |
| Sensitivity to Motion | High – susceptible to movement artifacts [93] | Low – more tolerant to subject movement [93] |
| Portability | High – lightweight and wireless systems available [93] | High – often used in mobile and wearable formats [93] |
| Best Use Cases | Fast cognitive tasks, ERP studies, sleep research [93] | Naturalistic studies, child development, motor rehab [93] |
Complementary Strengths and Limitations in Practice
The technical differences between EEG and fNIRS translate to distinctly different experimental applications. EEG excels at capturing rapid neural dynamics such as sensory processing, attentional shifts, and motor planning, where millisecond-level timing is crucial [93]. Its portability and relatively low cost make it accessible, but its sensitivity to environmental electrical noise and movement artifacts can limit its use in real-world settings [13] [94].
fNIRS is ideally suited for studying sustained cognitive states such as mental workload, problem-solving, and emotional processing, particularly when localized to specific cortical regions like the prefrontal cortex [93]. Its tolerance to movement artifacts makes it valuable for studies involving children, mobile participants, or real-world environments like classrooms or sports settings [93]. However, the inherent delay of the hemodynamic response (2–6 seconds) fundamentally limits its temporal precision [93] [94].
The Integrated Approach: Theoretical Foundations and Experimental Evidence
The Spatiotemporal Resolution Advantage
The core value proposition of integrated EEG-fNIRS imaging lies in overcoming the fundamental limitations of each modality when used independently. Simulation studies demonstrate that two neuronal sources separated by just 2.3–3.3 cm and activated with a temporal separation of only 50 ms can be accurately reconstructed using joint EEG-fNIRS methods, while remaining indistinguishable to either modality alone [30]. This represents a significant advancement in spatiotemporal resolution for non-invasive neuroimaging.
The integration leverages the principle of neurovascular coupling—the established relationship between neural electrical activity and subsequent hemodynamic responses [30]. While EEG directly captures the fast electrical activity and fNIRS measures the slower hemodynamic consequences, combining these signals enables researchers to create a more complete picture of brain dynamics than either modality can provide separately.
Experimental Validation in Brain-Computer Interfaces
The performance benefits of integrated EEG-fNIRS systems have been quantitatively demonstrated in multiple BCI studies. In a motor execution task involving left and right hand movements, a hybrid system achieved a classification accuracy of 91.02% ± 4.08%, significantly outperforming either modality alone (EEG: 85.64% ± 7.4%; fNIRS: 85.55% ± 10.72%) [94]. This performance enhancement was achieved while using a minimal number of channels and focusing on early temporal features (0–1 s for EEG, 0–2 s for fNIRS), thereby reducing system complexity and improving response time [94].
Table 2: Performance Comparison in Motor Imagery BCI Tasks
| Modality | Classification Accuracy | Key Features Utilized | Advantages | Limitations |
|---|---|---|---|---|
| EEG Alone | 85.64% ± 7.4% [94] | Sensorimotor rhythms (ERD/ERS) | Millisecond temporal resolution | Limited spatial specificity; sensitive to artifacts |
| fNIRS Alone | 85.55% ± 10.72% [94] | Hemodynamic responses (HbO/HbR) | Better spatial localization; robust to noise | Slow hemodynamic response (4-6 s delay) |
| Integrated EEG-fNIRS | 91.02% ± 4.08% [94] | Early EEG responses + initial fNIRS dip | Enhanced accuracy & information transfer rate | Increased system complexity & computational cost |
More recent studies have extended these findings to semantic decoding tasks. Simultaneous EEG-fNIRS recordings during imagination of animals and tools demonstrated the feasibility of decoding semantic categories from multimodal brain signals, with fNIRS potentially addressing EEG's poor spatial resolution while EEG compensates for fNIRS's slow temporal response [6]. Similarly, a 2025 dataset featuring eight different motor imagery tasks from 18 subjects demonstrated the utility of multimodal recordings for developing more intuitive BCI systems [95].
The following diagram illustrates the complementary relationship and integration benefits of EEG and fNIRS:
Technical Implementation: Methodologies and Experimental Protocols
Data Acquisition and Hardware Integration
Successful multimodal imaging requires careful technical implementation. Simultaneous EEG-fNIRS systems typically utilize integrated caps with pre-defined openings for both electrodes and optodes, often based on the international 10–20 system for standardized placement [93] [12]. Proper cap fitting is crucial, as excessive pressure can affect fNIRS signal quality, while loose fitting increases motion artifacts, particularly for EEG [12].
Hardware synchronization is achieved through various methods. Some vendors offer integrated systems with shared clocks, while others require synchronization via external triggers (e.g., TTL pulses) or shared acquisition software [93] [12]. The choice between these approaches involves trade-offs between synchronization precision and system flexibility. Unified processors that simultaneously process both signal types generally achieve higher synchronization accuracy compared to systems that merely synchronize data during post-processing [12].
Experimental Protocols and Data Processing
Motor Execution/Imagery Paradigm
A well-established protocol for evaluating hybrid EEG-fNIRS systems involves motor tasks. A typical experiment consists of multiple randomized trials of left and right hand movements or motor imagery [94]. Each trial begins with a rest period (e.g., 20 seconds), followed by a visual cue indicating the task (e.g., 5 seconds), during which participants perform the actual or imagined movement [94]. This design elicits robust, lateralized responses in both electrophysiological (EEG sensorimotor rhythms) and hemodynamic (fNIRS oxygenation changes) domains.
Data Preprocessing and Fusion Techniques
EEG and fNIRS data require distinct preprocessing pipelines before integration. EEG processing typically involves filtering, artifact removal (e.g., ocular, cardiac), and often re-referencing [96]. fNIRS processing includes converting raw light intensity measurements to optical density, then to hemoglobin concentration changes using the Modified Beer-Lambert Law, followed by filtering to remove physiological noise and motion artifacts [96] [82].
Data fusion techniques can be categorized into several approaches:
- Concatenation-based methods: Features from both modalities are combined into a single feature vector for classification [96].
- Model-based approaches: Incorporate priors or constraints from one modality into the analysis of the other [96] [30].
- Source-decomposition techniques: Methods like joint Independent Component Analysis (jICA) identify latent components that represent shared sources of variance across modalities [96].
- Decision-level fusion: Separate classifications are performed for each modality, with results combined at the decision stage [96].
The following diagram illustrates a typical experimental workflow for simultaneous EEG-fNIRS studies:
Essential Research Solutions and Equipment
Implementing a successful multimodal EEG-fNIRS research program requires specific technical resources. The following table details key components and their functions in typical experimental setups:
Table 3: Essential Research Solutions for EEG-fNIRS Studies
| Component Category | Specific Examples | Function & Importance | Technical Considerations |
|---|---|---|---|
| Acquisition Systems | NIRScout (fNIRS), BrainAMP (EEG) [12] [95] | Simultaneous signal recording with precise synchronization | Integrated systems preferred; otherwise external synchronization hardware needed |
| Integrated Caps/Holders | Custom 3D-printed helmets, modified EEG caps with optode holders [12] | Secure, co-registered placement of electrodes and optodes | Proper fit critical for signal quality; consider head size variability |
| Data Processing Tools | MATLAB toolboxes, Homer2, NIRS-KIT, EEGLAB [96] | Preprocessing, artifact removal, data fusion, and analysis | Separate preprocessing pipelines needed before multimodal integration |
| Stimulus Presentation | Presentation, Psychtoolbox, E-Prime | Precise timing and delivery of experimental paradigms | Synchronization with acquisition system crucial for event-related designs |
| Classification Algorithms | Support Vector Machines (SVM), Linear Discriminant Analysis (LDA), Deep Learning models [95] [94] | Pattern recognition and decoding of multimodal brain signals | Early temporal features (0-2s) often provide optimal performance [94] |
The integration of EEG and fNIRS represents a significant advancement in non-invasive brain imaging, offering spatiotemporal resolution capabilities that overcome fundamental limitations of either modality used independently. Experimental evidence consistently demonstrates enhanced classification accuracy in BCI applications and improved source localization for cognitive neuroscience research [30] [94].
Future developments in this field will likely focus on improving real-time processing capabilities, enhancing the mobility and wearability of integrated systems, and developing more sophisticated data fusion algorithms [96] [12]. As these technologies mature, simultaneous EEG-fNIRS imaging is poised to become an increasingly standard approach for studying brain function in both laboratory and real-world settings, offering unprecedented insights into the dynamic relationship between electrical and hemodynamic aspects of neural activity.
For researchers considering this approach, the decision to implement a multimodal system should be guided by specific research questions that require both high temporal and spatial resolution, particularly when studying complex cognitive processes or developing BCIs for naturalistic applications. While the technical complexity increases compared to single-modality studies, the complementary information gained through integrated EEG-fNIRS imaging provides a more comprehensive window into brain function.
In neuroscience and drug development, the quest to comprehend brain function is often hampered by the fundamental limitations of individual neuroimaging modalities. Electroencephalography (EEG), functional near-infrared spectroscopy (fNIRS), and functional magnetic resonance imaging (fMRI) each provide a unique, yet incomplete, window into brain activity. Data fusion addresses this challenge by combining data from multiple sources to achieve improved accuracy and more specific inferences than could be achieved by a single source alone [97]. This process is essential for creating a unified, comprehensive picture of neural events, marrying the millisecond-scale temporal dynamics of electrical activity with the millimeter-scale spatial localization of hemodynamic responses.
The inherent complementarity of EEG and fNIRS signals is a key driver for their joint use in multimodal studies. EEG measures the electrical activity of cortical neurons directly, offering unparalleled temporal resolution on the order of milliseconds, but suffers from limited spatial resolution (centimeter-scale) due to the blurring effect of the skull and scalp [13] [98]. Conversely, fNIRS measures changes in blood oxygenation (hemodynamic response), an indirect marker of neural activity. It provides superior spatial resolution for surface cortical areas but is constrained by the slow evolution of hemodynamics, resulting in a temporal resolution of several seconds [30] [98]. fMRI, while offering high spatial resolution and whole-brain coverage, is expensive, non-portable, and has very low temporal resolution, making it less suitable for real-world or bedside applications [13]. Table 1 summarizes the core characteristics of these modalities, highlighting their complementary strengths and weaknesses.
Table 1: Comparison of Key Non-Invasive Neuroimaging Modalities
| Feature | EEG | fNIRS | fMRI |
|---|---|---|---|
| What It Measures | Electrical activity from neurons | Hemodynamic response (HbO/HbR) | Blood oxygenation level-dependent (BOLD) signal |
| Temporal Resolution | High (milliseconds) [98] | Low (seconds) [98] | Low (seconds) [13] |
| Spatial Resolution | Low (centimeter-level) [98] | Moderate (better than EEG) [98] | High (millimeter-level) [13] |
| Portability & Cost | High portability, generally lower cost [98] | High portability, generally higher cost than EEG [98] | Non-portable, very high cost [13] |
| Key Strength | Timing of neural events | Localized cortical activity in real-world settings | Whole-brain anatomical mapping |
This guide objectively compares three prominent data fusion methods—Concatenation (Early Fusion), Model-Based Fusion, and Source-Decomposition Methods—within the context of EEG-fNIRS research, providing experimental data and detailed protocols to inform researchers and drug development professionals.
Foundational Data Fusion Concepts and Classifications
Data fusion techniques can be classified based on the stage at which integration occurs. The most established taxonomy, from Dasarathy, defines several levels [99] [97]:
- Data In-Data Out (DAI-DAO) / Low-Level Fusion: This involves raw data fusion, such as the simple concatenation of raw or pre-processed signals.
- Feature In-Feature Out (FEI-FEO) / Mid-Level Fusion: Features extracted from each modality are combined. This is also known as feature-level fusion.
- Decision In-Decision Out (DEI-DEO) / High-Level Fusion: Decisions or outputs from unimodal classifiers are fused, also known as decision-level fusion or late fusion.
Another critical perspective considers the relationship between the data sources, which can be complementary (providing different parts of the scene), redundant (providing information about the same target), or cooperative (combining to generate new, more complex information) [97]. The fusion of EEG and fNIRS is a quintessential example of a cooperative and complementary approach.
Concatenation-Based Fusion (Early Fusion)
Methodology and Theoretical Basis
Concatenation-based fusion, also known as early fusion or data-level fusion, involves directly combining raw or pre-processed data from multiple modalities into a single, unified feature vector before it is fed into a predictive model [99]. In the context of EEG-fNIRS, this typically means splicing the temporal signals or initial extracted features (e.g., EEG band powers and fNIRS HbO/HbR concentrations) into one long input vector.
Formally, within a generalized linear model framework, early fusion can be defined as finding a connection function ( gE ) such that the model output ( \etaE ) is a weighted sum of all features from all modalities [99]: [ gE(\mu) = \etaE = \sum{i=1}^{m} wi xi ] where ( xi ) are the features from the combined set of all modalities and ( wi ) are the non-zero weight coefficients. The final prediction is the inverse of the connection function, ( gE^{-1}(\eta_E) ) [99].
Experimental Evidence and Performance
Early fusion is straightforward but its performance is highly dependent on the dataset and task. Experimental results are mixed, with some studies showing modest improvements.
- Mental Workload Classification: A study on driver drowsiness using concatenated EEG-fNIRS features yielded a modest average improvement of 5.5% in classification accuracy compared to using features from a single modality [100].
- Parkinson's Disease Detection: In research aimed at distinguishing Parkinson's disease, simple feature concatenation provided only a 1% accuracy improvement over single-modality approaches [100].
- Theoretical Limitations: Mathematical analysis suggests that early fusion can fail when there are complex, nonlinear relationships between features and labels. Its performance is also highly sensitive to sample size, feature quantity, and the number of modalities [99].
The following diagram illustrates the straightforward workflow of concatenation-based fusion.
Model-Based Data Fusion
Methodology and Theoretical Basis
Model-based fusion techniques perform the integration of multimodal data and the estimation of the underlying source of interest simultaneously within a unified mathematical model. Unlike concatenation, this approach incorporates the physical and statistical properties of the measurement process directly into the inversion framework.
A prominent example is Model-Based Material Decomposition (MBMD) used in spectral CT imaging, which has parallels to neurovascular inverse problems. MBMD uses a polyenergetic forward model to directly estimate material density images from multi-channel measurement data, combining the reconstruction and decomposition steps [101]. The objective function in such a penalized weighted least-squares framework can be generalized as [101]: [ \psi = [\mathbf{y} - B \exp(-M \mathbf{x})]^T K^{-1} [\mathbf{y} - B \exp(-M \mathbf{x})] + R(\mathbf{x}) ] Here, ( \mathbf{y} ) represents the measurements, ( \mathbf{x} ) is the image (or source) to be estimated, ( B ) and ( M ) model the physical measurement process, ( K ) is the measurement covariance, and ( R(\mathbf{x}) ) is a regularizer. This direct approach avoids the trade-offs of traditional two-step methods and allows for the use of unmatched or undersampled data channels [101].
Experimental Evidence and Performance
Model-based approaches demonstrate high sensitivity and accuracy in challenging scenarios.
- Joint EEG-fNIRS Source Reconstruction: A simulation study using the ICBM152 brain atlas demonstrated that a model-based algorithm using DOT reconstruction as a spatial prior for EEG could accurately resolve two neuronal sources separated by only 2.3-3.3 cm and 50 ms. This level of spatiotemporal resolution was unattainable by either modality in isolation [30].
- Spectral CT Material Decomposition: In a kV-switching simulation, a model-based decomposition algorithm could reconstruct iodine at a very low concentration of 0.5 mg ml⁻¹, whereas traditional image-domain decomposition required a six times higher concentration (3.0 mg ml⁻¹) for a usable contrast-to-noise ratio [101].
The workflow for a model-based fusion approach, such as in joint source reconstruction, is more integrated, as shown below.
Source-Decomposition-Based Fusion
Methodology and Theoretical Basis
Source-decomposition methods aim to separate the underlying sources or components that generate the observed multimodal signals. These are often considered intermediate or feature-level fusion techniques. A common approach is Joint Independent Component Analysis (jICA), which assumes that the signals from different modalities are generated by the same set of statistically independent sources [100] [102].
Another powerful method uses Mutual Information (MI) as a criterion for feature selection and fusion. The goal is to select a subset of features from the combined EEG-fNIRS set that maximizes the relevance of features to the class labels while minimizing redundancy between features and maximizing their complementarity [100]. Complementarity exists when two features together provide more information about the output class than the sum of the information each provides individually [100].
Empirical Mode Decomposition (EMD) is a data-driven technique used to process nonlinear and non-stationary sequences, such as physiological signals, by decomposing them into Intrinsic Mode Functions (IMFs) and a residue [103]. This can be applied to pre-process signals before fusion to isolate distinct oscillatory modes.
Experimental Evidence and Performance
Decomposition and feature selection methods have shown significant improvements in classification performance across various tasks.
- Mental Stress Detection: A study using jICA to fuse EEG-fNIRS features achieved a detection rate of 98%, a substantial improvement over using fNIRS (91%) or EEG (95%) alone [102].
- Amyotrophic Lateral Sclerosis (ALS) Classification: A mutual information-based feature selection framework resulted in "considerably improved hybrid classification performance" compared to individual modalities and to conventional classification without feature selection [100].
- Motor Imagery and Mental Arithmetic Tasks: A multi-level progressive learning framework that included feature decomposition and selection achieved average classification accuracies of 96.74% for motor imagery and 98.42% for mental arithmetic tasks on a 29-subject EEG-fNIRS dataset [102].
The following diagram outlines the key steps in a mutual information-based feature selection pipeline for EEG-fNIRS fusion.
Comparative Analysis of Fusion Techniques
Table 2 provides a consolidated overview of the three fusion methods, summarizing their core principles, requirements, advantages, and disadvantages based on the cited experimental evidence.
Table 2: Comparative Analysis of Data Fusion Techniques for EEG-fNIRS
| Fusion Method | Core Principle | Data/Matching Requirements | Reported Advantages | Reported Disadvantages/Limitations |
|---|---|---|---|---|
| Concatenation (Early Fusion) | Simple splicing of raw data or features into a single input vector [99]. | Requires temporal alignment of data streams. | Simple to implement [100]. | Modest performance gains; can be sensitive to noise and redundant features; may fail with nonlinear feature-label relationships [99] [100]. |
| Model-Based Fusion | Simultaneous integration and inversion using a unified physico-statistical model [30] [101]. | Can work with unmatched or undersampled data [101]. | High sensitivity and accuracy; enables novel acquisition protocols; can achieve high spatiotemporal resolution [30] [101]. | Computationally complex; requires accurate forward models and priors. |
| Source-Decomposition (e.g., jICA, MI) | Blind source separation or optimal feature selection based on information theory [100] [102]. | Requires a common feature space or statistical assumptions (e.g., independence). | Can significantly improve classification accuracy; effectively exploits complementarity and reduces redundancy [100] [102]. | Performance depends on validity of underlying assumptions (e.g., statistical independence); feature selection can be computationally heavy. |
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful execution of multimodal EEG-fNIRS experiments requires specific hardware, software, and analytical tools. The following table details key components of a research pipeline for data fusion studies.
Table 3: Essential Research Reagents and Solutions for EEG-fNIRS Fusion
| Item | Function/Description | Example Use Case |
|---|---|---|
| Integrated EEG-fNIRS Caps | Head caps with pre-defined placements for both EEG electrodes and fNIRS optodes, often following the international 10-20 system. | Ensures proper spatial co-registration of electrical and hemodynamic measurements from the same cortical areas, minimizing sensor placement artifacts [98]. |
| Synchronization Trigger Box | Hardware device that sends simultaneous timing pulses (e.g., TTL pulses) to both EEG and fNIRS acquisition systems. | Critical for temporal alignment of data streams during post-processing, especially for event-related potential (ERP) and hemodynamic response analysis [98]. |
| High-Density DOT Systems | fNIRS systems with overlapping source-detector channels designed to achieve millimeter-scale spatial resolution for Diffuse Optical Tomography (DOT) [30]. | Provides high-quality spatial priors for model-based joint source reconstruction with EEG, enhancing localization accuracy [30]. |
| Mutual Information Feature Selection Algorithm | A computational filter method that selects an optimized subset of multimodal features based on relevance, redundancy, and complementarity criteria [100]. | Used in decomposition-based fusion to improve BCI classification performance for pathological conditions like ALS or for mental state monitoring [100]. |
| Variational Bayesian/ReML Framework | A model-based statistical framework (e.g., Variational Bayesian Multimodal Encephalography or Restricted Maximum Likelihood) used for solving ill-posed inverse problems. | Employed in joint EEG-fNIRS source reconstruction to incorporate spatial priors from fNIRS/DOT into the EEG inversion process [30]. |
The choice of an optimal data fusion technique for EEG-fNIRS research is not one-size-fits-all but is dictated by the specific goals, constraints, and nature of the scientific inquiry. Concatenation-based fusion offers a simple entry point but often yields only modest performance gains. Model-based fusion provides a powerful, principled framework for achieving high spatiotemporal resolution in source localization, which is crucial for mapping precise neural circuits. Source-decomposition and feature-selection methods currently demonstrate superior performance in classification tasks, such as BCI and disease diagnosis, by effectively leveraging the complementary information in the electrical and hemodynamic signals.
For researchers and drug development professionals, this implies that diagnostic applications seeking biomarkers may benefit most from decomposition and feature-selection fusion, while studies aimed at understanding the precise dynamics of neural networks might prioritize model-based approaches. As the field advances, the development of more integrated and computationally efficient fusion algorithms will continue to push the boundaries of our ability to decode the complex workings of the human brain.
In non-invasive neuroimaging, a fundamental trade-off exists between temporal and spatial resolution, often termed the "resolution paradox." Electroencephalography (EEG) provides direct measurement of neuronal electrical activity with millisecond-level temporal resolution, ideal for tracking the fast dynamics of brain function. However, its spatial resolution is limited, often on the centimeter scale, because electrical signals are blurred as they pass through the skull and scalp [104]. Conversely, functional Magnetic Resonance Imaging (fMRI), long considered the gold standard for in-vivo brain imaging, offers high spatial resolution but measures the slow hemodynamic response (blood flow changes), resulting in a temporal resolution on the scale of seconds [3]. Functional Near-Infrared Spectroscopy (fNIRS) occupies a unique middle ground; like fMRI, it measures the hemodynamic response but offers greater portability, lower cost, and higher tolerance for movement, albeit with more limited penetration depth than fMRI [104] [3].
Multimodal neuroimaging strategically integrates these techniques to overcome the limitations of individual modalities. By combining the millisecond temporal precision of EEG with the superior spatial localization of fNIRS or fMRI, researchers and clinicians can construct a more comprehensive and spatiotemporally detailed picture of brain function. This approach is increasingly vital for advancing cognitive neuroscience, improving diagnostic precision in neuropsychiatric disorders, and de-risking drug development pipelines [105] [56] [106]. This guide explores successful applications of this integrated approach, with a particular focus on the synergistic relationship between EEG and fNIRS.
Technical Comparison of Core Neuroimaging Modalities
The following table summarizes the key characteristics of EEG, fNIRS, and fMRI, highlighting their complementary strengths and weaknesses.
Table 1: Technical Comparison of EEG, fNIRS, and fMRI
| Feature | EEG | fNIRS | fMRI |
|---|---|---|---|
| What It Measures | Electrical activity from postsynaptic potentials [104] | Hemodynamic response (HbO & HbR concentration) [104] | Hemodynamic response (BOLD signal) [3] |
| Temporal Resolution | Very High (Milliseconds) [104] | Low (Seconds) [104] | Low (Seconds) [3] |
| Spatial Resolution | Low (Centimeter-level) [104] | Moderate (Better than EEG, cortical only) [104] | High (Millimeter-level) [3] |
| Depth of Measurement | Cortical surface [104] | Outer cortex (~1–2.5 cm deep) [104] | Whole brain |
| Portability & Tolerance | Moderate (Portable systems exist, sensitive to motion) [104] | High (Portable, robust to movement) [104] [3] | Low (Requires scanner, highly motion-sensitive) [3] |
| Best Use Cases | Fast cognitive tasks, ERPs, sleep studies, BCIs [104] | Naturalistic studies, child development, clinical populations, BCIs [104] | Detailed anatomical mapping, deep brain structure analysis [3] |
Fundamental Mechanisms and Signaling Pathways
The physiological rationale for combining EEG and fNIRS is rooted in the principle of neurovascular coupling [56]. This is the process where neural activity triggers a subsequent hemodynamic response to meet the increased metabolic demand for oxygen and nutrients.
The Neurovascular Coupling Pathway
The following diagram illustrates the sequential relationship between the electrical events measured by EEG and the hemodynamic events measured by fNIRS.
Figure 1: The Neurovascular Coupling Pathway. This sequence shows how a stimulus triggers neural activity (measured by EEG), which creates metabolic demand and leads to a hemodynamic response (measured by fNIRS).
Experimental Protocols in Multimodal Neuroimaging
Successful multimodal studies require careful experimental design, precise hardware synchronization, and specialized data processing pipelines. The general workflow for a concurrent EEG-fNIRS study is outlined below.
Table 2: Key Research Reagent Solutions for Multimodal Studies
| Item Category | Specific Function & Explanation |
|---|---|
| Integrated EEG-fNIRS Caps | Specially designed headcaps that pre-define optode (fNIRS) and electrode (EEG) holders according to the international 10-20 system, ensuring optimal placement and minimizing interference [104]. |
| Synchronization Hardware/Software | External hardware (e.g., TTL pulses) or shared software clocks are used to temporally align the EEG and fNIRS data streams, which is critical for subsequent integrated analysis [104]. |
| Motion Correction Algorithms | Computational tools applied during data preprocessing to minimize artifacts caused by subject movement, which is particularly important for the more motion-sensitive EEG signal [104]. |
| Data Fusion Algorithms | Advanced computational methods like joint Independent Component Analysis (jICA) or machine learning models that combine feature sets from both modalities to identify correlated patterns of brain activity [104] [56]. |
Figure 2: Generalized Workflow for a Concurrent EEG-fNIRS Experiment. This flowchart outlines the key stages, from design to interpretation, in a typical multimodal neuroimaging study.
Case Studies of Successful Multimodal Integration
Case Study 1: Enhancing Brain-Computer Interface (BCI) Performance
- Research Objective: To determine if combining fNIRS with EEG could improve the classification accuracy and stability of a Sensory Motor Rhythm (SMR)-based Brain-Computer Interface, a tool that allows users to control external devices through brain signals [107].
- Experimental Protocol: Participants were asked to perform both executed movements and motor imagery (imagining a movement without performing it) while whole-head EEG and fNIRS data were recorded simultaneously. Researchers developed a "meta-classifier" to integrate the real-time classification outputs from the EEG and fNIRS data streams [107].
- Results and Data: The hybrid fNIRS-EEG system significantly enhanced performance. It improved the classification accuracy of motor imagery in over 90% of subjects, with an average performance increase of 5%. Critically, some participants who were previously unable to operate a BCI using EEG alone became able to do so with the hybrid system. The study concluded that EEG and fNIRS provide different, complementary information, making them a viable multimodal technique for BCI [107].
Case Study 2: Semantic Decoding for Advanced Communication
- Research Objective: To investigate the feasibility of decoding imagined semantic categories (animals vs. tools) to develop a more intuitive BCI for direct communication, bypassing slow character-by-character spelling [6].
- Experimental Protocol: Participants were shown images of animals and tools and performed four distinct mental tasks: silent naming, visual imagery, auditory imagery, and tactile imagery. Simultaneous EEG and fNIRS data were recorded from 12 participants to capture both the fast electrical signatures and the localized hemodynamic changes associated with these semantic thoughts [6].
- Results and Data: The dataset provided a valuable resource for testing multimodal decoding algorithms. The combination of EEG's temporal resolution (to capture the rapid onset of the thought process) and fNIRS's spatial resolution (to help localize which brain regions were engaged) created a richer feature set for machine learning models to distinguish between the two semantic categories. This approach is a critical step toward practical semantic BCIs [6].
Case Study 3: Informing Drug Development in Psychiatry
- Research Objective: To use functional neuroimaging, particularly EEG and fMRI, as pharmacodynamic biomarkers in Phase 1 clinical trials to de-risk psychiatric drug development by confirming brain penetration, functional target engagement, and informing dose selection [106].
- Experimental Protocol: In studies on drugs for cognitive impairment associated with schizophrenia (CIAS), such as phosphodiesterase 4 inhibitors (PDE4i's), researchers used EEG/ERP to measure cognition-related brain signals across multiple drug doses. This functional data was compared with molecular target occupancy data from PET imaging and tolerability measures [106].
- Results and Data: This multimodal approach revealed a critical insight. While PET imaging suggested high drug doses were needed for molecular target engagement, EEG showed that pro-cognitive effects on brain function occurred at much lower, better-tolerated doses (~30% target occupancy). Relying on PET alone would have led to doses limited by adverse events, potentially causing promising drugs to fail. EEG provided a functional readout that was essential for correct dose selection [106].
The case studies presented here demonstrate that the integration of multimodal neuroimaging, particularly the combination of EEG with fNIRS, is transforming both basic neuroscience and clinical applications. By overcoming the inherent limitations of any single modality, researchers can achieve a more nuanced and comprehensive understanding of brain function. The complementary nature of EEG's temporal resolution and fNIRS's spatial localization provides a powerful toolkit for developing more robust brain-computer interfaces, probing high-level cognitive processes like semantic thought, and bringing greater precision to the development of new neurotherapeutics. As hardware integration becomes more seamless and data fusion algorithms more sophisticated, the use of multimodal neuroimaging is poised to become the standard for cutting-edge research and clinical translation in neuroscience.
The pursuit of understanding human brain function relies heavily on the ability to accurately capture the brain's dynamic, time-varying signals. Different neuroimaging modalities offer distinct trade-offs between temporal resolution (the precision in measuring when neural events occur) and spatial resolution (the precision in locating where they occur). This guide objectively compares the performance of Electroencephalography (EEG) and functional Near-Infrared Spectroscopy (fNIRS)—two portable, non-invasive techniques—within the broader context of temporal resolution research that includes functional Magnetic Resonance Imaging (fMRI). As machine learning (ML) and artificial intelligence (AI) become increasingly sophisticated, their role in fusing these multimodal temporal data streams is pivotal, enabling researchers to overcome the inherent limitations of any single method and achieve a more holistic view of brain activity [108] [6]. This is particularly relevant for applications in cognitive neuroscience and drug development, where understanding the precise timing and location of neural events is critical for assessing cognitive states and therapeutic outcomes.
Comparative Analysis of Neuroimaging Modalities
Quantitative Comparison of EEG, fNIRS, and fMRI
The following table summarizes the core technical specifications of EEG, fNIRS, and fMRI, highlighting their performance differences, especially regarding temporal dynamics [108].
Table 1: Technical Performance Comparison of Non-Invasive Neuroimaging Modalities
| Feature | EEG (Electroencephalography) | fNIRS (Functional Near-Infrared Spectroscopy) | fMRI (Functional Magnetic Resonance Imaging) |
|---|---|---|---|
| What It Measures | Electrical activity from postsynaptic potentials of cortical neurons [108] | Hemodynamic response (changes in oxygenated and deoxygenated hemoglobin) [108] | Blood-oxygen-level-dependent (BOLD) signal, a hemodynamic response [6] |
| Temporal Resolution | Very High (Milliseconds) [108] | Low (Seconds) [108] | Very Low (Seconds) [6] |
| Spatial Resolution | Low (centimeter-level) [108] | Moderate (better than EEG, but limited to cortex) [108] | High (Millimeters) [6] |
| Depth of Measurement | Cortical surface [108] | Outer cortex (~1–2.5 cm deep) [108] | Whole brain |
| Key Strength | Capturing rapid neural dynamics (e.g., event-related potentials) [108] | Localizing sustained cortical activity in real-world settings [108] | Detailed whole-brain spatial mapping [6] |
| Key Limitation | Poor spatial resolution due to signal dispersion [108] | Indirect, slow measure of neural activity [108] | Indirect, slow measure; expensive, non-portable [6] |
The Synergy of EEG and fNIRS for Multimodal Data
EEG and fNIRS offer complementary strengths. EEG provides a direct, millisecond-scale measure of neuronal firing, making it ideal for studying the rapid brain dynamics involved in sensory processing, motor planning, and instant cognitive responses [108]. Conversely, fNIRS provides an indirect measure of neural activity through neurovascular coupling, tracking changes in blood oxygenation with a delay of 2–6 seconds. While slower, it provides better spatial localization for surface cortical areas and is more robust to movement artifacts, making it suitable for studying sustained cognitive states like workload, attention, or emotional processing in naturalistic environments [108]. The combination of these two modalities yields a data stream with both high temporal and respectable spatial resolution, which is a prime target for multimodal AI analysis [108] [6].
Machine Learning for Multimodal Temporal Data Fusion
AI Architectures for Temporal Data
Multimodal AI designed to handle temporal data must integrate and model time-dependent information from multiple asynchronous sources. These AI architectures are specifically engineered to capture sequential patterns and dependencies across time [109].
- Recurrent Neural Networks (RNNs) and Temporal Convolutional Networks (TCNs): These are traditional workhorses for sequence modeling. RNNs process sequences step-by-step, maintaining a "memory" of previous inputs, while TCNs use convolutional layers with specific padding to capture temporal context. Both can be adapted to process time-series data from EEG or fNIRS [109].
- Transformers with Attention Mechanisms: This more advanced architecture has become dominant. The self-attention mechanism allows the model to weigh the importance of different time points across the entire sequence, regardless of their distance. This is exceptionally powerful for identifying long-range dependencies in neural signals. Cross-modal attention is a key technique for synchronizing and correlating temporal data from different modalities, for instance, by linking a specific EEG waveform to a corresponding fNIRS hemodynamic change [109].
- Handling Variable Time Scales: A significant challenge is that different modalities sample data at different rates (e.g., EEG at 500 Hz vs. fNIRS at 10 Hz). AI systems employ techniques like temporal pooling or interpolation to align these streams to a common time base, ensuring that a model can jointly analyze a fast EEG signal and a slow fNIRS signal that originate from the same neural event [109].
Application in Semantic Neural Decoding
The application of these ML techniques is vividly illustrated in the domain of semantic neural decoding for Brain-Computer Interfaces (BCIs). A 2025 study published in Scientific Data provides a clear example and a valuable dataset for training and testing such AI models [6].
Experimental Protocol: Decoding Imagined Categories
- Objective: To investigate the feasibility of distinguishing between imagined semantic categories (animals vs. tools) to develop a BCI that communicates conceptual meaning directly, bypassing slower character-by-character spelling [6].
- Participants: 12 right-handed native English speakers [6].
- Task Design: Participants performed four distinct mental tasks cued by images of animals or tools:
- Silent Naming: Silently naming the object.
- Visual Imagery: Visualizing the object in their mind.
- Auditory Imagery: Imagining the sounds associated with the object.
- Tactile Imagery: Imagining the feeling of touching the object [6].
- Data Acquisition: Simultaneous EEG and fNIRS data were recorded throughout the tasks, capturing both the direct electrical and indirect hemodynamic correlates of the imagined concepts [6].
- ML Integration Point: This simultaneous, multimodal dataset is ideal for training AI models like transformers. The model can learn a shared representation that associates the fast, temporally precise EEG markers of an "imagined animal" with the slower, spatially localized fNIRS activation patterns in regions like the prefrontal cortex, thereby improving decoding accuracy and robustness [6].
Visualizing Multimodal AI Analysis Workflows
Signaling Pathways and Data Flow
The following diagram illustrates the fundamental relationship between neural electrical activity and the hemodynamic response, which is the core physiological principle leveraged by combined EEG-fNIRS studies.
Neural to Hemodynamic Pathway
Multimodal AI Experimental Workflow
This diagram outlines a generalized workflow for conducting an experiment and analyzing data using simultaneous EEG-fNIRS and multimodal AI, as seen in semantic decoding research.
Multimodal AI Analysis Pipeline
The Scientist's Toolkit: Key Research Reagent Solutions
For researchers aiming to build and analyze multimodal EEG-fNIRS datasets, the following tools and concepts are essential.
Table 2: Essential Reagents and Tools for Multimodal EEG-fNIRS Research
| Item | Function & Explanation |
|---|---|
| Integrated EEG-fNIRS Systems | Hardware systems designed for simultaneous acquisition. They mitigate sensor interference and provide synchronized data streams, which is a foundational requirement for any subsequent multimodal analysis [108]. |
| High-Density EEG Caps with fNIRS-Compatible Openings | Specialized head caps that allow for the co-location of EEG electrodes and fNIRS optodes according to the international 10-20 system, ensuring optimal sensor placement for both modalities without physical interference [108]. |
| Synchronization Triggers (TTL Pulses) | External hardware signals used to mark the onset of experimental events (like stimulus presentation) in both the EEG and fNIRS data streams. This precise timing is critical for aligning neural events with behavioral tasks across modalities [108]. |
| Motion Correction Algorithms | Software algorithms used during data preprocessing to identify and correct for artifacts caused by head or body movements. This is especially important for EEG, which is highly susceptible to such artifacts, even in movement-tolerant fNIRS setups [108]. |
| Joint Independent Component Analysis (jICA) | A data fusion technique used after separate preprocessing of EEG and fNIRS. jICA identifies hidden underlying components that are statistically independent and shared across the two modalities, revealing coupled electrical and hemodynamic brain networks [108]. |
| Cross-Modal Attention (in Transformers) | An AI model mechanism that allows one modality to "attend to" relevant information in another. For example, when analyzing a fused EEG-fNIRS dataset, the model can learn to weight specific fNIRS features more heavily when a particular EEG pattern is detected [109]. |
Conclusion
The choice between EEG, fNIRS, and fMRI is not a matter of identifying a superior technology, but of strategically matching a modality's temporal (and spatial) resolution profile to the specific research or clinical question. EEG remains unparalleled for capturing neural events on a millisecond scale, fNIRS offers a robust balance for studying sustained cortical processes in naturalistic settings, and fMRI provides unmatched whole-brain spatial mapping for deeper structures. The future of neuroimaging lies not in the isolation of these techniques, but in their intelligent integration. Multimodal approaches, particularly portable systems like combined EEG-fNIRS, are poised to overcome the inherent limitations of single modalities. For drug development and clinical research, this synergy enables a more holistic view of brain function, from rapid electrophysiological changes to slower metabolic responses, ultimately accelerating the path to novel diagnostics and therapeutics.
References
- 1. fNIRS vs EEG: Comparing Non-Invasive Brain Imaging ... [https://imotions.com/blog/learning/research-fundamentals/fnirs-vs-eeg/?srsltid=AfmBOopNQ5YZjtAtXppWq1xqTXfYHwMyott-rBek2Z-E6CihEBwN4oOo]
- 2. Applications and advances of combined fMRI-fNIRs ... [https://www.frontiersin.org/journals/neurology/articles/10.3389/fneur.2025.1542075/full]
- 3. Comparison of fNIRS with other neuroimaging modalities [https://www.artinis.com/blogpost-all/comparison-fnirs-vs-fmri]
- 4. Current implications of EEG and fNIRS as functional ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11629311/]
- 5. Simultaneous EEG-fNIRS study of visual cognitive processing [https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0325017]
- 6. Simultaneous EEG and fNIRS recordings for semantic ... [https://www.nature.com/articles/s41597-025-04967-0]
- 7. Measuring Mental Workload with EEG+fNIRS [https://www.frontiersin.org/journals/human-neuroscience/articles/10.3389/fnhum.2017.00359/full]
- 8. Multimodal fNIRS–EEG sensor fusion: Review of data-driven ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12592382/]
- 9. EEG–fNIRS signal integration for motor imagery ... [https://www.sciencedirect.com/science/article/pii/S2772528625000299]
- 10. The applied principles of EEG analysis methods in ... [https://mmrjournal.biomedcentral.com/articles/10.1186/s40779-023-00502-7]
- 11. fNIRS vs EEG: Comparing Non-Invasive Brain Imaging ... [https://imotions.com/blog/learning/research-fundamentals/fnirs-vs-eeg/?srsltid=AfmBOoqW_bMHetB6k03LnJ_gbXu5dcbjmL10g7z3VTGgd9e25DWi-yW5]
- 12. A Cross-Disciplinary Review of the fNIRS-EEG Dual-Modality ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11506513/]
- 13. What is the difference between fMRI, EEG and TD-fNIRS? [https://docs.kernel.com/docs/what-is-the-difference-between-fmri-eeg-and-td-fnirs]
- 14. 5 Basics of EEG 101: Data Collection, Processing & Analysis [https://imotions.com/blog/learning/best-practice/5-basics-eeg-data-processing/?srsltid=AfmBOorzxcnrL6vWWc3ZdKkSAp0BJF-7fhUVD2zmc8YCfjTUnh5jzwnE]
- 15. What is the optimal combination of text and background ... [https://psychology.stackexchange.com/questions/6234/what-is-the-optimal-combination-of-text-and-background-color-for-eeg-experiments]
- 16. Methods of EEG Signal Features Extraction Using Linear ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4045570/]
- 17. Which experimental designs to use in fNIRS — Introduction [https://www.artinis.com/blogpost-all/fnirs-experimental-designs-introduction]
- 18. fNIRS vs EEG: Comparing Non-Invasive Brain Imaging ... [https://imotions.com/blog/learning/research-fundamentals/fnirs-vs-eeg/?srsltid=AfmBOoqhOXgezrqD64u5nS7UVQpjZMZoAoouCXP0bdSsnDVQucnpeM5i]
- 19. fNIRS in the developmental sciences - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC4979552/]
- 20. Simultaneous multimodal fNIRS-EEG recordings reveal ... [https://www.nature.com/articles/s41598-023-31609-5]
- 21. a simultaneous fNIRS-fMRI study [https://pmc.ncbi.nlm.nih.gov/articles/PMC9896013/]
- 22. How and when the fMRI BOLD signal relates to underlying ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3546820/]
- 23. The Underpinnings of the BOLD Functional Magnetic ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6741096/]
- 24. Evaluating the effects of multimodal EEG-fNIRS ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12425317/]
- 25. Apparent Diffusion Coefficient fMRI shines light on white ... [https://www.nature.com/articles/s42003-025-07889-0]
- 26. Physiological modeling of the BOLD signal and ... [https://www.sciencedirect.com/science/article/pii/S1053811923004007]
- 27. fNIRS vs EEG: Comparing Non-Invasive Brain Imaging ... [https://imotions.com/blog/learning/research-fundamentals/fnirs-vs-eeg/?srsltid=AfmBOopL5U0t8Tyg9uWKuv_1ZMvTllc3OYcZ_ZlmtNh6sdAFQIdkxk1H]
- 28. fNIRS, EEG, ECG, and GSR reveal an effect of complex ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12171126/]
- 29. Comparing structure–function relationships in brain ... [https://www.nature.com/articles/s41598-024-79817-x]
- 30. Enhanced spatiotemporal resolution imaging of neuronal ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7778454/]
- 31. fNIRS vs EEG: Comparing Non-Invasive Brain Imaging ... [https://imotions.com/blog/learning/research-fundamentals/fnirs-vs-eeg/?srsltid=AfmBOoqb7aNS1nbBXYnQGR8vJCbfwvdaFWqv69C7BvSpO-4x5h5amZJx]
- 32. Clinical applications of EEG as an excellent tool for event ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9123476/]
- 33. Event-Related Potential - an overview [https://www.sciencedirect.com/topics/neuroscience/event-related-potential]
- 34. Annotated interictal discharges in intracranial EEG sleep ... [https://www.nature.com/articles/s41597-024-04187-y]
- 35. Methodological considerations related to sleep paradigm ... [http://www.scielo.cl/scielo.php?script=sci_arttext&pid=S0716-97602008000300004]
- 36. fNIRS vs EEG: Comparing Non-Invasive Brain Imaging ... [https://imotions.com/blog/learning/research-fundamentals/fnirs-vs-eeg/?srsltid=AfmBOooHPEhoY40aPDPS6fIjNlxXU8bhRJgN5Dgz_NsEOTeGehcN5FQ3]
- 37. fNIRS - fMRI | Concurrent NIRS MRI Recording ... [https://nirx.net/fnirs-fmri]
- 38. Motor Training Using Mental Workload (MWL) With an ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8012849/]
- 39. fNIRS-EEG BCIs for Motor Rehabilitation: A Review [https://www.mdpi.com/2306-5354/10/12/1393]
- 40. Accuracy of functional near-infrared diffuse optical ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12370649/]
- 41. fNIRS vs EEG: Comparing Non-Invasive Brain Imaging ... [https://imotions.com/blog/learning/research-fundamentals/fnirs-vs-eeg/?srsltid=AfmBOopcQHRTZAjlSu68h9bf-bxcPVzxf3yHke6GyRpTJ4Vu_UzLwG3F]
- 42. Estimating Brain Activity with High Spatial and Temporal ... [https://arxiv.org/html/2510.09415v1]
- 43. Applications and advances of combined fMRI-fNIRs ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11958174/]
- 44. Simultaneous EEG and fNIRS recordings for semantic ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11993746/]
- 45. Functional connectivity of thalamic nuclei during ... [https://www.frontiersin.org/journals/neuroscience/articles/10.3389/fnins.2025.1568222/full]
- 46. Within-individual precision mapping of brain networks ... [https://www.sciencedirect.com/science/article/pii/S0896627325006646]
- 47. The history, current state and future possibilities of the non- ... [https://www.sciencedirect.com/science/article/pii/S2590093525000049]
- 48. State-of-the-Art on Brain-Computer Interface Technology [https://pmc.ncbi.nlm.nih.gov/articles/PMC10346878/]
- 49. Exploring Imagined Movement for Brain–Computer Interface ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12468679/]
- 50. EEG-based brain-computer interface enables real-time ... [https://www.nature.com/articles/s41467-025-61064-x]
- 51. Improved performance of fNIRS-BCI by stacking of deep ... [https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0314447]
- 52. STA-Net: Spatial–temporal alignment network for hybrid ... [https://www.sciencedirect.com/science/article/abs/pii/S156625352500096X]
- 53. HEFMI-ICH: a hybrid EEG-fNIRS motor imagery dataset for ... [https://www.nature.com/articles/s41597-025-06100-7]
- 54. Brain Computer Interfaces 2025-2045: Technologies, ... [https://www.idtechex.com/en/research-report/brain-computer-interfaces/1024]
- 55. fNIRS vs EEG: Comparing Non-Invasive Brain Imaging ... [https://imotions.com/blog/learning/research-fundamentals/fnirs-vs-eeg/?srsltid=AfmBOooWSlUiIaoxGzxC2M9Jdojt5EBP-BwonkcToii91-at4oqf3hpj]
- 56. Concurrent fNIRS and EEG for Brain Function Investigation [https://pmc.ncbi.nlm.nih.gov/articles/PMC9371171/]
- 57. Applications of near-infrared spectroscopy in epilepsy, with ... [https://www.sciencedirect.com/science/article/pii/S1878747924000096]
- 58. A Cross-Disciplinary Review of the fNIRS-EEG Dual ... [https://www.mdpi.com/2076-3425/14/10/1022]
- 59. Applications of Functional Near-Infrared Spectroscopy (fNIRS ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12347915/]
- 60. fNIRS vs EEG: Comparing Non-Invasive Brain Imaging ... [https://imotions.com/blog/learning/research-fundamentals/fnirs-vs-eeg/?srsltid=AfmBOoqyZXx34HHTDqOwcQBe9loiaWmh4uCz9yxTioedaN0jJlJd_y-8]
- 61. Environmental effects on inter-brain coupling: a systematic ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12351501/]
- 62. Brain benefits of deep learning-based noise management ... [https://www.nature.com/articles/s41598-025-25801-y]
- 63. Comparison of motion correction techniques applied to ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9900395/]
- 64. fNIRS vs EEG: Comparing Non-Invasive Brain Imaging ... [https://imotions.com/blog/learning/research-fundamentals/fnirs-vs-eeg/?srsltid=AfmBOooZzbbiBReJ4zudkgUvmR3MN7IPnUQg-XmEGgnutu5-445kxAev]
- 65. A Comparison of Approaches for Motion Artifact Removal from ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12349273/]
- 66. Eye-movement artifact correction in infant EEG [https://www.sciencedirect.com/science/article/abs/pii/S0165027025000469]
- 67. Comparing different pre-processing routines for infant fNIRS ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7985709/]
- 68. fNIRS: Non-stationary preprocessing methods [https://www.sciencedirect.com/science/article/pii/S1746809422005675]
- 69. Quantitative comparison of correction techniques for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7511246/]
- 70. How EEG preprocessing shapes decoding performance [https://www.nature.com/articles/s42003-025-08464-3]
- 71. Frontiers | Editorial: Multimodal approaches to investigating neural... [https://www.frontiersin.org/journals/systems-neuroscience/articles/10.3389/fnsys.2025.1495018/full]
- 72. vs. EEG vs. MRI - What are the Differences? - iMotions fMRI [https://imotions.com/blog/learning/research-fundamentals/eeg-vs-mri-vs-fmri-differences/]
- 73. fNIRS vs EEG: Comparing Non-Invasive Brain Imaging ... [https://imotions.com/blog/learning/research-fundamentals/fnirs-vs-eeg/?srsltid=AfmBOooR5CAjZ4veWqtdk9aGFLUxu8FPO6EkK7kah64qIos8H_cTr_wK]
- 74. Medical Science Monitor | Functional Near-Infrared Spectroscopy in... [https://medscimonit.com/abstract/full/idArt/949491]
- 75. Investigating the effective temporal resolution in a task- ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12290576/]
- 76. Dynamics of task-related electrophysiological networks [https://www.sciencedirect.com/science/article/pii/S1053811921001063]
- 77. The relationship between electrophysiological and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9669574/]
- 78. Early Detection of Hemodynamic Responses Using EEG [https://www.frontiersin.org/journals/human-neuroscience/articles/10.3389/fnhum.2018.00479/full]
- 79. fNIRS vs EEG: Comparing Non-Invasive Brain Imaging ... [https://imotions.com/blog/learning/research-fundamentals/fnirs-vs-eeg/?srsltid=AfmBOopp_gsuTBYv3eC3q8PE1n-Qzd3nLq-viIVmqiYBacL18loK5U3q]
- 80. Using the General Linear Model to Improve Performance in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7040364/]
- 81. Improving the SNR of EEG generated by deep sources with ... [https://pubmed.ncbi.nlm.nih.gov/19619646/]
- 82. Optimizing spatial specificity and signal quality in fNIRS [https://www.frontiersin.org/journals/neuroergonomics/articles/10.3389/fnrgo.2024.1286586/full]
- 83. Smooth dynamic T 2 * mapping in fMRI based on a novel ... [https://www.frontiersin.org/journals/neuroscience/articles/10.3389/fnins.2025.1544748/full]
- 84. The ecological validity of laboratory experiments in ... [https://www.sciencedirect.com/science/article/abs/pii/S0003682X25000544]
- 85. The 'Real-World Approach' and Its Problems: A Critique of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7204431/]
- 86. Opportunities and Limitations of Mobile Neuroimaging ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9292610/]
- 87. A direct comparison of laboratory and community EEG ... [https://www.nature.com/articles/s41598-025-03569-5]
- 88. Functional near infrared spectroscopy (NIRS) signal ... [https://www.sciencedirect.com/science/article/abs/pii/S105381190901235X]
- 89. Characterizing the Action-Observation Network Through ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7930074/]
- 90. Conducting Psychology Research in the Real World [https://nobaproject.com/modules/conducting-psychology-research-in-the-real-world]
- 91. The application of fNIRS in studies on occupational workload [https://pmc.ncbi.nlm.nih.gov/articles/PMC12053328/]
- 92. Sensitive and specific fNIRS-based approach for awareness ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12521986/]
- 93. fNIRS vs EEG: Comparing Non-Invasive Brain Imaging ... [https://imotions.com/blog/learning/research-fundamentals/fnirs-vs-eeg/?srsltid=AfmBOop25yVZxr7_cuZLmQonuONjM8o7dpjYiYUG3T1lA_cCNrGzH0ZZ]
- 94. Enhancing Performance of a Hybrid EEG-fNIRS System ... [https://www.frontiersin.org/journals/human-neuroscience/articles/10.3389/fnhum.2017.00462/full]
- 95. A multi-modal dataset of electroencephalography and ... [https://www.nature.com/articles/s41597-025-05286-0]
- 96. Multimodal fNIRS-EEG sensor fusion: Review of data- ... [https://pubmed.ncbi.nlm.nih.gov/41211102/]
- 97. A Review of Data Fusion Techniques - PMC - PubMed Central [https://pmc.ncbi.nlm.nih.gov/articles/PMC3826336/]
- 98. fNIRS vs EEG: Comparing Non-Invasive Brain Imaging ... [https://imotions.com/blog/learning/research-fundamentals/fnirs-vs-eeg/?srsltid=AfmBOoq2qxJH_7FH_WkPhZHBZ8s9CN_HGo0doL-gfFF3qTtQOvV3BeBy]
- 99. A Comparative Analysis of Three Data Fusion Methods and ... [https://www.mdpi.com/2227-7390/13/8/1218]
- 100. Multimodal fusion of EEG-fNIRS: a mutual information-based ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7984774/]
- 101. Model-based material decomposition with a penalized ... - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC6385868/]
- 102. Improved classification performance of EEG-fNIRS ... [https://www.frontiersin.org/journals/human-neuroscience/articles/10.3389/fnhum.2022.973959/full]
- 103. Connector based short time series prediction [https://www.nature.com/articles/s41598-024-83122-y]
- 104. fNIRS vs EEG: Comparing Non-Invasive Brain Imaging ... [https://imotions.com/blog/learning/research-fundamentals/fnirs-vs-eeg/?srsltid=AfmBOooZpJrjNWLn63qpDmK7h_Qb3RLT8T1jLUG-2kLmmFQKGgMh82Jt]
- 105. Multimodal neuroimaging computing: a review of the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4737664/]
- 106. Opportunities for use of neuroimaging in de-risking drug ... [https://www.nature.com/articles/s41386-024-01970-8]
- 107. Enhanced performance by a hybrid NIRS–EEG brain ... [https://www.sciencedirect.com/science/article/pii/S1053811911008792]
- 108. fNIRS vs EEG: Comparing Non-Invasive Brain Imaging ... [https://imotions.com/blog/learning/research-fundamentals/fnirs-vs-eeg/?srsltid=AfmBOoq3H8EjOpOLMwgGMvE3upmGjQDVvjrFoFlgJkGMyHwHtLqiPcXw]
- 109. How does multimodal AI handle temporal data? [https://milvus.io/ai-quick-reference/how-does-multimodal-ai-handle-temporal-data]